UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
FORM 10-K
(Mark One)
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2025
or
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the transition period from to
COMMISSION FILE NO. 000-26224
(EXACT NAME OF REGISTRANT AS SPECIFIED IN ITS CHARTER)
| (STATE OR OTHER JURISDICTION OF INCORPORATION OR ORGANIZATION) | (I.R.S. EMPLOYER IDENTIFICATION NO.) | |||||||||||||
| , | (ZIP CODE) | |||||||||||||
| (ADDRESS OF PRINCIPAL EXECUTIVE OFFICES) | ||||||||||||||
REGISTRANT’S TELEPHONE NUMBER, INCLUDING AREA CODE: (609 ) 275-0500
SECURITIES REGISTERED PURSUANT TO SECTION 12(b) OF THE ACT:
| Title of Each Class | Trading Symbol | Name of Exchange on Which Registered | ||||||
SECURITIES REGISTERED PURSUANT TO SECTION 12(g) OF THE ACT:
NONE
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act.
Yes ☐ No ☒
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
1
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| ☒ | Accelerated filer | ☐ | |||||||||
| Non-accelerated filer | ☐ | Smaller reporting company | |||||||||
| Emerging growth company | |||||||||||
If an emerging growth company, indicate by check if the registrant has elected not to use the extended transition period for complying with any new revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☒
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to § 240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
As of June 30, 2025, the aggregate market value of the registrant’s common stock held by non-affiliates was approximately $925.1 million based upon the closing sales price of the registrant’s common stock on The Nasdaq Global Select Market on such date. The number of shares of the registrant’s Common Stock, $0.01 par value, outstanding as of February 25, 2026 was 77,937,208 .
DOCUMENTS INCORPORATED BY REFERENCE:
2
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
TABLE OF CONTENTS
| Page | |||||
Unless otherwise stated or the context otherwise indicates, all references in this Annual Report on Form 10-K to “Integra LifeSciences,” “Integra,” “the Company,” “we,” “our,” and “us” refer to Integra LifeSciences Holdings Corporation, a Delaware corporation and its consolidated subsidiaries.
3
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
We have made statements in this report, including statements under “Business” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” that constitute forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, (“the Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). These forward-looking statements are subject to a number of risks, uncertainties and assumptions about us including, among other things:
•the on-going and possible future effects of global challenges, including macroeconomic uncertainties, such as supply chain disruptions, inflation, bank failures, high interest rates and availability of capital markets, geopolitical uncertainty and instability, the spread or escalation of wars and other armed conflict, other economic disruptions and U.S. and global recession concerns, on our customers and suppliers, and on our business, financial condition, results of operations and cash flows;
•general economic and business conditions, both domestically and in our international markets, including the effect of the continuing worldwide macroeconomic uncertainty and increasing trade regulations and tariffs;
•our expectations and estimates concerning future financial performance, financing plans and the impact of competition;
•trends in our business;
•demand for our products, including capital equipment;
•our ability to produce and deliver products in sufficient quantities to meet sales demands;
•our failure to comply with the substantial regulation related to quality standards applicable to our manufacturing and quality processes could have an adverse effect on our business, financial condition, or results of operations;
•our ability to remediate all matters identified in the United States Food and Drug Administration (the “FDA”) observations and warning letters that we received or may receive and resume the manufacture and sale of certain of our products identified in such observations and warnings letters;
•the ongoing and possible future effects of supply chain constraints, including the availability of critical raw materials and components, as well as cost inflation in materials, packaging and transportation;
•our expectations concerning our ongoing restructuring, integration and manufacturing transfer and expansion activities;
•existing and future regulations affecting our business, and enforcement of those regulations;
•conducting business internationally;
•our ability to obtain additional debt and equity financing, or to refinance our existing outstanding indebtedness, to fund capital expenditures, working capital requirements and acquisitions;
•physicians’ willingness to adopt our recently launched and planned products, third-party payors’ willingness to provide or continue reimbursement for any of our products and our ability to secure regulatory approval for products in development;
•initiatives launched by our competitors;
•our ability to protect our intellectual property, including trade secrets;
•our ability to complete acquisitions, integrate operations post-acquisition and maintain relationships with customers of acquired entities;
•the ability to successfully manage ongoing organizational and strategic changes, including our ability to attract, motivate and retain key employees and maintain engagement and efficiency in remote work environments;
•trends relating to our financial condition or results of operations, including the impact of interest rate and foreign currency exchange fluctuations; and
•other risk factors described in Item 1A. “Risk Factors” in this Annual Report on Form 10-K.
Forward-looking statements can be identified by forward-looking words such as “believe,” “may,” “could,” “might,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “seek,” “plan,” “expect,” “should,” “would,” “expect,” “target,” “pursue,” “forecast,” “hope” and similar expressions in this Annual Report on Form 10-K. Forward-looking statements in this Annual Report on Form 10-K include, but are not limited to, statements regarding our growth strategy; future business, operating and financial performance; our expectations and plans with respect to market opportunity, business and operational performance, strategic initiatives, capabilities, resources, manufacturing, product development, product availability and regulatory approvals, including expectations regarding our compliance master plan to improve our quality systems; our hope to secure PMA approval for SurgiMend and DuraSorb in 2026; our business strategy and plans, including plans to deliver future innovation both within the ENT business and across other CSS technology platforms; and our expectations regarding the operationalization of the Braintree facility and the timing thereof. These statements are only predictions and involve known and unknown risks, uncertainties, and other factors that may cause our actual results, levels of activity, performance, or achievements to be materially different from any future results, levels of activity, performance, or achievements expressed or implied by such forward-looking statements. Given these uncertainties, you should not place undue reliance on these forward-looking
4
statements. Also, these forward-looking statements represent our estimates and assumptions only as of the date of this report. Except as required by law, we undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. Factors that could cause or contribute to differences in our future financial results include the cautionary statements set forth herein and in our other filings with the Securities and Exchange Commission, including those set forth under “Risk Factors” set forth in Part I, Item 1A of this Annual Report on Form 10-K. We qualify all of our forward-looking statements by these cautionary statements.
5
PART I
ITEM 1. BUSINESS
OVERVIEW
Integra LifeSciences Holdings Corporation is a global medical technology company dedicated to restoring lives. We are advancing transformational care through impactful innovation and our portfolio of highly differentiated technologies is trusted by healthcare professionals to deliver transformative care.
We manufacture and sell medical technologies and products in two reportable business segments: Codman Specialty Surgical (“CSS”) and Tissue Technologies (“TT”). The CSS segment, which represents approximately 70% of our total revenue, consists of market-leading technologies and instrumentation used for a wide range of specialties, such as neurosurgery, neurocritical care, and otolaryngology, commonly referred to as ear, nose, and throat (“ENT”). We are the world leader in neurosurgery and one of the top three providers in the U.S. in instruments used in precision, specialty, and general surgical procedures. Our TT segment generates about 30% of our overall revenue and focuses on wound reconstruction and care and private label.
OUR PRODUCTS, SERVICES AND TECHNOLOGIES
We were the first company to receive an FDA claim for regeneration of dermal tissue and are a world leader in regenerative technology. We have developed numerous product lines from this technology for applications ranging from burn and deep tissue wounds to the repair of dura mater in the brain, as well as nerves and tendons. We have expanded our base regenerative technology business to include neurosurgical products, ENT, surgical instruments and advanced wound care through global acquisitions and product development to meet the evolving needs of our customers and enhance patient care. Each of these categories and the key products sold therein are described in more detail below. We include financial information regarding our reportable business segments and certain geographic information under “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” and Note 16. Segment and Geographic Information to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
CODMAN SPECIALTY SURGICAL
Neurosurgery: In neurosurgery, we are a global leader in neuro-access, neuro-surgical and neuro-monitoring technologies. Our product portfolio represents a continuum of care from pre-operative, to the neurosurgery operating room, to the neuro-critical care unit and post care for both adult and pediatric patients suffering from brain tumors, brain injury, cerebrospinal fluid pressure complications and other neurological conditions
We offer leading technologies in dural repair, ultrasonic tissue ablation, intracranial pressure (“ICP”) monitoring, hydrocephalus management, and cranial stabilization systems, while providing a rich research and development pipeline for growth.
Principal products include:
•CUSA® (Cavitron Ultrasonic Surgical Aspirator) Platform: An ultrasonic surgical aspirator platform, including the CUSA Clarity, which is used in various surgical procedures, primarily for the fragmentation, emulsification, and aspiration of soft and hard tissues;
•CereLink® intercranial pressure (“ICP”) Monitor: An advanced, digital intracranial pressure monitoring platform, designed for use in neuro-intensive care and operating room environments, which offers uncompromised advanced continuous ICP monitoring — with minimal drift, MRI conditional capability, durable, flexible ICP sensors, and advanced data presentation features;
•Codman® Neurosurgical Accessories: A portfolio of surgical consumables for use in the protection of tissue, including brain and other tissues of the central nervous system, during surgery and disposable perforators for use in perforating the cranium;
•DuraGen® Dural Graft Matrix: An absorbable implant for sutureless repair of dural defects after cranial or spinal surgery and which acts as an onlay dural graft which provide a highly porous scaffold for the purpose of patching large gaps in the dura and facilitating patient recovery;
•DuraSeal® Dural Sealant System: An absorbable polyethylene glycol (“PEG”) hydrogel designed to provide a watertight seal, particularly around suture lines or at the edge of an onlay graft, for cranial and spinal surgery patients;
•Mayfield® Standard Cranial Stabilization System: A surgical device used to stabilize the patient’s head during neurosurgical procedures;
6
•Bactiseal® Anti-Microbial Catheters: Antimicrobial external ventricular drainage (“EVD”) catheters and shunts designed to allow for the draining or shunting of cerebrospinal fluid (“CSF”) while reducing the risk of surgical site infections; and
•Certas® Plus Programmable Valve: A programable shunt valve which is designed to provide constant intraventricular pressure and drainage of CSF for the management of hydrocephalus
Specialty Instrumentation: Our specialty instrumentation portfolio includes a catalog of surgical headlamps, surgical instruments, as well as after-market service. With thousands of surgical instrument products, comprised of a comprehensive portfolio of reusable and disposable instruments, including forceps, retractors, scissors, and curettes, tailored for neurosurgery and spine surgery including specialty surgical instruments, we call on the central sterile processing unit of hospitals and acute care surgical centers. Additionally, through a strong U.S. distribution model, we can serve the needs of medical offices.
ENT Surgical Solutions: The Company’s ENT portfolio comprises a broad range of products including the TruDi® Navigation System featuring navigated surgical instrumentation, the RELIEVA SPINPLUS® Balloon Sinuplasty System, the AERA® Eustachian Tube Dilation System, and the MicroFrance ENT instrumentation line.
TISSUE TECHNOLOGIES
Our Tissue Technologies segment develops and markets a broad portfolio of regenerative tissue products and technologies primarily focused on wound reconstruction and care and private label. This segment serves a diverse range of specialties, including plastic and reconstructive surgery, general surgery, and wound management.
We currently utilize five unique regenerative technology platforms consisting of highly engineered bovine collagen (derived from bovine sources for structural support), bovine dermis (acellular dermal tissue for natural integration), porcine urinary bladder (extracellular matrix for cellular repopulation), human amniotic tissue (allografts for anti-inflammatory and protective properties), and resorbable synthetic mesh (biodegradable materials for temporary reinforcement). These technologies address clinical needs in treating acute wounds such as burns, chronic wounds including diabetic foot ulcers, and surgical tissue repair applications such as hernia reinforcement, tendon protection, and peripheral nerve repair.
Wound Reconstruction and Care: We offer a broad portfolio of products and solutions addressing a full spectrum of needs when managing complex wounds including for the treatment of acute wounds (e.g., burns and trauma), chronic wounds (e.g., diabetic foot ulcers and venous ulcers), and other complex wound types. The goal is to promote healing by providing scaffolds that encourage cellular invasion, revascularization, and tissue regeneration while managing infection, exudate, and inflammation. These products are often used in hospital settings, wound care clinics, or outpatient procedures to accelerate closure, reduce scarring, and prevent complications like amputation in diabetic cases.
Principal products include:
•Integra Skin: Comprised of the first FDA-approved bilayer matrix for the regeneration of dermal tissue, indicated for third-degree burns and scar contractures, which consists of a collagen and chondroitin-6-sulfate matrix with a silicone epidermal layer to protect against infection and dehydration and several other bilayer wound matrices used for a variety of wound types, providing a scaffold for cellular invasion and capillary growth, often used with negative pressure wound therapy;
•PriMatrix® Dermal Repair Scaffold: A collagen-based dermal repair scaffold for managing a broad range of wounds, including diabetic and venous ulcers;
•MicroMatrix® Urinary Bladder Matrix (“UBM”) and Cytal® Wound Matrix: These products use porcine urinary bladder matrix technology to support complex wound management;
•AmnioExcel® and AmnioExcel® Plus Placental Allograft Membrane: Human tissue-based allografts in various surgical and chronic wound applications; and
•MediHoney® Wound and Burn Dressings: Medical-grade Manuka honey-based dressings used for debridement and moisture balance in burns, ulcers, and surgical wounds.
We offer a continuum of advanced solutions for plastic and reconstructive surgery, complex hernias, and general surgery, including devices for implantation to reinforce soft tissue where weakness exists and for surgical repair of damaged or ruptured soft tissue membranes. These products provide structural support and promote tissue integration to address areas of tissue weakness. They are bioresorbable or acellular to minimize long-term foreign body reactions and reduce complications like adhesions or infections.
7
Principal products include:
•SurgiMend® Collagen Matrix: An implant for soft tissue reinforcement in plastic and reconstructive surgery, including hernia repair. It is derived from bovine collagen and is designed to integrate with host tissue while minimizing complications;
•Gentrix® Surgical Matrix: Porcine UBM matrix for hernia (including hiatal) and abdominal wall repair; and
•DuraSorb® Monofilament Mesh: A resorbable mesh used in reconstructive surgery to provide temporary support during tissue healing, particularly in hernia repair and other soft tissue applications.
Private Label: We offer extensive expertise in collagen biomaterials for other medical technology companies that sell to end markets primarily in spine, surgical and wound care. We manufacture a broad set of our regenerative and wound care technologies that are available for private label distribution by our customers, produce raw materials that can be integrated into our customers’ production processes, and have the expertise to design, develop and manufacture products to meet the specific needs of our customers.
OUR MARKETS
We sell our medical devices and therapies through a combination of direct sales representatives and independent distributors globally. Our global commercial network includes clinical specialists, direct sales force in key markets and strategic partnerships and distributors that serve hospitals, integrated health networks, group purchasing organizations, clinicians, surgery centers and health care providers.
Our marketing and sales strategy is focused on cost-effective delivery of high-quality products to a diverse group of customers worldwide. To achieve this objective, our direct global sales force teams are organized around physician specialties. This focus enables us to develop highly knowledgeable and dedicated sales representatives who are able to foster strong relationships with physicians and enhance our ability to support our customers and cross-sell complementary products.
Over the years, we have significantly expanded our global footprint by investing in our commercial and manufacturing operations, as well as introducing new products. In the last two years, we have expanded our presence in key markets such as Southeast Asia, India, South Korea and Brazil. As part of our In-China-For-China strategy, we continue to build out our assembly capabilities in our facility in Suzhou, China.
We did not have any customers that represented more than 10% of our consolidated revenues in 2025.
RESEARCH AND DEVELOPMENT STRATEGY
An important part of Integra’s growth strategy is introducing new products to strengthen and expand our portfolio through clinical evidence to support regulatory approval and strong reimbursement of our product portfolio around the world, including new indications for existing technologies. Our research and development activities focus on identifying unmet surgical needs and addressing those needs with innovative solutions and products. Investment in research and development is critical to driving our future growth. Our research and development efforts are focused on the further development and improvement of our existing products, the design and development of new innovative medical technologies and regulatory compliance across all our business segments. We apply our core competency in regenerative technology to innovate products for neurosurgical, wound applications, plastic surgery, and reconstructive surgery and we have extensive R&D development programs for our core platforms of electromechanical technologies.
Additionally, we conduct projects and clinical studies to generate efficacy and health economic evidence. The Company has continued its investments in clinical education as a key value driver to leverage its global footprint, enhanced digital content, and strengthened its clinical network. As part of this objective, the Company remains committed to participation in clinical research demonstrating the efficacy of its products prior to market introduction, and in supporting the clinical education and technical training.
Neurosurgical Solutions, Surgical Instruments, and ENT Solutions. The CSS neurosurgical business consists of a broad portfolio of market-leading brands, which are used for the management of multiple disease states, including brain tumors, traumatic brain injury, hydrocephalus and other neurological conditions. The growth in this business in recent years has been fueled by geographic expansion and new product registrations in markets, such as China, Japan, and Europe, which we expect to continue in the near-to-long term. We have several active programs focused on life cycle management and innovation for capital and disposable products in our portfolio. Our product development efforts are focused on core clinical applications in cerebrospinal fluid (“CSF”) management, neuro-critical care monitoring, minimally invasive instruments and electrosurgery and ultrasonic medical technologies, as well as our ambition to transform the standard of care in neurosurgery with product advancements in minimally invasive surgery (“MIS”) and the surgical management of intracerebral hemorrhage (“ICH”).
We continue to advance the CerebroFlo® external ventricular drainage (“EVD”), a catheter with Endexo® technology. The Endexo polymer in polyurethane is a permanent additive which has shown to be effective in reducing platelet adhesion in-vitro,
8
reducing thrombus accumulation in-vitro and in vivo, and reducing the clinical incidence of thrombus formation. In vitro evaluations and in vivo animal evaluations do not necessarily predict the clinical performance of the SureFlo EVD Catheter with respect to thrombus formation. The incidence of thrombus formation on polyurethane containing Endexo polymer in other medical devices and/or tissues systems does not necessarily predict the clinical performance of the SureFlo EVD Catheter for the intended use of CSF external drainage and monitoring. The CerebroFlo EVD catheter has demonstrated an average of 99% less thrombus accumulation onto its surface, in vitro, compared to a market leading EVD catheter. Our work to combine our Bactiseal antimicrobial technology with the Endexo anti-occlusive technology continues to progress for both a silicone-based hydrocephalus and EVD product.
We also continue to advance the Aurora® Surgiscope, which is the only tubular retractor system designed for cranial surgery with an integrated access channel, camera and lighting. The 15mm x 60mm and 15mm x 80mm Aurora Surgiscope System version received 510(k) clearance from the FDA in 2025.
In July 2025, we announced the inaugural enrollment of the first patient in the AERA Pediatric Registry, a prospective, multi-center observational registry evaluating the real-world use of the AERA Eustachian Tube Balloon Dilation System in children. This marks the focused effort to measure the ongoing, clinical performance of AERA in pediatric patients with obstructive Eustachian tube dysfunction. The registry is designed to capture both safety and efficacy outcomes for up to 300 pediatric patients who undergo Eustachian tube balloon dilation using AERA.
In September 2025 the Mayfield® Ghost Base Unit Post launched in the U.S., which is designed to help provide clear visualization of anatomical structure and to support surgical accuracy and patient positioning.
Regenerative Technologies. Our regenerative technology development program applies our expertise in bioengineering to a range of biomaterials including natural materials such as purified collagen, intact human or animal tissues, honey as well as resorbable synthetic polymers with our DuraSorb and DuraSeal® product lines. These unique product designs are used for neurosurgical and reconstructive surgical applications, as well as dermal regeneration. Our regenerative technology platform includes our legacy Integra® Dermal Regeneration Template (“IDRT”) products and complementary technologies that we have acquired. Our collagen manufacturing capability, combined with our history of innovation, provides us with strong platform technologies for multiple indications.
In the third quarter of 2021, we filed a PMA application for a specific indication for SurgiMend® in the use of post-mastectomy breast reconstruction and in July 2024 received approvable pending GMP status from FDA, which approved and closed out the clinical portion of this PMA application. We anticipate PMA approval following the operationalization of the Braintree facility, which is expected in 2026. We are also pursuing a PMA for DuraSorb for use in implant-based breast reconstruction (“IBBR”). We completed enrollment for the DuraSorb U.S. investigational device exemption clinical study for two-stage breast reconstruction in June 2023; and we continue to advance the PMA application. Currently, we hope to secure PMA approval for DuraSorb in 2026.
In 2024, we acquired the product rights for Durepair Dural Regeneration Matrix, a suturable dural graft which complements our portfolio of dural grafts and sealants, and subsequently launched the product for commercial sale in the U.S. in October 2025.
See Item 1A. Risk Factors, under the heading Risks Related to our Regulatory Environment and under Item 7. General Management’s Discussion and Analysis of Financial Condition and Results of Operations - FDA Matters of this Annual Report on Form 10-K for further discussion.
QUALITY SYSTEMS, SUPPLY CHAIN AND PRODUCT MANUFACTURE
We have been making investments to build more responsive and scalable processes, enhance the reliability of our quality systems and supply chain, and drive productivity initiatives to further supply and lower costs. We continue to invest in technologies, systems and processes to enhance the customer experience. We also continue to invest in our capacity expansion. This includes ongoing projects of transferring our Boston manufacturing to a new location in Braintree, Massachusetts and further investing in capacity and validating manufacturing processes in our manufacturing facility in Plainsboro, New Jersey.
In 2024, we initiated a Compliance Master Plan (the “CMP”), a systematic and holistic approach to improving our quality management system, or QMS, across our manufacturing and supply network. The primary objectives of the CMP are to remediate quality system gaps, harmonize the quality management system across the company, and enhance the quality culture across our organization. In executing our remediation plans, we have introduced objective metrics to assess opportunities in our QMS and supply chain which we believe has allowed for the effective allocation of resources and consistent tracking of our results.
In conjunction with our work under the CMP, we are focused on improving operational and execution excellence. Through leadership appointments and the development of a comprehensive plan to establish a robust end-to-end supply chain, we hope to enhance our supply chain capabilities and overall product available and operational reliability and resiliency.
9
We have key manufacturing and research facilities located in California, Maryland, Massachusetts, New Jersey, Ohio, Puerto Rico, Tennessee, Utah, France, Germany, Ireland, Israel and Switzerland. We source most of our handheld surgical instruments and dural sealant products through specialized third-party vendors
AVAILABILITY OF RAW MATERIALS
We purchase many of the components and raw materials used in manufacturing our products from numerous suppliers in various countries.
In general, raw materials essential to our businesses are readily available from multiple sources. For reasons of quality assurance, availability, or cost effectiveness, certain components and raw materials are available only from one or a limited number of suppliers. We have established long-term supply contracts with many of our suppliers and our practice is to maintain sufficient inventory of components so that our production will not be significantly disrupted even if a particular component or material is not available for a period of time. Due to the high standards applicable to manufacturing our products, such as the FDA’s Quality Management System Regulation (“QMSR”), which became effective on February 2, 2026, and which superseded the prior Quality System Regulation (“QSR”) and Good Manufacturing Practices, we may not be able to quickly establish additional or replacement sources for certain components or materials. Some of our manufacturing operations are located outside of the U.S., including Switzerland, Ireland, Israel, and France. Those manufacturing operations are also subject to additional challenges and risks associated with international operations described under the caption “Risk Factors” set forth in Part I, Item 1A of this Annual Report on Form 10-K. In the event we are unable to obtain sufficient quantities of raw materials or components on commercially reasonable terms or in a timely manner, our ability to manufacture our products on a timely and cost-competitive basis may be compromised, which may have a material adverse effect on our business, financial condition and results of operations.
Certain of our products, including but not limited to our dermal regeneration products, duraplasty products, wound care products, and nerve and tendon repair products, contain natural collagen material derived from bovine tissue. We take great care to provide medical products that are safe and free of agents that can cause disease. In particular, the collagen used in the products that we manufacture is derived from fetal bovine dermis from the U.S. and deep flexor tendon from the U.S. and New Zealand. New Zealand is a country that has never had a reported case of bovine spongiform encephalopathy (“BSE”) (otherwise known as mad cow disease). The World Health Organization classifies different types of cattle tissue for relative risk of BSE transmission. Fetal bovine dermis and deep flexor tendon are in the lowest-risk category for BSE transmission, and therefore considered to have a negligible risk of containing the agent that causes BSE.
INTELLECTUAL PROPERTY
We seek patent and trademark protection for our key technology, products and product improvements, both in the U.S. and in selected foreign countries. When determined appropriate, we have enforced and plan to continue to enforce and defend our patent and trademark rights. In general, however, we do not rely solely on our patent and trademark estate to provide us with significant competitive advantages as it relates to our existing product lines. We also rely upon trade secrets and continuing technological innovations to develop and maintain our competitive position. In an effort to protect our trade secrets, we have a policy requiring our employees, consultants and advisors to execute proprietary information and invention assignment agreements upon commencement of employment or consulting relationships with us. These agreements also provide that all confidential information developed or made known to the individual during the course of their relationship with us must be kept confidential, except in specified circumstances.
Acclarent Aera®, AccuDrain®, AmnioExcel®, Aquasonic®, Auragen®, Aurora® Surgiscope®, Bactiseal®, BioDFence®, BioDOptix®, Brainet®, Budde®, Buzz™, CereLink®, CerebroFlo® EVD Catheter with Endexo® Technology, Codman®, Codman Accu-Flo®, Codman Bicol®, Codman® Certas® Plus, Codman® Hakim®Programmable valve, Codman Holter®, Codman ICP Express®, Codman Microsensor®, Codman VersaTru®, Codman VPV®, Contour-Flex®, Cranioplastic®, CRW®, CRW Precision™, Ctherm™, CUSA®, Cytal®, DirectLink®, DuraGen®, DuraSeal®, DuraSorb®, Gentrix®, HeliCote®, HeliPlug®, HeliTape®, HeliMend®, Helistat®, Helitene®, Hermetic™, Hy-Tape®, Integra®, IntegraLink®, Isocool®, Jarit®, Lead-Lok™, Licox®, LimiTorr™, Luxtec®, Mayfield®, MatriStem UBM™, MediHoney®, MicroFrance®, MicroMatrix®, Miltex®, Mischler™, MoniTorr ICP™, Natus®, NeuraGen®, NeuraWrap™, Nicolet®, Omnigraft®, Omni-Tract®, OSV II®, Padgett®, PriMatrix®, Pureflow™, Q-Snor™, Redmond™, Relieva Spinplus®, Revize™, Ruggles®, Signacreme®, SurgiMend®, TCC-EZ®, TenoGlide®, TissueMend®, TruDi®, Ultra VS™, VersaTru®, Xtrasorb®, zRIP™, and the Integra logo are some of the material trademarks of Integra LifeSciences Corporation and its subsidiaries. MAYFIELD® is a registered trademark of SM USA, Inc., and is used by Integra under license.
SEASONALITY
Revenues during our fourth quarter tend to be stronger than other quarters because many hospitals increase their purchases of our products during the fourth quarter to coincide with the end of their budget cycles in the U.S. In general, our first quarter usually has lower revenues than the preceding fourth quarter, the second and third quarters have higher revenues than the first
10
quarter, and the fourth quarter revenues are the highest in the year. The main exceptions to this pattern occur because of material acquisitions.
COMPETITION
The markets in which we compete are highly competitive and are characterized by rapid change resulting from technological advances, innovations and scientific discoveries. Our product lines face a mix of competitors ranging from large manufacturers with multiple business lines to small manufacturers offering a limited selection of products. Many of these competitors offer a broader product portfolio and important competitive factors include product efficacy, safety and ease of use, price and demonstrated cost-effectiveness, marketing effectiveness, product labeling, customer service and R&D of new products and processes. Major shifts in industry market share have occurred in connection with product corrective actions, physician advisories, safety alerts, results of clinical trials to support superiority claims, and publications about our products, reflecting the importance of product quality, product efficacy and quality systems in the medical device industry.
Government and private sector initiatives to limit the growth of healthcare costs, including price regulation, competitive pricing, bidding and tender mechanics, coverage and payment policies, comparative effectiveness of therapies, technology assessments and managed-care arrangements, are continuing in many countries where we do business, including the U.S. These initiatives put increased emphasis on the delivery of more cost-effective medical devices and therapies. Government programs, including Medicare and Medicaid, private healthcare insurance, managed-care plans, and volume-based procurement tenders in China, have attempted to control costs by limiting the amount of reimbursement they will pay for particular procedures or treatments, tying reimbursement to outcomes, shifting to population health management, and other mechanisms. Hospitals, which purchase our technology, are also seeking to reduce costs through a variety of mechanisms, including, for example, centralized purchasing, and in some cases, limiting the number of vendors that may participate in the purchasing program. Hospitals are also aligning interests with physicians through employment and other arrangements, such as gainsharing, where a hospital agrees with physicians to share any realized cost savings resulting from changes in practice patterns such as device standardization. This has created an increased level of price sensitivity among customers for our products.
Our competitors for CSS include divisions within Medtronic, Inc., Stryker Corporation, Steris PLC, and B. Braun Medical, Inc. In addition, we compete with many smaller specialized companies and larger companies that do not otherwise focus on the offerings of Codman Specialty Surgical technologies. We rely on the depth and breadth of our sales and marketing organization, our innovative technologies, and our procurement and manufacturing operations to maintain our competitive position.
Our competitors for TT include Smith & Nephew plc, Organogenesis Holdings Inc., MiMedx Group, Inc., Allergan PLC, Becton Dickinson and Company, and Axogen, Inc. We compete with additional companies who partially participate in soft tissue reconstruction of complex wounds and surgical reconstruction. In addition, our products also compete against medical practices that treat a condition without using a medical device or any particular product, such as medical practices that utilize autograft tissue instead of our dermal regeneration products, duraplasty products and nerve repair products. Depending on the product line, we compete based on our products' features, strength of our sales force or distributors, sophistication of our technology and cost effectiveness of our solution.
In addition, our International team faces significant global and regional competition across our entire portfolio, with local competition also playing a role in countries where local suppliers may be advantaged.
We believe that the success of our products depends on our ability to differentiate ourselves and to demonstrate that our products deliver the clinical and operational attributes that are most important and cost-effective to customers. These attributes include, but are not limited to, superiority in efficacy, ease of use, reliability, accuracy, quality and cost. We believe our continued success depends in large part upon our ability to invest in product enhancements and technologies that will help us distinguish our product portfolio from our competitors.
GOVERNMENT REGULATION AND COMPLIANCE
We are a manufacturer and marketer of medical devices and Human Tissue and Cell Based Products (“HCT/Ps”) and therefore are subject to extensive regulation by the FDA, the Centers for Medicare & Medicaid Services (“CMS”) of the U.S. Department of Health and Human Services (“HHS”), other federal governmental agencies and, in some jurisdictions, by state and equivalent foreign governmental authorities. These regulations govern the development and introduction to the market of new medical devices and HCT/Ps, the observance of certain standards with respect to the design, manufacture, testing, labeling, promotion and sales of the products, the maintenance of certain records, the ability to track devices, the reporting of adverse affects and potential product defects, the import and export of products, and other matters. FDA product approvals, as well as equivalent approvals or certifications issued by foreign authorities or bodies, may be withdrawn or suspended, or other enforcement actions may occur, if compliance with regulatory standards is not maintained or if problems occur following initial marketing.
Our business is also affected by patient and data privacy laws and government payer cost containment initiatives, as well as environmental health and safety laws and regulations.
11
United States Food and Drug Administration
Our products are subject to extensive regulation particularly as to safety, efficacy and adherence to the FDA Quality System Regulation, and related manufacturing standards. Medical device products are subject to rigorous FDA and other governmental agency regulations in the United States and similar regulations of foreign agencies abroad. The FDA regulates the design, development, research, preclinical and clinical testing, introduction, manufacture, advertising, labeling, packaging, marketing, distribution, import and export, and record keeping for such products to ensure that medical products distributed in the United States are safe and effective for their intended use. In addition, the FDA is authorized to establish special controls to provide reasonable assurance of the safety and effectiveness of most devices. Non-compliance with applicable requirements can result in, without limitation, warning or untitled letters, the FDA’s delay or refusal to approve pending applications, a clinical hold on a clinical investigation, import detentions, fines, civil and administrative penalties, injunctions, suspensions or losses of regulatory approvals, product recalls, withdrawals from the market, product seizures, operating restrictions, including total or partial suspension of production or distribution, refusal of the government to approve product export applications or allow us to enter into supply contracts, and criminal prosecution. The regulatory process for obtaining product approvals and clearances can be onerous and costly.
Medical Device Regulation
Under the Federal Food, Drug and Cosmetic Act (the “FDCA”), unless a device is exempt or marketed under an FDA enforcement discretion, premarket authorization is required to commercially distribute a new medical device in the U.S market. A new medical device could either be a device that has not previously received marketing authorization in the U.S, or a previously authorized device that has been changed in such a way that would require a new marketing application. Changes that could require a new marketing application for an existing device may relate to, but are not limited to, the intended use of the device, the indications for use, manufacturing, and technological characteristics or functionalities. The type of marketing authorization is generally linked to the classification of the device. The FDA classifies medical devices into three classes based on risk. Regulatory control increases from Class I (lowest risk) to Class III (highest risk). The FDA generally must clear or approve the commercial sale of new medical devices in Classes II and III, respectively. Commercial sales of our Class II medical devices (except for Class II exempt devices) and Class III medical devices within the U.S. must be preceded by either a pre-market notification filing pursuant to Section 510(k) of the FDCA (Class II) or the granting of a pre-market approval, or a PMA (Class III). As the risk of the device increases, so do the FDA’s requirements to obtain market authorization.
Generally, all Class III and some Class II medical devices will require clinical testing to adequately demonstrate the safety and effectiveness of the device prior to FDA approval or clearance of the device for commercialization. The extent to which FDA is involved in the development of clinical trial protocols depends, in part, on whether the study is a significant risk (SR) study or a nonsignificant risk (NSR) study. Before testing begins, all clinical studies must obtain Institutional Review Board (IRB) approval and for SR medical devices, an Investigational Device Exemption (IDE) from the FDA must be obtained. This exemption allows for clinical testing while ensuring participant safety and adherence to ethical standards. Though the regulatory requirements for an NSR study are less burdensome than those for an SR study, all clinical trials can be time consuming and costly and may not result in our desired outcomes.
Class I devices are those for which safety and effectiveness can be reasonably assured by adherence to a set of requirements referred to as general controls, which previously required compliance with the applicable portions of FDA’s QSR, and more recently, aspects of the FDA’s QMSR. These include facility registration and device listing, reporting of adverse events and malfunctions, which is referred to as medical device reporting, and truthful and non-misleading labeling and promotional materials. Most Class I devices are exempt from the premarket notification requirements (i.e., 510(k)-exempt).
510(k) Clearance Process
Class II devices are those that are subject to general controls, as well as special controls, which can include performance standards, specialized labeling and post-market surveillance. Most Class II devices are subject to the premarket notification requirements or the 510(k) process. To obtain 510(k) clearance, we must submit a premarket notification, or 510(k), to the FDA and demonstrate that our proposed medical device is substantially equivalent to a medical device that is legally marketed for the same intended use or a device commercially distributed prior to May 28, 1976, for which the FDA has not yet called for the submission of a PMA.
The device to which the company must show substantial equivalence is known as a predicate device. A proposed device is substantially equivalent to a predicate device if, with respect to the predicate device, it has the same intended use and has either (i) the same technological characteristics or (ii) different technological characteristics that do not raise different questions of safety and effectiveness, and the information submitted demonstrates to the FDA that the proposed device is as safe and effective as the predicate device.
If the FDA determines that our device is “not substantially equivalent” to a previously cleared device, for example, due to a finding of a lack of a predicate device, or that the proposed device has a new intended use or different technological
12
characteristic that raise different questions of safety or effectiveness when the proposed device is compared to the cited predicate device, the device is automatically designated as a Class III device. As the device sponsor, we must then fulfill more rigorous PMA requirements, or we can request a risk-based classification determination for the device in accordance with the FDA’s de novo classification process. As a result, FDA clearance requirements may extend the device development process for a considerable length of time. If the FDA agrees that the proposed device is substantially equivalent to the predicate device proposed by the manufacturer, it will grant 510(k) clearance to commercially market the device.
De Novo Classification Process
For novel medical devices that are low to moderate risk and are not substantially equivalent to a predicate device, a manufacturer may request a risk-based classification determination for the device in accordance with de novo classification process. This procedure allows a de novo requester whose novel device is automatically classified into Class III to request down-classification of its medical device into Class I or Class II on the basis that the device presents low or moderate risk, rather than requiring the submission and approval of a PMA. A requestor may submit a de novo request for classification after receiving a “not substantially equivalent” determination in response to a 510(k) submission or, absent the prior submission of a 510(k), when the sponsor determines that there is no legally marketed device upon which to base a determination of substantial equivalence.
The FDA may reject the de novo request if it identifies a legally marketed predicate device that would be appropriate for a 510(k) or determines that the device is not low to moderate risk or that general controls would be inadequate, and special controls cannot be developed, to control the risks. In the event the FDA determines that the data and information submitted demonstrate that general controls or general and special controls are adequate to provide reasonable assurance of safety and effectiveness, the FDA will grant the de novo request and a classification regulation will be established for the device type. When the FDA grants a de novo request for classification, the device is granted marketing authorization and can further serve as a predicate device for a future 510(k) submissions of that device type.
PMA Process
Class III devices include devices deemed by FDA to pose the greatest risk, such as life-supporting or life-sustaining devices, or implantable devices. With a few exceptions for certain types of devices classified into Class III that were in commercial distribution in the U.S. before May 28, 1976, Class III devices are subject to the pre-market approval (“PMA”) process which requires us to independently demonstrate that a medical device is safe and effective for its intended use. This process is generally much more time-consuming and expensive than the 510(k) or de novo processes. The PMA process involves a complex and lengthy testing process and may require several years to complete. We may need to first obtain an investigational device exemption (for significant risk devices), known as an IDE, in order to conduct extensive clinical testing of the device to obtain the necessary clinical data for submission to the FDA. Prior to obtaining approval, the manufacturer typically undergoes a bioresearch monitoring (“BIMO”) audit of the supporting clinical trial and a manufacturing audit, which could lengthen the process and potentially raise unforeseen issues that undermine the approvability of the product. The FDA may also require review by an advisory panel, which can further lengthen the process.
The FDA will approve a PMA only if after evaluating the supporting technical data it finds that the PMA contains sufficient, valid scientific evidence to assure that the device is safe and effective for its intended use(s). This approval may be granted with post-approval requirements including inspection of manufacturing facilities, additional patient follow-up for an indefinite period of time and/or post-approval clinical study or studies.
Exempt Devices
If a manufacturer’s device falls into a generic category of Class I or Class II devices that FDA has exempted by regulation, a premarket notification is not required before marketing the device in the U.S. (most Class I devices, and some Class II devices, are 510(k)-exempt.) Manufacturers of such devices are required to comply with FDA’s general controls, including FDA’s establishment registration and device listing requirements. Some 510(k)-exempt devices are also exempt from QMSR requirements, except for their respective complaint handling and recordkeeping requirements.
Device Modifications Post-clearance or Approval
Medical devices can be marketed only for the indications for which they are cleared or approved. After a device has received 510(k) clearance for a specific intended use, any change or modification that significantly affects its safety or effectiveness, such as a significant change in the design, materials, method of manufacture or intended use, may require a new 510(k) clearance or, depending on the modification, a de novo classification request or PMA approval, as well as payment of an FDA user fee. The determination as to whether or not a modification could significantly affect the device’s safety or effectiveness is initially left to the manufacturer using available FDA guidance; however, the FDA may, if it disagrees with the manufacturer’s determination, review this determination to evaluate the regulatory status of the modified product at any time and may require the manufacturer to cease marketing and recall the modified device until a new 510(k) clearance or PMA approval is obtained.
13
The manufacturer may also be subject to significant regulatory fines or penalties for marketing a modified device without the requisite pre-marketing authorization.
Postmarket Requirements
After a device is cleared or approved for commercial distribution, numerous federal and state regulatory requirements apply. These include product listing and establishment registration requirements, which help facilitate FDA inspections and other regulatory actions, and our manufacturing sites are subject to periodic inspection by the FDA for compliance with the FDA’s Quality Management System Regulations, which cover the procedures and documentation of the design, testing, production, control, quality assurance, labeling, packaging, sterilization, storage and shipping of medical devices; the Medical Device Reporting regulation, which requires that manufacturers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if the malfunction were to recur; and the Reports of Corrections and Removals regulation, which requires manufacturers to report recalls and field corrective actions to the FDA if initiated to reduce a risk to health posed by the device or to remedy a violation of the FDCA. Postmarket requirements are also followed globally where our products are registered and approved. These foreign jurisdictions have similar requirements to the FDA, including product registration, reporting of adverse events or serious incidents, trend reporting and reporting of withdrawals, recalls, and other corrective measures.
In addition, labeling and promotional activities are subject to scrutiny by the FDA and, in certain circumstances, by the Federal Trade Commission. Medical devices that are approved or cleared by the FDA may not be promoted for unapproved or uncleared uses, otherwise known as “off-label” promotion. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability, including substantial monetary penalties and criminal prosecution.
If the FDA believes that a company is not in compliance with applicable regulations, it may issue a warning letter or untitled letter, institute proceedings to detain or seize products, issue a recall or market withdrawal order, impose operating restrictions including total or partial suspension of production or distribution, enjoin future violations, assess civil penalties against the company, its officers or its employees and may recommend criminal prosecution to the U.S. Department of Justice. The majority of Integra manufacturing facilities participate in the Medical Device Single Audit Program and are audited annually for compliance with the Quality System for U.S. FDA, Canada, Australia, Brazil, and Japan.
Moreover, after clearance or approval is given, if the product is shown to be hazardous or defective, the FDA and foreign regulatory agencies have the power to withdraw the clearance or approval, as the case may be, or require us to change the device, its manufacturing process or its labeling, to supply additional proof of the device’s safety and effectiveness or to recall, repair, replace or refund the cost of the medical device. Because we currently export medical devices manufactured in the U.S. that have not been approved by the FDA for distribution in the U.S., we are required to obtain approval/registration in the country to which we are exporting and maintain certain records relating to exports and make these available to the FDA for inspection, if required.
Human Cells, Tissues and Cellular and Tissue-Based Products
Integra, through its wholly-owned subsidiary BioD LLC (“BioD”), is involved with the recovery, processing, storage, transportation and distribution of donated amniotic tissue. The FDA has specific regulations governing HCT/Ps. An HCT/P is a product containing, or consisting of, human cells or tissue intended for transplantation into a human patient. Examples of HCT/Ps include bone, ligament, skin and cornea.
Some HCT/Ps fall within the definition of a biological product, medical device or drug regulated under the FDCA. These biologic, device or drug HCT/Ps must comply both with the requirements exclusively applicable to HCT/Ps and, in addition, with requirements applicable to biologics, devices or drugs, including premarket clearance or approval from the FDA.
Section 361 of the Public Health Service Act (“Section 361”) authorizes the FDA to issue regulations to prevent the introduction, transmission or spread of communicable disease. HCT/Ps regulated as “361” HCT/Ps are subject to requirements relating to registering facilities and listing products with the FDA, screening and testing for tissue donor eligibility, and Good Tissue Practices when processing, storing, labeling, and distributing HCT/Ps, including required labeling information, stringent record keeping, and adverse event reporting.
The American Association of Tissue Banks (“AATB”) has issued operating standards for tissue banking. Compliance with these standards is a requirement in order to become an AATB-accredited tissue establishment. In addition, some states have their own tissue banking regulations. We are licensed or have permits for tissue banking in California, Delaware, Illinois, Maryland, New York, Oregon, and Tennessee. In Tennessee, we are registered with the FDA Center for Biological Evaluations and Research.
Procurement of certain human organs and tissue for transplantation is subject to the restrictions of the National Organ Transplant Act, which prohibits the transfer of certain human organs, including skin and related tissue for valuable
14
consideration, but permits the reasonable payment associated with the removal, transportation, implantation, processing, preservation, quality control and storage of human tissue and skin. BioD is a registered Tissue Bank and is involved with the recovery, storage and transportation of donated human amniotic tissue.
Regulations Outside of the U.S.
Medical device regulations also are in effect in many of the countries in which we do business outside the U.S. These regulations may vary substantially from country to country. To market our products in most countries, we must obtain regulatory approvals or certifications and comply with extensive safety and quality regulations in those countries. The time required to obtain approval or certification to market our products in a foreign country may be longer or shorter than that required for FDA clearance or approval, and the requirements may differ.
In the European Economic Area (“EEA”), which is comprised of the 27 Member States of the European Union (the “EU”) plus Norway, Iceland and Liechtenstein, medical devices need to comply with specific requirements. Medical device manufacturers are required to affix the CE mark (i.e., a mandatory conformity marking for certain products sold within the EEA) to their medical devices, often after the intervention of a Notified Body and the issuing of a CE Certificate of Conformity.
Regulation 2017/745 on medical devices (the “EU MDR”) sets out the basic regulatory framework currently applicable to medical devices in the EEA. The EU MDR became applicable on May 26, 2021, repealing the prior Council Directive 93/42/EEC (the “EU MDD”), which had been regulating medical devices in the EEA for more than 20 years. This represented a major change in the regulatory landscape of medical devices in the EEA. The EU MDR sets out certain transitional provisions that allow for medical devices covered by the repealed EU MDD (called “legacy devices”) to still be marketed in the EEA for a certain period of time. Currently, manufacturers can place on the EEA market legacy devices until maximum December 31, 2027 or December 31, 2028, depending on the type of device and subject to meeting certain conditions.
The requirements set forth in the EU MDR are generally consistent with those laid out in the EU MDD, but in many topics there are additional or stricter requirements. Although we continue to transition our certification profile to meet the new EU MDR requirements, these stricter regulations set forth in the EU MDR may pose additional challenges for Integra to continue marketing products in the EEA. See “Item 1A. Risk Factors - We are subject to stringent domestic and foreign medical device regulations and oversight and any adverse action may adversely affect our ability to compete in the marketplace and our financial condition and business operations” of this Annual Report on Form 10-K.
In the EEA, medical devices are currently required to comply with the General Safety and Performance Requirements (or “GSPR”) in Annex I of the EU MDR (for legacy devices, this corresponds to the Essential Requirements of Annex I of the EU MDD). Compliance with GSPR is a prerequisite to be able to affix the CE mark to medical devices, without which they cannot be commercialized in the EEA. To demonstrate compliance with the GSPR and obtain the right to affix the CE mark, a manufacturer must undergo a conformity assessment procedure, which varies according to the type of medical device and its classification. In the EEA medical devices are classified into four different risk classes: Class I (which is further divided into (i) devices that are placed on the market in sterile condition, (ii) have a measuring function, (iii) are reusable surgical instruments, and (iv) all others), IIa, IIb and III. Apart from low risk medical devices (Class I if they have no measuring function, are not sterile, and are not reusable surgical instruments), where the manufacturer can issue an EU Declaration of Conformity based on a self-assessment of the conformity of the devices with the GSPR, a conformity assessment procedure requires the intervention of a Notified Body, which is an organization accredited by the competent authority of an EEA country to conduct conformity assessments. The Notified Body typically audits and examines the products’ technical documentation and the quality management system for the manufacture, design and final inspection of the medical devices before issuing a CE Certificate of Conformity. After receiving the CE Certificate of Conformity from the Notified Body upon successful completion of the conformity assessment, the manufacturer can draw up an EU Declaration of Conformity which allows it to affix the CE mark to the products.
Besides its involvement in the initial conformity assessment procedure, the Notified Body is required to carry out an annual audit (surveillance audit) and is also required to randomly perform unannounced audits at least once every five years. The quality management system and technical documentation of manufacturers will be required to be recertified periodically, as CE Certificates of Conformity issued by a Notified Body remain valid only for the period indicated in them, in no case exceeding five years. The EU MDR also provides various requirements relating to post-market surveillance and vigilance, including the obligation for manufacturers to implement a post-market surveillance system. Once a device is on the EEA market, manufacturers must comply with certain vigilance requirements, such as reporting serious incidents and field safety corrective actions (even those occurring outside the EEA) to the relevant competent authorities.
On December 16, 2025, the European Commission published a proposal to amend the EU MDR to simplify the regulation and address various concerns, including those related to notified body capacity constraints, certification timelines, regulatory burden and device availability. If adopted, the proposed revisions could impact, amongst others, certification timelines, recertification (the maximum period of validity of certificates, currently 5 years, is proposed to be removed), certification activities (for example, a reduced involvement of notified bodies in the conformity assessment of class IIa and IIb devices is proposed, as well
15
as the possibility to replace on-site audits by remote audits) and post-market surveillance requirements (the proposal provides the possibility to conduct surveillance audits only every two years, and unannounced audits only ‘for-cause’). The scope and timing for potential adoption of this revision is still unknown. The ultimate effect of any EU MDR amendments on our business will depend on the shape of the adopted revised legislation and corresponding application timelines.
The EU MDD, EU MDR, ISO 9000 series and ISO 13485 are recognized international quality standards that are designed to ensure that we develop and manufacture quality medical devices. A Notified Body audits our facilities annually to verify our compliance with the ISO 13485 Quality System standard. Other countries are also instituting regulations regarding medical devices or interpreting and enforcing existing regulations more strictly. Compliance with these regulations requires extensive documentation and clinical reports for our products, revisions to labeling, and other requirements such as facility inspections to comply with the registration requirements.
Certain countries, including those in the EEA, have issued regulations that govern products that contain materials derived from animal sources. Regulatory authorities are particularly concerned with materials infected with the agent that causes BSE. These regulations affect our dermal regeneration products, duraplasty products, hernia repair products, biomaterial products for the spine, nerve and tendon repair products and certain other products, all of which contain material derived from bovine tissue. Although we take great care to ensure that our products are safe and free of agents that can cause disease, products that contain materials derived from animals, including our products, may become subject to additional regulation, or even be banned in certain countries, because of concern over the potential for prior transmission. Significant new regulations, a ban of our products, or a movement away from bovine-derived products because of an outbreak of BSE could have a material, adverse effect on our current business or our ability to expand our business. See “Item 1A. Risk Factors – Risks Related to our Regulatory Environment” of this Annual Report on Form 10-K.
Regarding HCT/Ps and as happens in the U.S., these products can have various regulatory qualifications (medical devices, medicinal products or blood/tissues/cells products). The donation, collection, procurement, testing, processing, preservation, storage, import, export and distribution in the EEA of human blood, tissues and cells is currently mainly regulated by Directive 2002/98/EC (the “Blood Directive”), Directive 2004/23/EC (the “Tissues and Cells Directive”) and the implementing acts adopted by each EEA country. This legislation also partially applies to manufactured products derived from human blood, tissues and cells. In July 2024, Regulation (EU) 2024/1938 on standards of quality and safety for substances of human origin intended for human application (the “SoHO Regulation”) was published. This new Regulation will apply from August 7, 2027 and replace the Blood Directive and the Tissues and Cells Directive, changing the regulatory framework for the use of these substances in the EEA.
Regulations Governing Reimbursement
Market acceptance of our medical products in the U.S. and other countries is dependent upon the purchasing and procurement practices of our customers and patient need for our products and procedures and, the coverage and reimbursement of patients’ medical expenses by government healthcare programs, private insurers or other healthcare payors. The delivery of our devices is subject to regulation by the HHS and comparable state and non-U.S. agencies responsible for reimbursement and regulation of healthcare items and services. Healthcare providers that purchase medical devices generally rely on third-party payors, including, in the U.S., the Medicare and Medicaid programs and private payors, such as indemnity insurers, employer group health insurance programs and managed care plans, to reimburse all or part of the cost of the products. As a result, demand for our products is and will continue to be dependent in part on the coverage and reimbursement policies of these payors. The manner in which reimbursement is sought and obtained varies based upon the type of payor involved and the setting in which the product is furnished and utilized. Government payors, such as Medicare and national health systems of EU countries, are increasingly seeking additional clinical evidence beyond the data required to obtain marketing clearance, before covering our products for their patients. For example, in the EU, the Health Technology Assessment (HTA) Regulation (EU) 2021/2282 which began to apply from January 12, 2025 (subject to phased implementation), provides for the use of common HTA tools, methodologies, and procedures across the EU for certain categories of health technologies, including certain medical devices, and sets the basis for enhanced cooperation at the EU level for joint clinical assessments. Reimbursement from Medicare, Medicaid and other third-party payors may be subject to periodic adjustments as a result of legislative, regulatory and policy changes, as well as budgetary pressures. For instance, in the 2026 Medicare physician, hospital outpatient, and ambulatory surgical center payment system final rules, Medicare revised its payment methodology for most sheet skin substitute products to a single per square centimeter payment rate regardless of product and settings, which may significantly alter Medicare payment for such products in these settings. Possible reductions in, or eliminations of, coverage or reimbursement by third-party payors, or denial of, or provision of uneconomical reimbursement for new products may affect our customers' revenue and ability to purchase our products. Any changes in the healthcare regulatory, payment or enforcement landscape relative to our customers’ healthcare services have the potential to significantly affect our operations and revenue.
Implementation of legislative or regulatory reforms to reimbursement systems, including price regulation, reference pricing systems, competitive bidding and tendering, coverage and payment policies, comparative effectiveness of therapies, health technology assessments and managed-care arrangements, or adverse decisions relating to our products by administrators of these systems in coverage or reimbursement, could significantly reduce reimbursement or result in the denial of coverage,
16
which could have an impact on the acceptance of and demand for our products and the prices that our customers are willing to pay for them.
Healthcare Reform
A primary trend in the U.S. healthcare industry and elsewhere is cost containment. For example, the Affordable Care Act (“ACA”), went into effect in 2010, and, among other things, includes changes to the coverage and payment for products under government health care programs. In the United States, there have been, and we expect there to continue to be, a number of legislative and regulatory initiatives, at both the federal and state government levels, to change the healthcare system in ways that, if approved, could affect our ability to sell our products profitably. Among policy makers and payors in the United States and elsewhere, there is significant interest in promoting changes in healthcare systems with the stated goals of containing healthcare costs, improving quality and/or expanding access. Current and future legislative proposals to further reform healthcare or reduce healthcare costs may limit coverage of or lower reimbursement for the procedures associated with the use of our products. The cost containment measures that payors and providers are instituting and the effect of any healthcare reform initiative implemented in the future could impact our revenue from the sale of our products.
Other legislative changes have been proposed and adopted in the United States since the ACA was enacted. For example, in August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. When Congress was unable to reach required deficit reduction, it triggered the legislation’s automatic reduction to several government programs. This included aggregate reductions of Medicare payments to providers currently at 2% per fiscal year and, due to subsequent legislative amendments, will remain in effect through 2033.
On July 4, 2025, the “One Big Beautiful Bill Act,” or OBBBA, was signed into law. The OBBBA is projected to decrease federal health care spending by approximately $1 trillion by reducing Medicaid spending and enrollment and making changes to federal Medicare spending. The law also made changes to ACA marketplace enrollment that are projected to decrease the number of individuals with marketplace coverage. It is unclear if these changes will impact demand for our products.
Some of the provisions of the ACA and related laws have been, and may continue to be, subject to judicial and Congressional challenges, and to modifications in their interpretation or implementation. We plan to monitor health care legislation and the impact these changes, if enacted, may have on our business.
There has also been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several recent Congressional inquiries and proposed bills designed to, among other things, bring more transparency to product pricing and review the relationship between pricing and manufacturer programs. Individual states in the U.S. have also become increasingly active in enacting legislation and implementing regulations designed to control product pricing. We expect that additional foreign, federal and state healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in limited coverage and reimbursement and reduced demand for our products.
There have been, and likely will continue to be, legislative and regulatory proposals at the national level in the U.S. and other jurisdictions globally, as well as at some regional, state and/or local levels within the U.S. or other jurisdictions, directed at broadening the availability of healthcare and containing or lowering the cost of healthcare. Such reforms could have an adverse effect on anticipated revenues from product that we may successfully develop and for which we may obtain marketing approval and may affect our overall financial condition and ability to develop product.
Other regulations
Healthcare Fraud and Abuse Laws. In the U.S., we are subject to laws and regulations pertaining to healthcare fraud and abuse, including anti-kickback laws, false claims, and other health care fraud laws that regulate, among other things, the means by which companies in the health care industry may sell and market their products to hospitals and health care professionals and regulate the arrangements and engagements with customers. We are also subject to other federal and state laws that constrain the way we can market and sell our products, or require reporting of certain transfers of value to health care professionals. Some of the laws and regulations we are subject to include:
•The U.S. federal Anti-Kickback Statute (the “AKS”), which prohibits, among other things, persons or entities from knowingly and willfully soliciting, offering, receiving or paying any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, in exchange for or to induce either the referral of an individual for the furnishing or arranging for a good or service, or for the purchasing, leasing, ordering, or arranging for or recommending any good, facility, service or item for which payment may be made in whole or in part under federal healthcare programs, such as the Medicare and Medicaid programs. The term “remuneration” expressly includes kickbacks and bribes, and also has been broadly interpreted to include anything of value, including, for example, gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash, waivers of payments, ownership interests and providing anything at less than its fair market value. The AKS is broad and prohibits many arrangements and practices that are lawful in businesses outside of the healthcare industry. There are a number of statutory exceptions and regulatory safe
17
harbors that may protect certain business arrangements from prosecution if strictly complied with; however, those exceptions and safe harbors are drawn narrowly. There are also no available exceptions or safe harbors for many common business activities. Practices that involve remuneration to those who prescribe, purchase, recommend or arrange for the purchase, order or recommendation of medical devices, including discounts, providing items or services for free or engaging such individuals as consultants, advisors, or speakers, may be subject to scrutiny if they do not fit squarely within an available exception or safe harbor and would be subject to a facts and circumstances analysis to determine compliance with the AKS. A violation of the AKS can be established without proving that the person or entity had actual knowledge of the statute or specific intent to violate it. A claim that includes items or services resulting from an AKS violation constitutes a false or fraudulent claim for purposes of the federal civil False Claims Act (the “False Claims Act”).
•The False Claims Act, which prohibits, among other things, persons, or entities from knowingly presenting or causing to be presented a false or fraudulent claim for payment of government funds or knowingly presenting or causing to be presented a false record or statement material to an obligation to pay money to the government or knowingly and improperly avoiding, decreasing, or concealing an obligation to pay money to the federal government. Medical device manufacturers have been investigated and have reached substantial financial settlements with the federal government under the False Claims Act for a variety of alleged improper activities, including causing false claims to be submitted as a result of the marketing of their products including for unapproved and thus non-reimbursable uses and interactions with prescribers and other customers, including those that may have affected their billing or coding practices and submission of claims to the federal government. Actions under the False Claims Act can be brought by qui tam relators, or whistleblowers, on behalf of themselves and the government, and those relators can share in any settlement or resolution.
•The Health Insurance and Portability Act of 1996, and its implementing regulations (collectively, “HIPAA”), which imposes criminal and civil liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, or knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement, in connection with the delivery of, or payment for healthcare benefits, items or services by a healthcare benefit program, which includes both government and privately funded benefits programs. Similar to the U.S. federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
•Analogous state and foreign law equivalents of each of the above U.S. federal laws, such as anti-kickback and false claims laws that apply to items or services reimbursed by any third-party payor, including commercial insurers.
•State and foreign laws that require medical device companies to comply with the industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict payments (or require reporting transfers of value) that may be made to healthcare providers and other potential referral sources.
•The federal Physician Payments Sunshine Act, implemented as the Open Payments program, and its implementing regulations, requires certain manufacturers of drugs, devices, biologics and medical supplies that are reimbursable (directly or indirectly) under Medicare, Medicaid, or the Children’s Health Insurance Program to report annually to the CMS information related to certain payments or others transfers of value made in the preceding calendar year to physicians, other health care professionals, such as physician assistants and nurse practitioners, and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Certain foreign countries, including the EEA, have similar requirements for the reporting of transfers of value to healthcare professionals.
Anti-bribery laws exist in many of the countries in which we sell our products outside the U.S., as well as the United States Foreign Corrupt Practices Act (the “FCPA”) which addresses the activities of U.S. companies in foreign markets. Violations of these laws are punishable by criminal and civil sanctions, including, but not limited to, civil and criminal penalties, up to treble damages, fines, restitution and exclusion from participation in federal and state healthcare programs, including Medicare and Medicaid. Our products also are subject to regulation regarding reimbursement, and U.S. healthcare laws apply when a customer submits a claim for a product that is reimbursed under a federally funded healthcare program. These global laws require that we exercise care in designing our sales and marketing practices, including interactions with healthcare professionals, and customer discount arrangements. See “Item 1A. Risk Factors – We are exposed to a variety of risks relating to our international sales and operations” of this Annual Report on Form 10-K for further details.
Import-export. Our international operations subject us to laws regarding sanctioned countries, entities and persons, customs, and import-export. Among other things, these laws restrict, and in some cases can prevent, U.S. companies from directly or indirectly selling goods, technology or services to people or entities in certain countries. These laws also require that we exercise care in our business dealings with entities in and from foreign countries. In addition to our need to comply with such regulations in connection with our direct activities, we also sell and provide goods, technology and services to agents, representatives and distributors who may export such items to customers and end-users. If we, or the third parties through which we do business, are not in compliance with applicable import, export control or economic sanctions laws and regulations,
18
we may be subject to civil or criminal enforcement action, and varying degrees of liability. Such actions may disrupt or delay sales of our products or services or result in restrictions on our distribution and sales of products or services that may materially impact our business.
Environmental Health and Safety. Our research, development and manufacturing processes involve the controlled use of certain hazardous materials. We are subject to country-specific, federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of these materials and certain waste products. We believe that our environmental, health and safety procedures for handling and disposing of these materials comply with the standards prescribed by the controlling laws and regulations. However, risk of accidental releases or injury from these materials is possible. These risks are managed to minimize or eliminate associated business impacts. In the event of this type of accident, we could be held liable for damages and face a liability that could exceed our resources. We could be subject to a regulatory shutdown of a facility that could prevent the distribution and sale of products manufactured there for a significant period of time, and we could suffer a casualty loss that could require a shutdown of the facility in order to repair it, any of which could have a material, adverse effect on our business. Although we continuously strive to maintain full compliance with respect to all applicable global environmental, health and safety laws and regulations, we could incur substantial costs to fully comply with future laws and regulations, and our operations, business or assets may be negatively affected. Furthermore, global environmental, health and safety compliance is an ongoing process. We have compliance procedures in place for compliance with Employee Health & Safety laws, driven by a centrally led organizational structure that ensures proper implementation, which is essential to our overall business objectives.
In addition, we are subject to numerous federal, state, foreign and local laws relating to safe working conditions, environmental protection and fire hazard control, among others. We may be required to incur significant costs to comply with these laws and regulations in the future and complying with these laws may result in a material adverse effect upon our business, financial condition and results of operations.
Data Privacy and Cybersecurity Laws and Regulations. As a business with a significant global footprint, compliance with evolving regulations and standards in data privacy and cybersecurity (relating to the confidentiality and security of our information technology systems, products such as medical devices, and other services provided by us) may result in increased costs, lower revenue, new complexities in compliance, new challenges for competition, and the threat of increased regulatory enforcement activity. Our business relies on the secure electronic transmission, storage and hosting of sensitive information, including personal information, financial information, intellectual property, and other sensitive information related to our customers and workforce.
In the U.S., the collection, maintenance, protection, use, transmission, disclosure and disposal of certain personal information and the security of medical devices are regulated at the U.S. federal, state, and industry levels. U.S. federal and state laws govern the confidentiality of certain patient health information, including patient medical records, and restrict the use and disclosure of patient health information by health care providers and other persons or entities that create, receive, maintain, or transmit health information. For example, in the U.S., depending on the facts and circumstances, we could be subject to the requirements of HIPAA. HIPAA imposes privacy and security obligations on covered entity health care providers, health plans, and health care clearinghouses, as well as their “business associates” – certain persons or entities that create, receive, maintain, or transmit protected health information in connection with providing a specified service or performing a function on behalf of a covered entity. HIPAA also regulates standardization of data content, codes and formats used in health care transactions and standardization of identifiers for health plans and providers. Penalties for violations of HIPAA include significant civil and criminal penalties for each violation. In addition, the FDA has issued guidance advising manufacturers to take cybersecurity risks into account in product design for connected medical devices and systems, to assure that appropriate safeguards are in place to reduce the risk of unauthorized access or modification to medical devices that contain software and reduce the risk of introducing threats into healthcare systems that are connected to such devices. The FDA also issued guidance on post market management of cyber security in medical devices.
The Federal Trade Commission (“FTC”) also sets expectations for taking appropriate steps to safeguard consumers’ personal information, and providing a level of privacy or security commensurate to promises made to individuals. Pursuant to Section 5 of the FTC Act, which governs unfair and deceptive trade practices, the FTC expects a company’s data privacy and security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities. Failure to meet these standards may constitute unfair or deceptive acts or practices in violation of the FTC Act. The FTC also has the power to enforce the Health Breach Notification Rule, which imposes notification obligations on companies for breaches of certain health information contained in personal health records. Enforcement by the FTC under the FTC Act and Health Breach Notification Rule can result in civil penalties or enforcement actions.
At the state level, the California Consumer Privacy Act (“CCPA”)establishes certain requirements for processing personal data, including obligations related to transparency and the collection, use, retention, and disclosure of personal data, and provides California consumers (as defined in the law) certain rights. Such rights include rights to access, correct, and delete personal
19
information, opt out of certain personal information sharing, and receive detailed information about how personal information is used. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches—involving certain types of personal information—that is expected to increase data breach litigation. The CCPA may increase our compliance costs and potential liability. Nearly two dozen other states have enacted privacy laws similar to the CCPA, and some states, like Washington and Nevada, have enacted health privacy specific-specific consumer laws that grant heightened rights with respect to health information. Moreover, as a result of the broad scale release and availability of Artificial Intelligence (“AI”) technologies such as generative AI, there is a global trend towards more regulation (e.g., the EU AI Act and AI laws passed in U.S. states) designed to ensure the ethical use, privacy, and security of AI and the data that it processes. Compliance with such laws will likely be an increasing and substantial cost in the future.
Outside the U.S., we are impacted by the privacy and data security requirements at the international, national and regional level, and on an industry specific basis. Legal requirements in these countries relating to the collection, use, retention, disclosure, and international transfer of personal data and, potentially, intellectual property continue to evolve with increasingly strict enforcement regimes. In Europe, for example, we are subject to the EU General Data Protection Regulation (EU GDPR) and the UK General Data Protection Regulation (UK GDPR) which impose restrictions on the collection, use and transfer of personal data and includes, among other things, a requirement for prompt notice of data breaches to data subjects and supervisory authorities in certain circumstances and significant fines for non-compliance, which can go up to €20 million/ GBP 17.5 million or up to 4% of the total worldwide annual turnover of the preceding financial year, whichever is higher. The EU and UK GDPR also confer the right for data subjects to lodge complaints with supervisory authorities, seek judicial remedies and obtain compensation for damages resulting from violations of the GDPR. The EU and UK GDPR also requires companies processing personal data (including health data from clinical trials) of individuals residing in the EU or UK to comply with local privacy and data protection rules. The EUK and UK GDPR governs the lawfulness of processing personal data (including obtaining valid consent of the individuals to whom the personal data relates, where applicable), the disclosure of processing details to individuals, the adequacy, relevance and necessity of the personal data collected, the retention of personal data, the sharing of personal data with third parties, the transfer of personal data out of the European Economic Area/UK to third countries including the U.S., contracting requirements (such as with clinical trial sites and vendors), the use of personal data in accordance with individual rights, the security of personal data and security breach/incident notifications. Data protection authorities from the different European Member States and the UK may interpret the GDPR and applicable related national laws differently and impose requirements additional to those provided in the GDPR that sit alongside the GDPR, as set out under applicable local data protection law. In addition, guidance on implementation and compliance practices may be issued, updated or otherwise revised. Enforcement by European and UK regulators is generally active, and failure to comply with the GDPR or applicable Member State/UK local law may result in fines or other penalties. Further, the EU or UK Government may amend or update data protection law, which may result in changes to our business operations and potentially incur commercial cost.
In particular, when we rely on third-party service providers processing personal data of subjects in the EU or UK, we must enter into suitable agreements with these providers and receive sufficient assurances that the providers meet the requirements of the EU and UK GDPR. The obligations under the EU and UK GDPR may therefore be onerous and adversely affect our business, financial condition, results of operations and prospects. Please refer to “Item 1A. Risk Factors – Failure to comply with laws relating to the confidentiality of sensitive personal information or standards related to the transmission of electronic health data, may require us to make significant changes to our products, or incur penalties or other liabilities” of this Annual Report on Form 10-K for additional discussion of the risks accompanying compliance with data privacy and cybersecurity laws and regulations.
These laws and regulations impact the ways in which we use and manage personal data, protected health information, and our information technology systems. They also impact our ability to move, store, and access data across geographic boundaries. Compliance with these requirements may require changes in business practices, complicate our operations, and add complexity and additional management and oversight needs. They also may complicate our clinical research activities, as well as product offerings that involve transmission or use of clinical data.
HUMAN CAPITAL
Our people are our greatest asset and we view human capital management and the strength of our employees as integral to the long-term success of our business. We understand that we rely on our employees worldwide to propel our organization forward with great innovations, strong leadership and a focus on execution excellence.
In seeking to sustain a culture of excellence and accountability, we focus on employee empowerment, professional development and building an environment where all employees can contribute to their fullest potential. These efforts have been recognized through our inclusion in several best workplace lists globally in 2025 and 2024. We continue to advance our broader organizational sustainability initiatives, and published our annual environmental, social and governance (“ESG”) report in the third quarter of 2025. For more information on our ESG strategy, goals, performance, and achievements, please visit “Our
20
Company—ESG Report” at https://www.integralife.com/esg-report. Information on our website is not incorporated by reference herein and is not part of this Annual Report on Form 10-K.
WORKFORCE DEMOGRAPHICS
As of December 31, 2025, we had 4,427 regular full and part time employees.
69.6% of our employees are located in the United States, 20.6% in Europe, 4.9% in Asia Pacific, 3.6% in China and 1.3% in Latin America and Canada.
BUILDING A STRONG WORKFORCE
A talented and engaged workforce is a business priority and a key to our long-term success. We believe our company is stronger when we leverage broad perspectives to meet the needs of our shareholders, customers, colleagues and the communities we serve. Our commitment to workforce development starts at the top with our Board of Directors and Chief Executive Officer. At all levels of the Company, we focus on attracting, retaining, and developing talent that drives innovation and results. Through various Business Resource Groups (“BRGs”), leadership councils and external partnerships, we provide opportunities for colleagues to contribute to our culture, develop professionally and provide feedback to our executive team. As of December 31, 2025 there are eight (8) Integra-sponsored BRGs, employee-led groups, which provide career development, leadership opportunities and networking connections across the organization.
COMPENSATION BENEFIT
Our compensation philosophy is designed to reinforce and align with our mission, business strategy, and financial needs. We invest in the physical, emotional and financial well-being of our employees through our compensation and benefit programs. We provide market-competitive compensation and benefits based on benchmarking surveys we conduct regularly for all position levels against relevant peer companies. Our annual and long-term incentive packages are linked directly to business and individual performance, with a balance of short- and long-term financial and strategic objectives. We have an employee stock purchase plan. Eligibility for non-salary benefits such as salary continuance, life insurance, health insurance, and similar benefits, follows local regulations and practices.
We are a pay-for-performance company committed to fair pay. All compensation decisions are made without regard to personal characteristics such as, but not limited to, gender, race, color, national or ethnic origin, age, disability, sexual orientation, gender identity or expression, genetic information, religion, or veteran status. As part of our commitment to compensation equity, Integra regularly conducts a pay equity analysis, reviewing how our organization compensates employees against external and internal data in conjunction with the role and scope of each position and making adjustments if necessary.
TALENT DEVELOPMENT AND RETENTION
We have comprehensive and effective human capital development programs in place because we believe that the personal success of our employees is critical to the overall success of our business. To build a diverse and talented organization, we have invested in honing our recruiting and hiring processes to attract top talent and engage new hires from the very beginning of their experience at Integra.
We offer a variety of opportunities for our employees to learn and grow. Continued learning and development is a critical component of employee job satisfaction, retention, and career advancement—and ultimately, a driver of business success. We encourage and promote experiential, collaborative, and formal learning programs. Employees are also encouraged to discuss with their managers the skills, training, and experience needed to grow and develop. In addition to several skills-based trainings available (technical, sales, leadership ability) to all employees, managers may recommend external job-specific development programs to employees. These programs are paid for directly by Integra.
EMPLOYEE HEALTH AND SAFETY
We are committed to providing a safe environment for all employees and visitors. This commitment is supported by our environmental, health, and safety management systems, alongside entrusting managers to oversee and ensure compliance with local laws and safety standards at their respective sites to foster a safe workplace culture. We implement our approach globally by our systems and support at regional and country levels from colleagues that implement proper safety protocols, identify and correct hazards, and remain safety conscious at all times. Our Environmental Health and Safety (“EH&S”) organizational structure incorporates both workplace EH&S coordinators and compliance teams. We have developed an Incident Procedure Policy and General Safety Rules that guide our colleagues to improve our workplace environment, improve safety, and reduce risk and costs.
EMPLOYEE ENGAGEMENT AND WELLBEING
We regularly seek employee feedback and sentiment about our workplace through global engagement pulse surveys conducted on at least an annual basis. After each survey is complete, we share detailed results with senior management and all employees
21
within each department. We are incorporating employee survey results into our corporate strategies – across company, division and function levels – and have further used this employee feedback to modify corporate programs and initiatives. We believe this process enables us to monitor employee engagement and create a continuously improving, satisfying work environment for our employees.
We are committed to improving the quality of life of our employees and their families. Our health and wellbeing programs differ by country and typical benefits include comprehensive health insurance, disability coverage, workplace accommodations, parental leave and other leaves of absence based on health or life events, employee assistance programs, fitness reimbursement, and flu shots fully covered by the company. We also provide on-demand health advocates to help employees navigate the health insurance system, access to digital health solutions, a weight management program, smoking cessation assistance, a substance use disorder helpline, a diabetes health program and other similar programs to drive healthy behaviors and awareness.
FINANCIAL INFORMATION ABOUT GEOGRAPHIC AREAS
Financial information about our geographical areas is set forth in our financial statements Note 16. Segment and Geographic Information, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
CORPORATE HISTORY AND AVAILABLE INFORMATION
Integra LifeSciences Holdings Corporation was founded in 1989. Our common stock trades on the Nasdaq Global Select Market (“Nasdaq”) under the symbol “IART.”
We are subject to the informational requirements of the Exchange Act of 1934. In accordance with the Exchange Act, we file annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and any amendments to those reports, proxy statements and other information with the Securities and Exchange Commission, (the “SEC”). Our financial information may be viewed, including the information contained in this report, and other reports we file with the SEC, on the Internet, without charge as soon as reasonably practicable after we file them with the SEC, in the “SEC Filings” page of the Investor Relations section of our website at investor.integralife.com. A copy may also be obtained for any of these reports, without charge, from our Investor Relations department, 1100 Campus Road, Princeton, NJ 08540. Alternatively, reports filed may be viewed or obtained through the SEC’s website at www.sec.gov.
Investors and others should note that we announce material financial information to our investors using our investor relations website (investor.integralife.com), SEC filings, press releases, public conference calls and webcasts. We use these channels as well as social media to communicate with the public about our Company, our services and other issues. It is possible that the information we post on social media could be deemed to be material information. Therefore, we encourage investors, the media, and others interested in our Company to review the information we post on the social media channels listed on our investor relations website. We have used, and intend to continue to use, our investor relations website as means of disclosing material non-public information and for complying with our disclosure obligations under Regulation FD. Additional corporate governance information, including our certificate of incorporation, bylaws, corporate governance guidelines, board committee charters, and global code of conduct, is also available on our investor relations website under the heading “Corporate Governance.” The contents of our websites are not intended to be incorporated by reference into this Annual Report on Form 10-K or in any other report or document we file with the SEC, and any references to our website are intended to be inactive textual references only.
ITEM 1A. RISK FACTORS
Our business faces significant risks. These risks include those described below and may include additional risks and uncertainties not presently known to us or that we currently deem immaterial. Our business, financial condition and results of operations could be materially adversely affected by any of these risks, and the trading prices of our common stock could decline by virtue of these risks. Some of the factors, events and contingencies discussed below may have occurred in the past, but the disclosures below are not representations as to whether or not the factors, events or contingencies have occurred in the past and instead reflect our beliefs and opinions as to the factors, events or contingencies that could materially and adversely affect our business in the future.
GLOBAL CHALLENGES AND MACROECONOMIC CONDITIONS
The continuing worldwide macroeconomic and geopolitical uncertainty may adversely affect our business and prospects.
The United States and foreign countries have experienced recessionary pressures and face continued concerns about the systemic impacts of adverse economic conditions and geopolitical issues. Any negative impact on economic conditions and international markets, including increased geopolitical instability and other macroeconomic factors, including heightened inflation, trade barriers and related restrictions (including tariffs and related countermeasures), import or export licensing requirements, armed conflict and acts of terrorism, geopolitical tension and instability, supply chain disruptions, interest rate and foreign currency rate fluctuations, and volatility in the capital markets could negatively impact our business, financial condition, and results of operations.
22
Our business and results of operations have been and may continue to be adversely impacted by changes in macroeconomic conditions, including inflation, rising interest rates, bank failures and the accessibility of capital markets. Uncertainty about global economic and geopolitical conditions may also cause decreased demand for our products and services and increased competition, which could result in lower sales volume and downward pressure on the prices for our products, longer sales cycles, and slower adoption of new technologies. Such conditions may also constrain the liquidity of hospitals and other customers or limit their access to financing, which could further delay or reduce capital equipment purchases. A weakening of macroeconomic conditions may also adversely affect our suppliers, which could result in interruptions in supply. In addition, heightened macroeconomic and geopolitical uncertainty may impair our ability to accurately forecast demand and plan our business and operational activities.
Market acceptance of our medical products in the U.S. and other countries is dependent upon the medical equipment purchasing and procurement practices of our customers, patient need for our products and procedures and the reimbursement of patients' medical expenses by government healthcare programs and third-party payors. The continuing uncertainty surrounding global economic conditions and financial markets may cause the purchasers of medical equipment to decrease their procurement activities, particularly the sales of capital equipment such as our ultrasonic surgical aspirators, neuro monitors and cranial stabilization products. Economic uncertainty, an increase in unemployment rates, as well as increasing health insurance premiums, co-payments and deductibles may adversely affect demand for our products and elective or non-reimbursed procedures. Furthermore, governments and other third-party payors around the world facing tightening budgets could move to further reduce the reimbursement rates or the scope of coverage offered, which could adversely affect the sales of our products.
RISKS RELATING TO OUR BUSINESS
Our operating results may fluctuate.
Our operating results, including components of operating results such as gross margin and operating expenses, may fluctuate from time to time. Our operating results have fluctuated in the past and can be expected to do so from time to time in the future. Some of the factors that may cause these fluctuations include:
•economic conditions worldwide, which could affect the ability of hospitals and other customers to purchase our products and could result in a reduction in elective and non-reimbursed operative procedures;
•changes in import and export policies, including new, increased or retaliatory tariffs, sanctions and countersanctions and customs restrictions by the U.S. and foreign governments, which may disrupt our supply chain, adversely affect our relationships with customers, and impact our competitiveness;
•inspections of our manufacturing facilities for compliance with FDA’s Quality Management System Regulations (Good Manufacturing Practices), which have resulted in, and could result in future Form 483 observations, warning letters, injunctions or other adverse findings from the FDA or from equivalent regulatory bodies, and corrective actions, procedural changes and other actions that we determine are necessary or appropriate to address the results of those inspections, any of which may affect production and our ability to supply our customers with our products, including resuming the manufacture and sale of products subjection to prior voluntary recalls;
•the increased regulatory scrutiny of certain of our products, including products which we manufacture for others, could result in removal from the market or involve field corrective actions that could affect the marketability of our products;
•expenditures for major initiatives, including acquired businesses and integrations thereof, restructuring and the development and implementation of the CMP;
•various cost reduction initiatives to align our cost structure with our operations, improve operational performance and reduce costs might not provide the anticipated benefits on our expected timeline, or at all, and may yield unintended consequences, including business disruption, the loss of institutional knowledge as a result of turnover and reduced employee productivity
•the timing of significant customer orders, which tend to increase in the fourth quarter coinciding with the end of budget cycles;
•increased competition for a wide range of customers across all our product lines in the markets our products are sold;
•potential difficulties in recapturing market acceptance for any of our products which were subject of a voluntary recall and which we are now seeking to resume the manufacture and sale following the remediation of the issues which prompted the initiation of any such voluntary recall;
•market acceptance of our existing products, as well as products in development;
•retention of current employees and recruiting of new employees in light of market competition for talent and relevant skills;
•the timing of regulatory approvals as well as changes in country-specific regulatory requirements;
•changes in the exchange rates between the U.S. dollar and foreign currencies of countries in which we do business;
23
•changes in the variable interest rates of our debt instruments which could impact debt service requirements;
•an inability to refinance our indebtedness or to do so upon attractive terms could materially and adversely affect our business and results of operations, financial condition and cash flows;
•potential backorders, lost sales and expenses incurred in connection with product recalls or field corrective actions;
•disruption of our operations and sales resulting from political instability, war, insurrections, extreme weather conditions, the outbreak of disease, natural disasters, or other events outside our control that damage our manufacturing, distribution, or infrastructure of those facilities, or the suppliers and service providers for those facilities;
•our ability to manufacture and ship our products efficiently or in sufficient quantities to meet sales demands;
•changes in the cost or decreases in the supply of raw materials and services, including sterilization, energy, steel and honey;
•the timing of our research and development expenditures;
•reimbursement for our products by private and public health insurers, such as Medicare and Medicaid, and foreign governmental health systems;
•the impact of acquisitions, our ability to integrate acquisitions, and our restructuring activities including portfolio rationalization, and divestitures;
•risks related to public health concerns or crises, including epidemics and pandemics, which may negatively impact certain aspects of our business, including the demand for and supply of certain of our products, operations, supply chains and distribution systems, and our ability to generate cash flow;
•the ability to maintain existing distribution rights to and from certain third parties;
•the ability to maintain business if or when we opt to convert such business from distributors to a direct sales model;
•the ability of our commercial sales representatives to obtain sales targets in a reasonable time frame;
•the impact of changes to our sales organization and continued channel expansion, including increased specialization;
•peer-reviewed publications discussing the clinical effectiveness of the products we sell;
•changes in regulations or guidelines that impact the sales and marketing practices for products that we sell;
•enforcement or defense of intellectual property rights;
•changes in tax laws, or their interpretations; and
•the impact of goodwill and intangible asset impairment charges if future operating results of the acquired businesses are significantly less than the results anticipated at the time of the acquisitions.
Fluctuations in our operating results, including any of the above factors, may cause the market price of our common stock to fluctuate.
The industry and market segments in which we operate are highly competitive, and we may be unable to compete effectively with other companies.
There is intense competition among medical device companies. We compete with established medical technology companies in many of our products. Competition also comes from early-stage companies, universities, research institutions and other non-profit entities. In certain cases, our products compete primarily against medical practices that treat a condition without using a device or any particular product, such as the medical practices that use autograft tissue instead of our dermal regeneration products, duraplasty products and nerve repair products, or that use other technologies that cost less than our products. Many of our competitors have access to greater financial, technical, research and development, marketing, manufacturing, sales, distribution, administrative, consulting and other resources than we do. Our competitors may be more effective at developing commercial products or navigating the regulatory approval process in the markets in which we operate. In addition, unfavorable payment amounts or adverse coverage determinations of private and public health insurers, such as Medicare and Medicaid, and foreign governmental health systems, regarding our products or third-party determinations that favor a competitor’s product over ours, could harm acceptance or continued use of our products.
Our competitive position depends on our ability to achieve market acceptance for our products, develop new products, enhance existing products, implement marketing plans, secure regulatory approval for products under development and maintain previously-obtained approvals, demonstrate clinical and economic effectiveness, obtain and maintain funding, coverage and reimbursement under third-party payors and foreign governmental health systems, obtain patent protection and produce products consistently in sufficient quantities to meet demand. We may need to develop new applications for our products to remain competitive. Technological advances by one or more of our current or future competitors or their achievement of superior reimbursement from third-party payors and foreign governmental health systems could render our present or future products obsolete or uneconomical. Our future success will depend upon our ability to compete effectively against current
24
technology as well as to respond effectively to technological advances, changes in customers' requirements or in payor or regulatory evidence requirements. Additionally, purchasing decisions of our customers may be based on clinical evidence or comparative effectiveness studies and, because of our vast array of products, we might not be able to fund the studies necessary to gain entry or maintain our position or provide the required information to compete effectively. Other companies may have more resources available to fund such studies. For example, competitors have launched and are developing products to compete with our dural repair products, regenerative skin, neuro critical care monitors and ultrasonic tissue ablation devices, among others. In the current environment of managed care, consolidation among health care providers, increased competition, and declining reimbursement rates, we have been increasingly required to compete on the basis of price. Competitive pressures could adversely affect our profitability. Given these factors, we cannot guarantee that we will be able to compete effectively or continue our level of success in the areas in which we compete.
Changes in the healthcare industry may require us to decrease the selling price for our products, may reduce the size of the market for our products, or may eliminate a market, any of which could have a negative impact on our financial performance.
Trends toward managed care, healthcare cost containment and other changes in government and private sector initiatives in the U.S. and other countries in which we do business are placing increased emphasis on the delivery of more cost-effective medical therapies that could adversely affect the sale and/or the prices of our products. For example:
•third-party payors of hospital and physician services, including private and public health insurers, such as Medicare and Medicaid, and foreign governmental health systems, annually revise their payment methodologies, which can result in stricter standards for reimbursement of hospital charges for certain medical procedures or the elimination of reimbursement;
•several foreign countries have implemented reforms of their respective healthcare sectors in an effort to reduce healthcare spending, including restricting funding to only those medical technologies and procedures with proven effectiveness, increasing patient co-payments and providing for payback measures. Governmental health systems have revised and continue to consider revisions of healthcare budgets, which could result in stricter standards for implementing certain medical procedures, increased scrutiny of medical devices, and downward pricing pressure;
•Medicare, Medicaid, private and public health insurer and foreign governmental cutbacks could create downward pricing pressure on our products;
•in the U.S., Medicare and Medicaid coverage as well as commercial payor coverage determinations could reduce or eliminate reimbursement or coverage for certain of our wound matrix, amniotic, surgical reconstruction and advanced wound dressing products as well as other products in most regions, negatively affecting our market for these products, and future determinations could reduce or eliminate reimbursement or coverage for these products in other regions and could reduce or eliminate reimbursement or coverage for other products;
•there has been a consolidation among healthcare facilities and purchasers of medical devices in the U.S., some of whom prefer to limit the number of suppliers from whom they purchase medical products, and these entities may decide to stop purchasing our products or demand discounts on our prices;
•in the U.S., we are party to contracts with group purchasing organizations, which negotiate pricing for many member hospitals, require us to discount our prices for certain of our products and limit our ability to raise prices for certain of our products, particularly surgical instruments;
•there is economic pressure to contain healthcare costs in domestic and international markets, and, regardless of the consolidation discussed above, providers generally are exploring ways to cut costs by eliminating purchases or driving reductions in the prices that they pay for medical devices, implementing national and provincial tender pricing, such as the volume-based procurement policy implemented in China, or increasing clinical or economic evidence thresholds for product formularies;
•there are proposed and existing laws, regulations and industry policies in domestic and international markets regulating the sales and marketing practices and the pricing and profitability of companies in the healthcare industry;
•proposed laws or regulations may permit hospitals to provide financial incentives to doctors for reducing hospital costs, will award physician efficiency, and will encourage partnerships with healthcare service and goods providers to reduce prices; and
•there have been initiatives by third-party payors and foreign governmental health systems to challenge the prices charged for medical products that could affect our ability to sell products on a competitive basis.
Any and all of the above factors could materially and adversely affect our levels of revenue and our profitability.
We cannot guarantee that any of our acquisitions, investments or alliances will be successful.
25
We seek to supplement our internal growth through strategic acquisitions, investments and alliances. Such transactions are inherently risky, and the integration of any newly-acquired business requires significant effort and management attention. The success of any acquisition, investment or alliance may be affected by a number of factors, including our ability to properly assess and value the potential business opportunity or to successfully integrate any business we may acquire into our existing business. Even if the operations of an acquired business are integrated successfully, we may not realize the full benefits of such acquisition, including the synergies, cost savings or sales or growth opportunities, that we expect. There can be no assurance that any past or future transaction will be successful.
Our global business exposes us to certain operational, compliance and economic risks.
A significant portion of our current operations are conducted and located outside the United States, and our growth strategy involves expanding our existing foreign operations and entering into new foreign jurisdictions. We have significant manufacturing and distribution sites in North America, Europe, China and Japan. Our acquisition of Acclarent resulted in our acquiring research-and-development facilities and personnel located in Israel. As we seek to continue to expand and strengthen our international operations, we may experience difficulty in growing our sales in certain new markets and other international markets in which we are attempting to increase our presence due to, among other things, customer acceptance, undeveloped and/or unfamiliar distribution channels, regulatory restrictions and changes, and business knowledge of these markets.
The success of our operations outside the U.S. also depends, in part, on our ability to make necessary infrastructure enhancements to, among other things, our production facilities and sales and distribution networks, and our strategic staffing plans required to support our international operations. These and other factors may adversely impact our ability to pursue our growth strategy in these markets.
Our international operations increase our compliance risk, as such operations subject us to laws regarding sanctioned countries, entities and persons, customs, import-export, laws regarding transactions in foreign countries, the U.S. Foreign Corrupt Practices Act and local anti-bribery and other laws regarding interactions with healthcare professionals, and product registration requirements. Among other things, these laws restrict, and in some cases prevent, U.S. companies from directly or indirectly selling goods, technology or services to people or entities in certain countries. In addition, these laws require that we exercise care in structuring our sales and marketing practices and effecting product registrations in foreign countries. Global enforcement of anti-corruption and bribery laws has increased substantially in recent years, with more enforcement proceedings by foreign governmental agencies and the imposition of significant fines and penalties. While we have implemented policies, procedures and training related to compliance with these laws, our international operations, which often involve customer relationships with foreign governments, create the risk that there may be unauthorized payments or offers of payments made by employees, consultants, sales agents or distributors. Any alleged or actual violations of these laws may subject us to government investigations and significant criminal or civil sanctions and other liabilities, and negatively affect our reputation which could result in a material adverse effect on our business, results of operations, financial condition and cash flows.
The current tensions in international trade and rising international political tensions and uncertainties may materially adversely affect our business, financial condition, and results of operations.
Our operations and performance are significantly impacted by global, regional and U.S. economic and geopolitical conditions and increased trade barriers, including tariffs, from the U.S., China, the EU or other nations could materially adversely affect our business. The current tariff environment is dynamic and uncertain, as the U.S. government has imposed, modified and paused tariffs multiple times since the beginning of 2025. Changes to tariffs and other trade restrictions can be announced at any time with little or no notice. Additionally, potential tariffs or other U.S. trade policy measures have already prompted, and may prompt further, retaliatory actions by other countries, including by countries that are significant markets for our products, such as China. In addition, the U.S. Department of Commerce recently initiated an investigation under Section 232 of the Trade Expansion Act of 1962, as amended, into (among other things) imports of personal protective equipment, medical consumables and medical equipment (including devices), to determine whether they threaten U.S. national security, which further creates policy uncertainty in terms of tariffs.
The use of tariffs as a policy tool has created significant uncertainty about the future trading relationship among the U.S., China, the EU, Canada, Mexico and other exporting countries, including with respect to trade policies, treaties, government regulations and tariffs, and has led to concerns regarding the potential for extended trade barriers. The escalation of trade tensions could impact us in a variety of ways, including increases in manufacturing costs, disruptions or delays to our global supply chain, limitations on our ability to sell our products; and reductions in sales volumes and gross margins for our products, any of which could materially affect our business, financial condition and results of operations. Political uncertainty surrounding trade and other international disputes could also have a negative effect on customer confidence and spending, including capital equipment purchases by hospitals and some of our other customers. Changing our operations in accordance with new or changed trade restrictions may be expensive, time-consuming, disruptive to our operations and distracting to management. Further, such trade barriers could also lead to fluctuations in both the U.S. dollar and foreign currencies and our financial results may be materially adversely affected by such fluctuations.
26
Owing to the complex relationships between the U.S. and such other countries, political, diplomatic, military, or other events could result in business disruptions, including increased regulatory enforcement against companies, tariffs, trade embargoes, capital controls, export restrictions and the termination or modification of existing trade agreements. The imposition of such restrictions could increase the cost of the Company’s products and the components and raw materials that go into making them, require the Company to change its operations and the products it offers and negatively impact consumer confidence and spending, all of which, both individually and in the aggregate, could materially and adversely affect our business, results of operations and financial condition.
Exchange rate fluctuations and foreign currency hedges could adversely affect our financial results.
We generate significant revenues outside the U.S. in multiple foreign currencies, and in U.S. dollar-denominated transactions conducted with customers who generate revenue in currencies other than the U.S. dollar. For those foreign customers who purchase our products in U.S. dollars, currency fluctuations between the U.S. dollar and the currencies in which those customers do business may have a negative impact on the demand for our products in foreign countries where the U.S. dollar has increased in value compared to the local currency.
Since we have operations based outside the U.S. and we generate revenues and incur operating expenses in multiple foreign currencies, we experience currency exchange risk with respect to those foreign currency-denominated revenues and expenses. Our most significant currency exchange risk relates to transactions conducted in Euros, British pounds, Swiss francs, Canadian dollars, Japanese yen, Israeli shekel, Australian dollars and Chinese yuan.
We cannot predict the consolidated effects of exchange rate fluctuations upon our future operating results because of the number of currencies involved, the variability of currency exposure and the potential volatility of currency exchange rates. Although we address currency risk management through regular operating and financing activities, and, on a limited basis, through the use of derivative financial instruments, those actions may not prove to be fully effective. For a description of our use of derivative financial instruments, see Note 6. Derivative Instruments to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
Our future financial results could be adversely affected by impairments or other charges.
We are required to test both goodwill and indefinite-lived intangible assets for impairment on an annual basis based upon a fair value approach, rather than amortizing them over time. We are also required to test goodwill and indefinite-lived intangible assets for impairment between annual tests if an event occurs such as a significant decline in revenues or cash flows for certain products, or the discount rates used in the calculations of discounted cash flows change significantly, or circumstances change that would more likely than not reduce our enterprise fair value below its book value. If such a decline, rate change or circumstance were to materialize, we may record an impairment of these intangible assets that could be material to the financial statements. For example, during the second quarter of 2025 we recognized an aggregate charge of $511.4 million in goodwill impairment expense in our consolidated statement of operations following our completion of a quantitative assessment of our Tissue Technologies, Neurosurgery, and Instruments and ENT reporting units in accordance with ASC 350. The quantitative assessment was initiated following the decrease in the price per share of our common stock related to a number of factors including recent tariff changes that have created broad economic uncertainty and the impact of quality, operational, and supply issues. For more information concerning the goodwill impairment charge we recorded in 2025 and related matters, please see Item 7 “Management’s Discussion and Analysis of Financial Condition and Results of Operations - Critical Accounting Estimates” of this Annual Report on Form 10-K, and Note 7. Goodwill and Other Intangibles to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
The guidance on long-lived assets requires that we assess the impairment of our long-lived assets, including finite-lived intangible assets, whenever events or changes in circumstances indicate that the carrying value may not be recoverable as measured by the sum of the expected future undiscounted cash flows.
Also, Company decisions and other economic factors relating to our trade names may occur over time. For instance, we may discontinue certain products in the future as we continue to assess the profitability of our product lines. As a result, we may need to record impairment charges or accelerate amortization on certain trade names or technology-related intangible assets in the future.
The value of a medical device business is often volatile, and the assumptions underlying our estimates made in connection with our assessments under the guidance may change as a result of that volatility or other factors outside our control and may result in impairment charges. The amount of any such impairment charges could be significant and have a material, adverse effect on our reported financial results for the period in which the charge is taken and could have an adverse effect on the market price of our securities, including the notes and the common stock into which they may be converted.
We rely on independent suppliers and third-party providers in our supply chain for raw materials, packaging materials and components, sterilization services, and some finished goods; we could experience inventory shortages if any of these suppliers encounter a manufacturing or distribution disruption.
27
Outside vendors, some of whom are sole-source suppliers, provide key components, raw materials, packaging materials and components, sterilization services, and some finished goods used in the manufacture of our products. Although we believe that alternative sources for many of these components, raw materials, packaging materials and some finished goods are available, any interruption in supply of a limited or sole-source component or raw material could harm our ability to manufacture our products until a new or alternative source of supply is identified and qualified. In addition, an uncorrected defect or supplier’s variation in a component or raw material, either unknown to us or incompatible with our manufacturing process, could harm our ability to manufacture products and create inventory shortages of our products. We may not be able to find a sufficient alternative supplier in a reasonable time period, or on commercially reasonable terms, if at all, and our ability to produce and supply our products could be impaired. We believe that these factors are most likely to affect the following products that we sell:
•our collagen-based products and bovine-based products, such as the Integra Dermal Regeneration Template and wound matrix products, the DuraGen® family of products, our Absorbable Collagen Sponges, PriMatrix® and SurgiMend products;
•our products made from silicone, such as our neurosurgical shunts and drainage systems and hemodynamic shunts;
•products which use many different specialty parts, electrical components, or chemicals from numerous suppliers, such as our intracranial monitors, shunts, catheters, tissue ablation, and headlights;
•our biosynthetic products, including the DuraSeal sealant system and DuraSorb biosynthetic mesh scaffold;
•products which are amniotic tissue-based
•products which are porcine tissue-based;
•products that use medical grade leptospermum honey, such as our MediHoney products; and
•our TCC-EZ® total contact cast system products.
For example, in 2025, we initiated voluntary recalls of our MediHoney products due to a variety of packaging failures which compromised the sterility of such products. Although MediHoney supply resiliency has been incorporated into our broader CMP efforts to strengthen our supply chains, a definitive relaunch date for our MediHoney products remains unknown at this time.
The availability of amniotic tissue-based products depends upon, among other factors, the availability of tissue from human donors. Access to donated amniotic tissue could also be adversely impacted by regulatory changes or evolving public perceptions of the donor process.
Additionally, many of our products require sterilization by third-party suppliers. To the extent these suppliers are unable to provide sterilization services, whether due to lack of capacity, regulatory requirements, environmental concerns such as those relating to ethylene oxide or otherwise, we may be unable to transition sterilization to other suppliers in a timely or cost-effective manner, or at all, which could have an adverse impact on our operating results. For example, there is increased focus on the use and emission of ethylene oxide by the EPA and state environmental regulatory agencies. Additional regulatory requirements associated with the use and emission of ethylene oxide for sterilization may be imposed in the future, both domestically and outside the U.S. This increased regulation has required certain of the sterilization suppliers we use to temporarily suspend operations and it is possible that additional sterilization suppliers we use might also have to suspend operations, install additional emissions control technology, limit the use of ethylene oxide or take other actions, which would impact or further reduce the available capacity to sterilize medical devices and healthcare products. Although Integra has business continuity plans in place to mitigate the impact of any such disruptions, these plans may not be able to fully offset the full impact of such regulations.
Our supply chain and our cost of goods also may be negatively impacted by unanticipated price increases due to factors such as global economic disruptions, electronic component shortages, trade wars, inflation, including wage inflation, recessionary conditions, geopolitical conflict and instability, including wars and acts of terrorism, and fear of future or ongoing pandemics, all of which are beyond our control or the control of our suppliers.
While it is our policy to maintain sufficient inventory of components so that our production will not be significantly disrupted even if a particular component or material is not available for a period of time, we remain at risk that we will not be able to qualify new components or materials quickly enough to prevent a disruption if one or more of our suppliers ceases production of important components, materials or sterilization services.
We may experience difficulties, delays, performance impact or unexpected costs from consolidation of facilities and transfer of manufacturing facilities.
In recent years, we consolidated several facilities or transferred manufacturing operations (including between third parties, from third parties to our existing internal manufacturing facilities, and from our existing manufacturing facilities to new internal manufacturing facilities) and may further undertake similar consolidations or transfers in the future in order to improve our cost
28
structure, achieve increased operating efficiencies and reliability, and improve our competitive standing or results of operations and/or to address unfavorable economic conditions. For example, the Company has announced its plans to operationalize its Braintree, Massachusetts manufacturing facility (the “Braintree facility”) by the first half of 2026 and to transition the restart of the manufacture of PriMatrix and SurgiMend to the Braintree facility rather than attempt to restart the manufacture of these products at the Company’s Boston, Massachusetts manufacturing facility (the “Boston facility”). The consolidation, transfer or operationalization of manufacturing facilities requires such facilities to meet all applicable requirements related to quality control, quality assurance, and the maintenance of records and documentation as required by the FDA and any other applicable regulatory body both before and following the commencement of the manufacture of any approved product at such facility. For more information concerning the regulation related to our products, please refer to the risk factors appearing under the heading “—Risks Related to Our Regulatory Environment” below. As part of these initiatives, we may also lose favorable tax incentives or not be able to renew leases on acceptable terms. We may further reduce staff, make changes to certain capital projects, close certain production operations and abandon leases for certain facilities that will not be used in our operations. In conjunction with any actions, we will continue to make significant investments and build the framework for our future growth. We may not realize, in full or in part, the anticipated benefits and savings from these efforts because of unforeseen difficulties, delays, implementation issues or unexpected costs. If we are unable to achieve or maintain all of the resulting savings or benefits to our business or other unforeseen events occur, our business and results of operations may be adversely affected.
If any of our facilities or those of our suppliers were damaged and/or our manufacturing or business processes interrupted, we could experience lost revenues and our business could be seriously harmed.
Damage to our manufacturing, distribution, development and/or research facilities because of fire, extreme weather conditions, natural disaster, power loss, communications failure, geopolitical disruption, unauthorized entry or other events, such as a flu or other health epidemic, could significantly disrupt our operations, the operations of suppliers and critical infrastructure and delay or prevent product manufacture and shipment during the time required to repair, rebuild or replace the damaged facilities. Certain of our manufacturing facilities are located in Puerto Rico, which in the past has experienced both severe hurricanes and other natural disasters. Although we maintain property damage and business interruption insurance coverage on these facilities, our insurance might not cover all losses under such circumstances, and we may not be able to renew or obtain such insurance in the future on acceptable terms with adequate coverage or at reasonable costs. Moreover, climate change may increase both the frequency and severity of extreme weather conditions and natural disasters and, consequently, risks to our operations and growth.
Supply constraints have and may continue to adversely affect our ability to meet customer demand, and increase our costs to manufacture, transport and warehouse a certain subset of our products. In addition, supply constraints have resulted in increases to the costs of production of certain of our products that we may not be able to pass on to our customers. We expect these factors will continue to impact us in the future and obtaining alternative sources of raw materials and components could involve significant costs and regulatory challenges and may not be available to us on commercially reasonable terms, if at all.
We may have significant product liability exposure and our insurance may not cover all potential claims.
We are exposed to product liability and other claims if our technologies or products are alleged to have caused harm. We may not be able to obtain insurance for the potential liability on acceptable terms with adequate coverage or at reasonable costs. Any potential product liability claims could exceed the amount of our insurance coverage or may be excluded from coverage under the terms of the policy. Our insurance may not be renewed at a cost and level of coverage comparable to that then in effect.
Our private label product lines depend significantly on key relationships with third parties, which we could be unable to establish and maintain.
Our private label business depends in part on entering into and maintaining long-term supply agreements with third parties. The third parties with whom we have entered into agreements might terminate these agreements for a variety of reasons, including developing other sources for the products that we supply. The diminution or termination of our most important relationships could adversely affect our expectations for the growth of private label products.
RISKS RELATED TO OUR REGULATORY ENVIRONMENT
We are subject to stringent domestic and foreign medical device regulations and oversight and any adverse action may adversely affect our ability to compete in the marketplace and our financial condition and business operations.
Our medical devices and technologies, development activities and manufacturing processes are subject to extensive and rigorous regulation by numerous government agencies, including the FDA and comparable foreign agencies, as discussed in “Part 1, Item 1. Business – Government Regulation” of this Annual Report on Form 10-K. To varying degrees, each of these agencies monitors and enforces our compliance with laws and regulations governing the development, testing, manufacturing, labeling, marketing and distribution of our medical devices. We are also subject to regulations that may apply to certain of our products that are Drug/Device Combination products or are considered to be subject to pharmaceutical regulations outside the U.S. The process of obtaining marketing approval or clearance from the FDA and comparable foreign regulatory agencies for
29
new products, or for enhancements or modifications to existing products could be costly, time consuming and burdensome, may be impacted by failed clinical trials or weakened clinical evidence, involve modifications, repairs or replacements of our products and result in limitations on the indicated use of our products, which may negatively impact our ability to market our products and services, result in delays or prevent full commercial realization of future products or service. Furthermore, failure to obtain timely approvals, certifications or renewals may result in penalties and fines. Additional regulations govern the approval, initiation, conduct, monitoring, documentation and reporting of clinical studies to regulatory agencies in the countries or regions in which they are conducted. Failure to comply, could subject us to significant enforcement actions and sanctions, including halting the study, rejection of data generated in the study, seizure of investigational devices or data, sanctions against investigators, civil or criminal penalties, and other actions. In addition, without the data from one or more clinical studies, it may not be possible for us to secure the data necessary to support certain regulatory submissions, to secure reimbursement or demonstrate other requirements. We cannot assure that access to clinical investigators, sites and subjects, documentation and data will be available on the terms and timeframes necessary. We also cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. In addition, government shutdowns, recent reductions in U.S. government agency staffing and government spending more generally could impact ordinary course operations of agencies with which we interact routinely, such as the FDA. Following these reductions, the agencies may lack adequate staff and resources to meet current review, approval and inspection schedules, which could delay the receipt of or otherwise adversely affect the outcomes of product authorizations we seek.
We are subject to extensive complex regulatory requirements by domestic and foreign government agencies and any failure to comply with our ongoing responsibilities under their applicable laws and regulations could result in a material adverse impact on our business. Failure to comply with applicable regulations could result in reduced sales, increased costs, delays to new product introductions, harm to our reputation or competitiveness, future product recalls, injunctions preventing the shipment of products or other enforcement actions, all of which could have a material adverse effect on our business and financial results. In addition, the FDA has taken the position that device manufacturers are prohibited from promoting their products other than for the uses and indications set forth in the cleared or approved product labeling, and any failure to comply could subject us to significant civil or criminal exposure, administrative obligations and costs, and/or other potential penalties from, and/or agreements with, the federal government. Similar restrictions exist in many other countries where we do business, including the EEA.
As we disclosed above in “Part 1, Item 1. Business – Government Regulation,” we also are subject to the EU MDR, which was adopted by the EU as a common legal framework for all EU Member States (and also applies to Norway, Iceland and Liechtenstein). The EU MDR also sets out certain transitional provisions that permit certain legacy devices to remain on the market for a limited period, and failure to obtain, maintain, or timely transition required certifications under the EU MDR, including within those transitional periods, could restrict our ability to market certain products in the EEA and other jurisdictions that rely on EU certifications, and could adversely affect our business, financial condition, and results of operations.
Under the EU MDR, companies that wish to manufacture, import and distribute medical devices in EEA must meet certain quality system, performance and safety requirements as well as ongoing product monitoring responsibilities. Complying with the requirements of these regulations may require us to incur significant expenditures. Expenditures for EU MDR compliance activities amounted to $41.9 million for the year ended December 31, 2025 and we anticipate incurring additional expenditures in connection with our on-going efforts to obtain certification for our products under the EU MDR. Various penalties exist for non-compliance with the requirements of the EU MDR and the related laws of EEA countries which, if incurred, could have a material adverse impact on our business, results of operations and cash flows.
Further, the regulatory environment in China continues to evolve, and officials in the Chinese government exercise broad discretion in deciding how to interpret and apply regulations. It is possible that the Chinese government’s current or future interpretation and application of existing or new regulations will negatively impact our China operations, result in regulatory investigations or lead to fines or penalties.
Some of our activities may subject us to risks under federal and state laws prohibiting “kickbacks” and false or fraudulent claims.
We are subject to laws and regulations that govern the means by which companies in the healthcare industry may market their products to healthcare professionals and may compete by discounting the prices of their products, including for example, the federal AKS, the False Claims Act, HIPAA, state law equivalents to these federal laws that are meant to protect against fraud and abuse and analogous laws in foreign countries. Violations of these laws are punishable by criminal and civil sanctions, including, but not limited to, in some instances, civil and criminal penalties, damages, fines, restitution and exclusion from participation in federal and state healthcare programs, including Medicare and Medicaid. For a more detailed discussion of these laws, see “Item 1. Business — Government Regulation and Compliance — Other Regulations — Healthcare Fraud and Abuse Laws.”
30
The sales and marketing practices of our industry have been the subject of increased scrutiny from federal and state government agencies, and we believe that this trend will continue. Various hospital organizations, medical societies and trade associations are establishing their own practices that may require detailed disclosures of relationships between healthcare professionals and medical device companies or ban or restrict certain marketing and sales practices such as gifts and business meals. Since these laws, regulations and ultimate enforcement continue to evolve, we cannot predict with certainty, what, if any, impact, changes to them may have on our business or our customers.
Our international operations are subject to the provisions of the U.S. FCPA of 1977, which prohibits U.S. companies and their representatives from offering or making improper payments to foreign officials for the purpose of obtaining or retaining business. In many countries, the healthcare professionals we regularly interact with may meet the definition of a foreign official for purposes of the FCPA. Our international operations are also subject to various other international anti-bribery laws such as the UK Anti-Bribery Act. Despite meaningful measures that we undertake to facilitate lawful conduct, which include training and compliance programs and internal policies and procedures, we may not always prevent unauthorized, reckless or criminal acts by our employees or agents, or employees or agents of businesses or operations we may acquire. Violations of these laws, or allegations of such violations, could disrupt our operations, involve significant management distraction and have a material adverse effect on our business, financial condition and results of operations. We also could be subject to adverse publicity, severe penalties, including criminal and civil penalties, disgorgement, further changes or enhancements to our procedures, policies and controls, personnel changes and other remedial actions. Moreover, our failure to comply with domestic or foreign laws could result in various adverse consequences, including possible delay in approval or refusal to approve a product, recalls, seizures, and withdrawal of an approved product from the market.
Our medical device products are subject to reporting requirements and recalls, even after receiving regulatory clearance, approval or certification, which could harm our reputation, business and financial results.
Maintaining compliance with multiple regulators, and multiple centers within the FDA, adds complexity and cost to our manufacturing processes. Both before and after a device is placed on the market, numerous regulatory requirements apply, which require manufacturers to follow, among other things, design, testing, production, control, documentation and other quality assurance procedures during the manufacturing process; labeling regulations, which prohibit the promotion of unapproved products or unapproved or “off-label” uses of cleared or approved products and impose other restrictions on labeling; and medical device reporting regulations that require us to report to FDA or similar governmental bodies in other countries if our products are ineffective or may have caused or contributed to a death or serious injury or malfunction in a way that would be reasonably likely to contribute to death or serious injury if the malfunction were to recur. The FDA and similar governmental bodies in other countries have the authority to require the recall, repair, replacement, or refund of such products, refuse to grant pending pre-market approval applications or require certificates of non-U.S. governments for exports, and/or require us to notify health professionals and others that the devices present unreasonable risks of substantial harm to the public health, and in certain rare circumstances, ban medical devices. We may voluntarily recall a product if a reasonable possibility of serious injury or any material deficiency in a device is found, or withdraw a product to improve device performance or for other reasons. For example, in May 2023, after consultation with the FDA, we initiated a voluntary global recall of all products manufactured in our Boston facility distributed between March 1, 2018 and May 22, 2023. Additionally, in response to Form 483s issued to the Company by the FDA at the conclusion of the FDA’s inspection of three of the Company’s facilities located in Mansfield, Massachusetts, Plainsboro, New Jersey, and Princeton, New Jersey during June and August of 2024, the Company took a number of voluntary actions including the initiation of shipping holds for several products and a voluntary recall of certain disposable cottonoid patties and strips. In July 2024, we announced plans to implement an enterprise-wide CMP, a systematic and holistic approach to improving our quality management system across our manufacturing and supply network. We currently cannot predict with certainty whether we will be able to effectively implement the CMP and realize the benefits contemplated thereby within the anticipated timeframe, or at all. Further, there can be no assurance that the Company will build and operationalize the Braintree facility, transition manufacturing activities to the Braintree facility or realize the anticipated benefits of the Company’s consolidation of its efforts at Braintree on the planned timeline, or at all, which could have a material adverse effect on our business and financial results. For more information concerning our remediation efforts, including the status of the implementation of the CMP, and our expectations regarding the Company’s plans to build and operationalize the Braintree manufacturing facility and to transition manufacturing activities from the Boston facility to the Braintree facility and respond to the December 2024 Warning Letter, please see “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations - FDA Matters” in this Annual Report on Form 10-K.
Recalls of any of our products may divert managerial and financial resources and have an adverse effect on our financial condition and results of operations and cash flows. A recall might not only impact our results of operations and financial results but also could harm our reputation with customers and consumers which could reduce the future sales of our products. In addition, the FDA or other foreign governmental agencies may implement enforcement actions Any adverse regulatory action, depending on its magnitude, may place us under heightened scrutiny by the FDA and other regulators, including more frequent or more extensive inspections and increased monitoring of our quality and compliance activities, may restrict us from effectively marketing and selling our products and subject us to increased requirements for documentation or data in connection
31
with future submissions or otherwise limit our ability to obtain future pre-market clearances or approvals, and could result in a substantial modification to our business practices and operations.
The adoption of healthcare reform in the U.S. and initiatives sponsored by other governments may adversely affect our business, results of operations and/or financial condition.
Our operations may be substantially affected by potential fundamental changes in the global political, economic and regulatory landscape of the healthcare industry. Government and private sector initiatives to limit the growth of healthcare costs are continuing in the U.S., and in many other countries in which we do business, causing the marketplace to put increased emphasis on the delivery of more cost-effective treatments. These initiatives include price regulation, competitive pricing, coverage and payment policies, comparative effectiveness of therapies, technology assessments and managed-care arrangements. For example, the OBBBA is projected to decrease federal health care spending by approximately $1.0 trillion by reducing Medicaid spending and enrollment and making changes to federal Medicare spending. Congress also drafts and introduces, from time to time, legislation that could significantly change the statutory provisions governing the regulation of medical devices. In addition, the FDA may change its clearance and approval policies, adopt additional regulations or revise existing regulations, or take other actions, which may prevent or delay approval or clearance of our future products under development or impact our ability to modify our currently cleared products on a timely basis. The adoption of some or all of these initiatives could have a material, adverse effect on our financial condition and results of operations.
We cannot predict what impact ongoing uncertainty regarding federal and state health reform proposals, instability of the insurance markets, changes in the U.S. administration, laws and policies, an expansion in government’s role in and/or additional proposals and/or changes to the U.S. health care system will have on our customer’s purchasing decisions and/or reimbursement which could have a material adverse effect on our business. We expect that additional state and federal and foreign health care reform measures will be adopted in the future, including those initiatives affecting coverage and reimbursement for our products, any of which could limit the amounts that federal and state governments will pay for health care products and services, which could adversely affect the growth of the market for our products or demand for our products, or result in additional pricing pressures. We cannot predict the ultimate content, timing or effect of any healthcare reform legislation or policies or the impact on us. We continue to monitor the implementation of such legislation and, to the extent new market or industry trends or new governmental programs evolve, we will consider implementing or implement programs in response.
Certain of our products contain materials derived from animal sources and may become subject to additional regulation.
Certain of our products are derived from bovine or porcine tissue sources. As a result, we may experience difficulties in processing and producing our bovine and porcine tissue products at scale, including problems related to yields, quality control and assurance, tissue availability, adequacy of control policies and procedures and availability of skilled personnel.
With respect to bovine, among other products, our dermal regeneration products, duraplasty products, wound care products, bone void fillers, nerve and tendon repair products and certain other products, contain material derived from bovine tissue. In 2025, 26% of our revenues derived from products containing material derived from bovine tissue. Products that contain materials derived from animal sources, including food, pharmaceuticals and medical devices, are subject to scrutiny in the media and by regulatory authorities. Regulatory authorities are concerned about the potential for the transmission of disease from animals to humans via those materials. This public scrutiny has been particularly acute in Japan and Western Europe with respect to products derived from animal sources, because of concern that materials infected with the agent that causes bovine spongiform encephalopathy, otherwise known as BSE or mad cow disease, may, if ingested or implanted, cause a variant of the human Creutzfeldt-Jakob Disease, an ultimately fatal disease with no known cure. The World Organization for Animal Health recognizes the U.S. as having a negligible risk for BSE, which is the highest status available.
We take care to provide that our products are safe and free of agents that can cause disease. In particular, we qualified a source of collagen from a country outside the U.S. that is considered BSE/TSE-free. The World Health Organization classifies different types of bovine tissue for relative risk of BSE transmission. Deep flexor tendon and bovine fetal skin, which are used in our products, are in the lowest-risk categories for BSE transmission and are therefore considered to have a negligible risk of containing the agent that causes BSE (an improperly folded protein known as a prion). Nevertheless, products that contain materials derived from animals, including our products, could become subject to additional regulation, or even be banned in certain countries, because of concern over the potential for the transmission of prions. Significant new regulations, or a ban of our products, could have a material, adverse effect on our current business or our ability to expand our business.
Certain countries, such as Japan, China, Taiwan and Argentina, have issued regulations that require our collagen products be sourced from countries where no cases of BSE have occurred, and the EU has requested that our dural replacement products and other products that are used in neurological tissue be sourced from a country where no cases of BSE have occurred. Currently, we source bovine fetal hides from the U.S. and purchase tendon from the U.S. and New Zealand. New Zealand has never had a case of BSE. We received approval in the U.S., the EU, Japan, Taiwan, China, Argentina as well as other countries for the use of New Zealand-sourced tendon in the manufacturing of our products. If we cannot continue to use or qualify a
32
source of tendon from New Zealand or another country that has never had a case of BSE, we could be prohibited from selling our collagen products in certain countries.
We are subject to current and potential future requirements relating to protection of the environment, such as hazardous materials regulations, which may impose significant compliance or other costs on us.
Certain of our processes in manufacturing and research and development involve the controlled use of certain hazardous materials. In addition, we own and/or lease a number of facilities at which hazardous materials have been used in the past. Finally, we have acquired various companies that historically have used certain hazardous materials and that have owned and/or leased facilities at which hazardous materials have been used. For all of these reasons, we are subject to federal, state, foreign, and local laws and regulations governing the use, manufacture, storage, transportation, handling, treatment, remediation, and disposal of hazardous materials and certain waste products (“Environmental, Health, Safety and Transportation Laws”). Although we believe that our procedures for handling, transporting, and disposing of hazardous materials comply with the Environmental, Health, Safety and Transportation Laws, such laws may be amended in ways that increase our cost of compliance, perhaps materially.
Furthermore, the potential risk of accidental contamination or injury from these materials cannot be eliminated, and there is also a risk that such contamination previously has occurred in connection with one of our facilities or in connection with one of the companies we have purchased. In the event of such an accident or contamination, we could be held liable for any damages that result and any related liability could exceed the limits or fall outside the coverage of our insurance and could exceed our resources and could have a material impact on our operations and cash flows. We may not be able to maintain insurance on acceptable terms or at all.
Environmental, social and corporate governance issues, including those related to climate change and sustainability, may have an adverse effect on our business, financial condition and results of operations and damage our reputation.
There continues to be an increasing focus from foreign, federal, state and local regulatory and legislative bodies and certain investors, customers, consumers, employees and other stakeholders concerning environmental policies relating to climate change, regulating greenhouse gas emissions, sustainability, human rights and inclusion matters, and disclosure regarding the foregoing, many of which may be ambiguous, inconsistent, dynamic or conflicting. We expect to experience or be subject to increased restrictions, transition risks (from regulatory requirements or technology changes), compliance and assurance costs, recurring investments in data gathering and reporting systems, and legal expenses related to such new or changing legal or regulatory requirements, which could increase our operating costs. In addition, we may still be subject to penalties or potential litigation if such laws and regulations are interpreted or applied in a manner inconsistent with our practices.
Moreover, from time to time we establish and publicly announce environmental, social and governance aspirational goals and commitments. Implementation of our environmental, social and governance aspirational goals and initiatives involves risks and uncertainties, requires investments, and depends in part on third-party performance or data that is outside of our control. In addition, some stakeholders may disagree with the Company’s environmental, social and governance aspirational goals, targets or objectives. If we do not meet, are perceived not to meet, or if stakeholders disagree with, our environmental, social and governance aspirational goals, targets or objectives, we risk negative stakeholder reaction as well as damage to our reputation, reduced demand for our products, inability to attract capital investment from investors, inability to attract and retain employee talent or other negative impacts on our business, operations and financial condition.
If we do not retain our key personnel and attract and retain other highly skilled employees, our business could suffer.
If we fail to recruit, develop and retain the necessary personnel, our business and our ability to obtain new customers, develop new products, provide acceptable levels of customer service and achieve our research and development, operational or strategic or business objectives could suffer. The success of our business is heavily dependent on the leadership of our key management personnel. Our success also depends on our ability to recruit, develop and retain and motivate highly skilled sales, marketing, manufacturing, quality, regulatory and scientific personnel. Competition for these persons in our industry is intense, and we may not be able to successfully recruit, train or retain qualified personnel. In addition, we recognize that attracting, retaining and developing a diverse workforce is a critical success factor for our business. In that regard, we are continuously facing significant competition in our markets and at all levels in the workforce. We also continue to face the challenges of maintaining employee well-being, recognizing that the continued additional financial, family and health burdens that many employees may be experiencing due to macroeconomic uncertainties, including inflation, and other factors, may adversely impact job performance, employee engagement and employee retention. Additionally, in our industry, there is substantial competition for key personnel in the regions in which we operate and plan to expand our business. Labor shortages and competition for qualified personnel, particularly as employees are increasingly able to work remotely, could cause disruptions in our business operations. Also, facilitating seamless leadership transitions for key positions is a critical factor in sustaining the success of our organization. If we fail to effectively manage any organizational and/or strategic changes, our financial condition, results of operations, and reputation, as well as our ability to successfully attract, motivate and retain key employees, could be harmed.
33
RISKS RELATED TO TAX AND DEBT
We may have additional tax liabilities.
We are subject to income taxes in the U.S. and many foreign jurisdictions and are commonly audited by various tax authorities. In the ordinary course of our business, there are many transactions and calculations where the ultimate tax determination is uncertain. Significant judgment is required in determining our worldwide provision for income taxes. Although we believe that our tax estimates are reasonable, tax authorities may disagree with certain positions we have taken, and the final determination of tax audits and any related litigation could be materially different from our historical income tax provisions and accruals. In addition, economic and political pressures to increase tax revenue in various jurisdictions may make resolving tax disputes favorably more difficult. The results of an audit or litigation could have a material, adverse effect on our financial statements in the period or periods for which that determination is made and could result in the imposition of fines and penalties.
Changes in tax laws or exposures to additional tax liabilities could negatively impact the Company’s operating results.
We are subject to income taxes, as well as taxes that are not income-based, in both the U.S. and many foreign jurisdictions. Taxes could significantly increase due to changes in tax laws, changes in the interpretation or application of those laws, or differences in how tax authorities enforce those laws. The U.S. has not adopted the Organization for Economic Cooperation’s global minimum tax initiative (“Pillar Two”), and as of December 31, 2025, the G7 countries announced an agreement to exempt U.S. companies from certain elements of the Pillar Two framework. Although certain proposals could mitigate the impact on U.S.-based multinational companies, there is no assurance such proposals will be enacted, implemented consistently, or sustained. In addition, Pillar Two remains in effect in other countries, and there is significant uncertainty regarding the implementation of the G7 agreement, the interpretation and consistent application of existing Pillar Two rules, their interaction with national tax laws, and their consistency with current tax treaty obligations. We cannot provide any assurance that there will not be a material impact to our effective tax rate because of these developments and evolving tax legislation in 2025 and beyond.
Taxes could also significantly increase due to changes in accounting guidance. Our future effective tax rate could be unfavorably affected by numerous factors including a change in, or the interpretation of, tax rules and regulations in the jurisdictions in which we operate (including changes in legislation currently being considered), the expiration of or disputes about certain tax agreements in a particular jurisdiction, a change in our geographic earnings mix, and/or to the jurisdictions in which we operate, or a change in the measurement of our deferred taxes.
Our leverage and debt service obligations could adversely affect our business.
Our leverage and debt service obligations could adversely affect our business. As of December 31, 2025, our total consolidated external debt was approximately $1.9 billion (See Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations and Note 5. Debt, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for a discussion of our consolidated external debt). We may also incur additional indebtedness in the future. Our substantial indebtedness could have material, adverse consequences, including:
•making it more difficult for us to satisfy our financial obligations;
•increasing our vulnerability to adverse economic, regulatory and industry conditions, and placing us at a disadvantage compared to our competitors that are less leveraged;
•limiting our ability to compete and our flexibility in planning for, or reacting to, changes in our business and the industry in which we operate;
•limiting our ability to borrow additional funds for working capital, capital expenditures, acquisitions and general corporate or other purposes; and
•result in greater interest rate risk and volatility.
Our debt service obligations will require us to use a portion of our operating cash flow to pay interest and principal on indebtedness instead of for other corporate purposes, including funding future expansion of our business, acquisitions, and ongoing capital expenditures, which could impede our growth. Our ability to comply with, renegotiate or extend the Company’s debt obligations will depend on various factors, including the accessibility of the capital markets and our operating and financial performance, which, in turn, is subject to prevailing economic conditions and financial, business and other factors beyond our control. Our Senior Credit Facility (as defined below) has a maturity date of March 24, 2028 (See Note 5. Debt, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for a discussion of our consolidated external debt, including the Senior Credit Facility). We may seek to refinance all of our outstanding debt in 2026 and we may not be able to refinance our existing debt on terms acceptable to us or at all. Any disruptions in our operations, the financial markets, or the overall economy, may adversely affect the availability and cost of credit to us and/or our ability to comply with our existing obligations.
34
In addition, prevailing interest rates or other factors at the time of refinancing could increase our interest expense. A refinancing of our indebtedness could also require us to comply with more onerous covenants and further restrict our business operations. Our inability to refinance our indebtedness or to do so upon attractive terms could materially and adversely affect our business, prospects, results of operations, financial condition and cash flows, and make us vulnerable to adverse industry and general economic conditions.
RISKS RELATED TO OUR INTELLECTUAL PROPERTY
Our intellectual property rights may not provide meaningful commercial protection for our products, potentially enabling third parties to use our technology or very similar technology and could reduce our ability to compete in the market.
To compete effectively, we depend, in part, on our ability to maintain the proprietary nature of our technologies and manufacturing processes, which includes the ability to obtain, protect and enforce patents on our technology and to protect our trade secrets. We own or have licensed patents that cover aspects of some of our product lines. Our patents, however, may not provide us with any significant competitive advantage. Others may challenge our patents and, as a result, our patents could be narrowed, invalidated or rendered unenforceable. Competitors may develop products similar to ours that our patents do not cover. In addition, the approval or rejection of patent applications may take several years and our current and future patent applications may not result in the issuance of patents in the U.S. or foreign countries.
Our competitive position depends, in part, upon unpatented trade secrets, which we may be unable to protect.
Our competitive position also depends upon unpatented trade secrets, which are difficult to protect. We cannot assure that others will not independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets, that our trade secrets will not be disclosed or that we can effectively protect our rights to unpatented trade secrets.
In an effort to protect our trade secrets, we require our employees, consultants and advisors to execute confidentiality and invention assignment agreements upon commencement of employment or consulting relationships with us. These agreements provide that, except in specified circumstances, all confidential information developed or made known to the individual during the course of their relationships with us must be kept confidential. We cannot assure, however, that these agreements will provide meaningful protection for our trade secrets or other proprietary information in the event of the unauthorized use or disclosure of confidential information.
Our success will depend partly on our ability to operate without infringing or misappropriating the proprietary rights of others.
We may be sued for infringing the intellectual property rights of others. In addition, we may find it necessary, if threatened, to initiate a lawsuit seeking a declaration from a court that we do not infringe the proprietary rights of others or that their rights are invalid or unenforceable. If we do not prevail in any litigation, in addition to any damages we might have to pay, we would be required to stop the infringing activity (which could include a cessation of selling the products in question) or obtain a license for the proprietary rights involved. Any required license may be unavailable to us on acceptable terms, if at all. In addition, some licenses may be nonexclusive and allow our competitors to access the same technology we license.
If we fail to obtain a required license or are unable to design our products so as not to infringe on the proprietary rights of others, we may be unable to sell some of our products, and this potential inability could have a material, adverse effect on our revenues and profitability and cash flows.
We may be involved in lawsuits relating to our intellectual property rights and promotional practices, which may be expensive.
The medical device industry is characterized by extensive intellectual property litigation and to protect or enforce our intellectual property rights, we may have to initiate or defend legal proceedings, such as infringement suits or opposition proceedings, against or by third parties. In addition, we may have to institute proceedings regarding our competitors’ promotional practices or defend proceedings regarding our promotional practices. Legal proceedings are costly, and, even if we prevail, the cost of the legal proceedings could affect our profitability and cash flows. In addition, litigation is time-consuming and could divert management’s attention and resources away from our business. Moreover, in response to our claims against other parties, those parties could assert counterclaims against us.
RISKS RELATED TO CYBERSECURITY AND DATA PRIVACY
Cybersecurity incidents or other disruptions to our information technology systems could adversely affect our business.
We are increasingly dependent on sophisticated information technology for our infrastructure and to support business decisions. Our information systems require an ongoing commitment of significant resources to maintain, protect, and enhance existing systems and develop new systems to keep pace with continuing changes in information processing technology, evolving systems and regulatory standards, the increasing expectations regarding protection of patient, customer, and employee
35
information, and changing customer patterns. An experienced third party maintains the enterprise business system used to support our transaction processing, accounting and financial reporting, and supply chain and manufacturing processes. Any significant breakdown, intrusion, interruption, corruption, or destruction of these systems, as well as any other cybersecurity incident, could have a material, adverse effect on our business. A cybersecurity incident may also result in improper use of our information systems, unauthorized access, use, disclosure, modification or destruction of information, defective products, production downtimes and operational disruptions.
Cybersecurity incidents may involve social engineering/phishing, cyber-attacks (including ransomware, malware attacks, unauthorized access attempts, and denial of service and other unintentional intrusions or malicious cyber-attacks), cyber extortion or other fraudulent schemes, or attempts to exploit vulnerabilities and may result in a compromise of our information systems, including unauthorized access to data relating to patients, proprietary or other sensitive information. We may face increased cybersecurity risks due to our reliance on internet technology and the number of our employees who are working remotely, which may create additional opportunities for cybercriminals to exploit vulnerabilities. If we, or third parties on whom we rely, fail to maintain or protect our information systems and data effectively, we could lose existing customers, have difficulty attracting new customers, suffer backlash from negative public relations, experience increased regulatory scrutiny or have regulatory sanctions or penalties imposed, have increases in operating expenses, incur expenses or lose revenues as a result of a data privacy incident (including from class action settlements or awards), or suffer other adverse consequences (including from litigation, class action settlements, or awards), have increased cybersecurity protection and insurance costs, miss reporting deadlines, or suffer other adverse consequences. For example, a vulnerability affecting a third-party provider’s enterprise software we use was exploited in a threat actor campaign that affected numerous of the third-party provider’s customers. As a result, we have an ongoing investigation to assess the nature and scope of any impacted Company data. Several putative class action lawsuits also have been filed relating to this incident, including against us, and additional litigation, regulatory inquiries, or claims may arise and we cannot predict the ultimate outcome, scope or impact of this matter.
We have programs, processes (including ongoing improvements) and technologies in place to prevent, detect, contain, respond to and mitigate cybersecurity related threats and potential incidents. Because the techniques used to obtain unauthorized access or interrupt services change frequently and can be difficult to detect, anticipating, identifying or preventing these threats or mitigating them if and when they occur, may be challenging. And as increased regulatory compliance for cybersecurity protocols and disclosures are required or expected by regulatory authorities, there is no guarantee that the increased amount of resources, both time and expense, will not adversely affect our business, or that a court or regulator will agree that the measures we have put in place are reasonable, appropriate, or adequate. Further, adoption of artificial intelligence (“AI”) tools by us or by third parties may pose new cybersecurity challenges. Threat actors may use AI tools to automate and enhance cybersecurity attacks against us. We use software and platforms designed to detect such cybersecurity threats but these threats could become more sophisticated and harder to detect and counteract, which may pose significant risks to our data security and systems. We are also dependent on third party vendors to supply and/or support certain aspects of our information technology systems which may contain defects in design or manufacture or other problems that could result in system disruption or unexpectedly compromise the information security of our own systems. In addition, as we grow in part through new acquisitions we may face risks due to implementation, modification, or remediation of controls, procedures, and policies relating to data privacy and cybersecurity at the acquired business that may or may not have been identified as part of due diligence or encounter issues as part of integration. We continue to consolidate and integrate the number of information systems we operate, and to upgrade and expand our information system capabilities for stable and secure business operations. While we have obtained cybersecurity insurance, there are no assurances that the coverage would be adequate in relation to any incurred losses. Moreover, as cyber attacks increase in frequency and magnitude, we may be unable to obtain cybersecurity insurance in amounts and on terms we view as adequate for our operations in the future.
Failure to comply with laws relating to the confidentiality of sensitive personal information or standards related to the transmission of electronic health data, including in international jurisdictions, may require us to make significant changes to our products, or incur penalties or other liabilities.
State, federal and foreign laws, such as HIPAA, Section 5 of the FTC Act, or the California Consumer Privacy Act and other similar state laws regulate the confidentiality of personal information, including sensitive information and the circumstances under which such information may be released. These measures may govern the disclosure and use of personal and patient medical record information and may require users of such information to implement specified security measures and to notify individuals in the event of privacy and cybersecurity incidents. Evolving laws and regulations in this area could restrict the ability of our customers to obtain, use or disseminate patient information, or could require us to incur additional costs to re-design our products in a timely manner, either of which could have an adverse impact on our results of operations. Other health information standards, such as regulations under HIPAA and FDA guidance and requirements, establish standards regarding device security, electronic health data transmissions and transaction code set rules for specified electronic transactions, for example transactions involving submission of claims to third-party payors. These standards also continue to evolve and are often unclear and difficult to apply. We have incurred and expect that we will continue to incur costs implementing additional
36
security measures to protect against new or enhanced data security or privacy threats, or to comply with current and new federal, state and international laws governing data privacy and cybersecurity which are frequently being enacted and proposed. Moreover, as a result of the broad scale release and availability of AI technologies such as generative AI, there is a global trend towards more regulation (e.g., the EU AI Act and AI laws passed in U.S. states) designed to ensure the ethical use, privacy, and security of AI and the data that it processes. Compliance with such laws will likely be an increasing and substantial cost in the future. Outside the U.S., we are also impacted by privacy and data security requirements at the international, national and regional level, and on an industry specific basis.
See “Item 1. Business – Government Regulation and Compliance – Other Regulations – Data Privacy and Cybersecurity Laws and Regulations.” Failure to maintain the confidentiality of personal data in accordance with the applicable regulatory requirements, or to abide by electronic health data transmission standards, could expose us to breach of contract claims, regulatory fines and penalties, (which may be significant in EU or other non-US jurisdictions), litigation expenses, costs for remediation and harm to our reputation.
ITEM 1B. UNRESOLVED STAFF COMMENTS
As of the filing of this Annual Report on Form 10-K, we had no unresolved comments from the staff of the SEC that were received not less than 180 days before the end of our 2025 fiscal year.
ITEM 1C. CYBERSECURITY
Information Technology and Cybersecurity
Our business relies on the secure electronic transmission, storage and hosting of information, including personal information, financial information, intellectual property, and other sensitive information related to our business, customers and workforce (“sensitive information”) on both our information systems and those of third-party service providers. Given the importance of cybersecurity to our business, we maintain a robust information technology and cybersecurity program to increase both the effectiveness of our systems and our preparedness for cybersecurity risks, including security monitoring for internal and external threats to bolster the confidentiality, integrity and availability of our information assets. We perform evaluations of our cybersecurity program, including periodic internal and external audits, penetration tests and incident response simulations, and our information technology infrastructure and cybersecurity management system are subject to external program assessments on a regular basis. In addition, we require new employees to complete cybersecurity training so they are better able to understand how to identify, protect, and preserve sensitive data and minimize risks related to cybersecurity matters. We supplement this new hire training with annual training and certification programs, which includes social engineering simulations. We continue to expand and improve our global training programs to raise employee awareness of security obligations and members of senior management regularly provide employees with communications regarding the cybersecurity environment to increase employee awareness of cybersecurity trends and emerging risks. We have designed our cybersecurity program to leverage core principles of the National Institute of Standards and Technology Cybersecurity Framework (“NIST CSF”) to help inform our cybersecurity management system and reduce cybersecurity risks, although we do not fully implement all aspects of NIST CSF.
Management and Board Oversight
37
Processes for Assessing, Identifying and Managing Material Risks from Cybersecurity Threats
Our monitoring capabilities, including our internal auditing procedures, internal control over financial reporting and corporate compliance programs, are designed in part to inform management about our significant risks, including those related to cybersecurity risks. In the event of a cybersecurity incident, we maintain a regularly tested incident response program. Pursuant to the incident response program and its escalation protocols, designated personnel are responsible for assessing the severity of the cybersecurity incident and associated threat, containing the threat, remediating the threat, including recovery or data and access to systems, analyzing the reporting and disclosure obligations associated with the incident, and performing post-incident analysis and program improvements. Although the particular personnel assigned to an incident response team will depend on the particular facts and circumstances, the team is generally led by the CIO or another member of the cybersecurity executive steering committee and will include other information technology and legal personnel. For incidents that meet certain characteristics under the program, the incident response team will escalate and update both the Company’s Board and members of senior management, including the Chief Executive Officer, Chief Financial Officer and Chief Legal Officer.
To support current needs and future growth, we use a strategic information systems multi-year planning process that involves senior management and is integrated into our overall business planning. Information systems projects, including those for cybersecurity, are prioritized based upon strategic, financial, regulatory, risk and other business advantage criteria.
Cybersecurity Risks
ITEM 2. PROPERTIES
As of December 31, 2025, we lease approximately 166,991 square feet of space in Princeton, NJ, where we house our principal headquarters, sales operations, and support functions. This lease expires in 2035.
We own facilities in Saint Aubin Le Monial, France, Rietheim-Weilheim, Germany, Plainsboro, New Jersey and Cincinnati, Ohio and we lease all of our other facilities.
We have key manufacturing and research facilities located in California, Maryland, Massachusetts, New Jersey, Ohio, Puerto Rico, Tennessee, Utah, France, Germany, Ireland, Israel, and Switzerland. Our instrument procurement operations are located in Germany.
Our primary distribution centers are located in Kentucky, Nevada, Australia, Belgium, Canada, Italy, Japan, and China. In addition, we lease several smaller facilities to support additional administrative, assembly, and distribution operations.
In addition to the Company’s primary operations, we have dedicated repair facilities located in multiple countries around the world.
38
Our manufacturing facilities are registered with the FDA. Our facilities are subject to FDA inspection to ensure compliance with Quality System regulations. For further information regarding the status of FDA inspections, see the “Item 1. Business –Government Regulation and Compliance” and “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations – FDA Matters” in this Annual Report on Form 10-K.
ITEM 3. LEGAL PROCEEDINGS
Please refer to Note 15. Commitments and Contingencies, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information, Holders and Dividends
Our common stock trades on The Nasdaq Global Select Market under the symbol “IART.” The number of stockholders of record as of February 25, 2026 was approximately 675, which includes stockholders whose shares were held in nominee name.
Dividend Policy
We have not paid any cash dividends on our common stock since our formation. Our Senior Credit Facility (as defined below) limits the amount of dividends that we may pay. Any future determinations to pay cash dividends on our common stock will be at the discretion of the Board and will depend upon our financial condition, results of operations, cash flows and other factors deemed relevant by the Board.
Sales of Unregistered Securities
There were no sales of unregistered securities during the years ended December 31, 2025, 2024 or 2023.
Sale of Registered Securities
There were no sales of registered securities during the years ended December 31, 2025, 2024, or 2023.
Issuer Purchases of Equity Securities
On December 31, 2025, our share repurchase program expired with approximately $50.0 million remaining available for repurchase. Upon expiration of the existing share repurchase program, we did not adopt a new share repurchase program. During the three months ended December 31, 2025, we did not repurchase any of our equity securities.
Securities Authorized for Issuance under Equity Compensation Plan
The information required by this item regarding our equity compensation plans is incorporated herein by reference to Item 12 of Part III of this Annual Report on Form 10-K.
39
Stock Performance Graph
The graph below compares the five-year total return to stockholders on our common stock with the return of the Standard & Poor’s (“S&P”) 500 Stock Index and the S&P Healthcare Equipment Index for the five years ended December 31, 2025. The Company’s cumulative shareholder return is based on an investment of $100 on December 31, 2020 and is compared to the cumulative total return of the S&P indices mentioned above over the period with a like amount invested. Measurement points are the last trading day of each respective fiscal year.
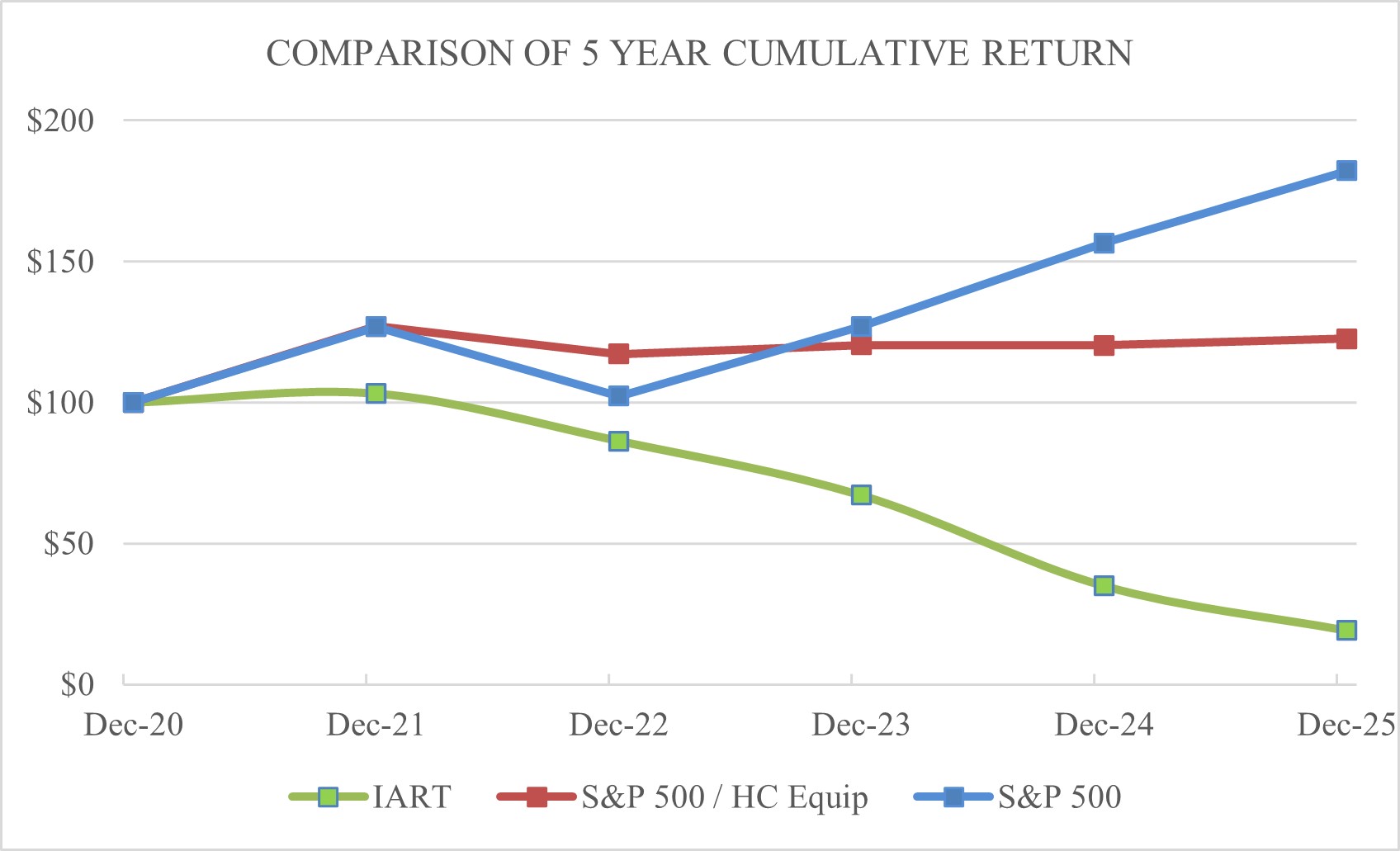
Note: The stock price performance shown on the graph above is not indicative of future price performance. This graph shall not be deemed filed for purposes of Section 18 of the Exchange Act or otherwise subject to the liabilities of that section nor shall it be deemed incorporated by reference in any filing under the Securities Act or the Exchange Act, regardless of any general incorporation language in such filing.
ITEM 6. [Reserved]
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis provides information management believes to be relevant to understanding our financial condition and results of operations. For a full understanding of financial condition and results of operations, it should be read together with the selected audited consolidated financial data and our financial statements with the related notes appearing elsewhere in this report. The discussion focuses on our financial results for the year ended December 31, 2025 and 2024. The comparison of fiscal 2024 to 2023 has been omitted from this Form 10-K, but can be referenced in our Form 10-K for the fiscal year ended December 31, 2024—“Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations” filed with the SEC on February 25, 2025.
We have made statements in this report which constitute forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Exchange Act. These forward-looking statements are subject to a number of risks, uncertainties and assumptions about the Company and other matters. The Company’s actual results could differ materially from those anticipated in these forward-looking statements as a result of many factors, including but not limited to those set forth under Item 1A. Risk Factors. Please refer to “Special Note Regarding Forward-Looking Statements” and Item 1A. Risk Factors for a discussion of the factors that could cause actual results to differ materially from those projected in these statements. The following information concerning our business, results of operations and financial condition should also be read in conjunction with the information included under Item 1. Business, Item 1A. Risk Factors and Item 15. Exhibits and Financial Statement Schedules.
40
GENERAL
Integra LifeSciences Holdings Corporation is a global medical technology company dedicated to restoring lives. We are advancing transformational care through impactful innovation and our portfolio of highly differentiated technologies is trusted by healthcare professionals to deliver transformative care.
We manufacture and sell medical technologies and products in two reportable business segments: Codman Specialty Surgical (“CSS”) and Tissue Technologies (“TT”). The CSS segment, which represents approximately 70% of our total revenue, consists of market-leading technologies and instrumentation used for a wide range of specialties, such as neurosurgery, neurocritical care, and otolaryngology, commonly referred to as ear, nose, and throat (“ENT”). We are the world leader in neurosurgery and one of the top three providers in the U.S. in instruments used in precision, specialty, and general surgical procedures. Our TT segment generates about 30% of our overall revenue and focuses on wound reconstruction and care and private label.
NEW PRODUCT INTRODUCTIONS AND RESEARCH AND DEVELOPMENT
We continue to invest in collecting clinical evidence to support our existing products and new product launches, and to ensure that we obtain market access for broader and more cost-effective solutions.
Neurosurgical Solutions, Surgical Instruments, and ENT Solutions. The CSS neurosurgical business consists of a broad portfolio of market-leading brands, which are used for the management of multiple disease states, including brain tumors, traumatic brain injury, hydrocephalus and other neurological conditions. The growth in this business in recent years has been fueled by geographic expansion and new product registrations in markets, such as China, Japan, and Europe, which we expect to continue in the near-to-long term. We have several active programs focused on life cycle management and innovation for capital and disposable products in our portfolio. Our product development efforts are focused on core clinical applications in cerebrospinal fluid (“CSF”) management, neuro-critical care monitoring, minimally invasive instruments and electrosurgery and ultrasonic medical technologies, as well as our ambition to transform the standard of care in neurosurgery with product advancements in minimally invasive surgery (“MIS”) and the surgical management of intracerebral hemorrhage (“ICH”).
We continue to advance the CerebroFlo® external ventricular drainage (“EVD”), a catheter with Endexo® technology. The Endexo polymer in polyurethane is a permanent additive which has shown to be effective in reducing platelet adhesion in-vitro, reducing thrombus accumulation in-vitro and in vivo, and reducing the clinical incidence of thrombus formation. In vitro evaluations and in vivo animal evaluations do not necessarily predict the clinical performance of the SureFlo EVD Catheter with respect to thrombus formation. The incidence of thrombus formation on polyurethane containing Endexo polymer in other medical devices and/or tissues systems does not necessarily predict the clinical performance of the SureFlo EVD Catheter for the intended use of CSF external drainage and monitoring. The CerebroFlo EVD catheter has demonstrated an average of 99% less thrombus accumulation onto its surface, in vitro, compared to a market leading EVD catheter. Our work to combine our Bactiseal antimicrobial technology with the Endexo anti-occlusive technology continues to progress for both a silicone-based hydrocephalus and EVD product.
We also continue to advance the Aurora® Surgiscope, which is the only tubular retractor system designed for cranial surgery with an integrated access channel, camera and lighting. The 15mm x 60mm and 15mm x 80mm Aurora Surgiscope System version received 510(k) clearance from the FDA in 2025.
In July 2025, we announced the inaugural enrollment of the first patient in the AERA Pediatric Registry, a prospective, multi-center observational registry evaluating the real-world use of the AERA Eustachian Tube Balloon Dilation System in children. This marks the focused effort to measure the ongoing, clinical performance of AERA in pediatric patients with obstructive Eustachian tube dysfunction. The registry is designed to capture both safety and efficacy outcomes for up to 300 pediatric patients who undergo Eustachian tube balloon dilation using AERA.
In September 2025 the Mayfield® Ghost Base Unit Post launched in the U.S., which is designed to help provide clear visualization of anatomical structure and to support surgical accuracy and patient positioning.
Regenerative Technologies. Our regenerative technology development program applies our expertise in bioengineering to a range of biomaterials including natural materials such as purified collagen, intact human or animal tissues, honey as well as resorbable synthetic polymers with our DuraSorb and DuraSeal® product lines. These unique product designs are used for neurosurgical and reconstructive surgical applications, as well as dermal regeneration. Our regenerative technology platform includes our legacy Integra® Dermal Regeneration Template (“IDRT”) products and complementary technologies that we have acquired. Our collagen manufacturing capability, combined with our history of innovation, provides us with strong platform technologies for multiple indications.
In the third quarter of 2021, we filed a PMA application for a specific indication for SurgiMend® in the use of post-mastectomy breast reconstruction and in July 2024 received approvable pending GMP status from FDA, which approved and closed out the clinical portion of this PMA application. We anticipate PMA approval following the operationalization of the Braintree facility, which is expected in 2026. We are also pursuing a PMA for DuraSorb for use in implant-based breast reconstruction (“IBBR”).
41
We completed enrollment for the DuraSorb U.S. investigational device exemption clinical study for two-stage breast reconstruction in June 2023; and we continue to advance the PMA application. Currently, we hope to secure PMA approval for DuraSorb in 2026.
In 2024, we acquired the product rights for Durepair Dural Regeneration Matrix, a suturable dural graft which complements our portfolio of dural grafts and sealants, and subsequently launched the product for commercial sale in the U.S. in October 2025.
EUROPEAN UNION MEDICAL DEVICE REGULATION UPDATES
We continue to work towards certifying our products under the EU MDR. In recent years, we received EU MDR certification in our CSS segment for Hakim Programmable Valves, Certas Plus with and without Bactiseal catheters, Surgical Patties and Strips, DuraSeal Dural, CUSA Clarity and DuraGen Suturable, Cranial Drills and Perforators, as well as IDRT, BioPatch, MicroMatrix, and Cytal in our TT segment. We do not currently anticipate any significant disruption to our commercial activities in Europe related to EU MDR.
FDA MATTERS
On December 19, 2024, the Company received a warning letter from the FDA (the “2024 Warning Letter”). The 2024 Warning Letter relates to quality system issues identified during FDA inspections at three of the Company’s facilities located in Mansfield, Massachusetts; Plainsboro, New Jersey; and Princeton, New Jersey. The 2024 Warning Letter did not identify any new observations that had not already been provided in the Form 483s previously issued to the Company by the FDA at the conclusion of its three inspections in June and August of 2024 (the “2024 Form 483s”). In the 2024 Form 483s, the FDA deemed certain of the Company’s devices, including cranial perforators, disposable cottonoid patties and strips, and collagen-based products, to be out of compliance with respect to quality system regulations. At that time, the Company took a number of voluntary actions including the initiation of shipping holds for several products and a voluntary recall of the disposable patties and strips. The 2024 Warning Letter does not restrict the Company’s ability to manufacture or ship products, require recall of any products, nor restrict the Company’s ability to seek FDA 510(k) clearance of products. The 2024 Warning Letter states that premarket approval applications for Class III devices to which the quality system regulation violations are reasonably related will not be approved until the violations have been corrected. The Company has submitted several responses to the 2024 Form 483s issued to each of the three manufacturing facilities to the FDA and has submitted several updates to the 2024 Warning Letter throughout 2025.
On March 7, 2019, TEI Biosciences, Inc. (“TEI”), one of our wholly-owned subsidiaries, received a Warning Letter (the “2019 Warning Letter”), dated March 6, 2019, from the FDA. The 2019 Warning Letter was related to quality systems issues at TEI’s manufacturing facility located in Boston, Massachusetts (the “Boston facility”). The Boston facility manufactured extracellular bovine matrix products in our TT segment that were sold both in wound reconstruction and care and surgical reconstruction franchises, and in private label channels. The 2019 Warning Letter resulted from an inspection held at the Boston facility in October and November 2018 and did not identify any new observations that were not already provided in the Form 483 that followed the inspection. We submitted our initial response to the 2019 Warning Letter on March 28, 2019 and provide regular progress reports to the FDA as to our corrective actions. On October 28, 2021, the FDA initiated an inspection of the Boston facility and at the conclusion of the inspection, issued an FDA Form 483 on November 12, 2021 (the “2021 Form 483”). On March 1, 2023, the FDA commenced an inspection of the Boston facility and issued an FDA Form 483 at the conclusion of this inspection (the “2023 Form 483”). In May 2023, after consultation with the FDA, the Company initiated a voluntary global recall of all products manufactured at the Boston facility, including PriMatrix, SurgiMend, Revize™, and TissueMend™, distributed between March 1, 2018 and May 22, 2023 (the “Boston recall”). On July 19, 2023, TEI received a Warning Letter, dated July 17, 2023, from the FDA related to quality system issues at the Boston facility (the “2023 Warning Letter”). The 2023 Warning Letter did not identify any new observations that had not already been provided in the 2023 Form 483. The Company has submitted periodic responses to the FDA for both the 2023 Form 483 and the 2023 Warning Letter. We are committed to resolving the matters identified in the warning letters and Form 483s and are continuing significant efforts to remediate the observations.
Although the warning letters do not restrict the Company’s ability to seek FDA 510(k) clearance of products, PMAs for Class III devices to which the quality system regulation violations are reasonably related will not be approved until the violations have been addressed. Following its assessment of the results of a third-party audit of the Boston facility, the Company announced in the second quarter of 2024 that it no longer planned to restart the manufacture of PriMatrix and SurgiMend at its Boston facility. The restart of the manufacturing of SurgiMend will occur at the Company’s new tissue manufacturing facility in Braintree, Massachusetts (the “Braintree transition”). The Company anticipates the Braintree facility to be operational in 2026. In addition, the Company entered into a new third-party agreement, which facilitated the relaunch of PriMatrix, as well as Durepair Dural Regeneration Matrix, ahead of previously disclosed timelines.
42
We cannot give any assurances that the FDA will be satisfied with our response to the issues identified by the FDA in any of the foregoing Form 483s or warning letters or as to the expected date of the resolution of such issues. Until the issues cited by the FDA are resolved to the FDA’s satisfaction, the FDA may initiate additional regulatory action without further notice. Any adverse regulatory action, depending on its magnitude, may restrict us from effectively manufacturing, marketing and selling our products and could have a material adverse effect on our business, financial condition and results of operations.
OPTIMIZATION AND INTEGRATION ACTIVITIES
As a result of our ongoing acquisition strategy and significant growth in recent years, we have undertaken cost-saving initiatives in 2024 and 2025 to consolidate manufacturing operations, distribution facilities and transfer activities, eliminate duplicative positions, realign various sales and marketing activities, and expand and upgrade production capacity for our regenerative technology products. These efforts are expected to continue and while we expect a positive impact from ongoing restructuring, integration, and manufacturing transfer and expansion activities, such results remain uncertain.
As a result of audits and inspections by regulatory agencies as well as our own review of the Company’s quality management system, we have implemented an enterprise-wide Compliance Master Plan (the “CMP”), a systematic and holistic approach to improving our quality management system across our manufacturing and supply network. The primary objectives of the CMP are to remediate quality system gaps, harmonize the quality management system and enhance the quality culture across the Company. The Company has completed baseline audits across all manufacturing facilities, conducted CMP training, and has made significant progress in its prioritized work streams.
During the fourth quarter of 2025, we approved a restructuring initiative to improve the Company’s operational performance by strengthening the stability and resilience of our supply chain and advancing our prioritization and execution discipline. We expect to incur aggregate restructuring costs associated with this initiative of approximately $12.6 million related to severance and other employee costs. The costs will be incurred as specific actions required as part of the initiative are identified and approved and are expected to continue through the end of 2026. The amounts and timing of estimated restructuring costs are subject to change until finalized; actual amounts and timing may vary materially based on various factors. We incurred $8.5 million of restructuring related costs related to this initiative during the year ended December 31, 2025. Restructuring costs were included in accrued expenses and other current liabilities in the consolidated balance sheet for the year ended December 31, 2025. See Note 2. Summary of Significant Accounting Policies, of the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for further details.
ACQUISITIONS & DIVESTITURES
Acquisitions
Our growth strategy includes the acquisition of businesses, assets, or product lines to increase the breadth of our offerings and the reach of our product portfolios and drive relevant scale to our customers. As a result, our financial results for the year ended December 31, 2025 may not be directly comparable to those of the corresponding prior-year periods. See Note 4. Acquisitions and Divestitures, of the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for a further discussion.
Durepair® Acquisition
On October 2, 2024, the Company completed the acquisition of the product rights for the Durepair® Regeneration Matrix (“Durepair”), a non-synthetic dura substitute for repair of the dura mater during neurosurgical procedures, from Medtronic plc for total cash consideration of $45.0 million. The Company made a cash payment of $10.0 million upon the closing of the acquisition in October 2024, $15.0 million on the first anniversary of the acquisition in October 2025, and will make an additional cash payment of $20.0 million upon the second anniversary of the acquisition in October 2026. The additional cash payment to be made in October 2026 is included at its present value in accrued expenses and other current liabilities as of December 31, 2025.
The acquisition of the product rights for Durepair, which consist of certain patents and trademarks, regulatory approvals, and other records, has been accounted for as an asset acquisition in accordance with FASB Topic 805, Business Combinations (“ASC 805”) as the acquisition does not include an assembled workforce and substantially all of the fair value of the assets acquired is concentrated in a single identifiable intangible asset.
43
Acclarent, Inc. Acquisition
On April 1, 2024, the Company completed the acquisition of all of the outstanding capital stock of Acclarent, Inc. (“Acclarent”), a developer and marketer of medical devices used in ear, nose, throat (“ENT”) procedures, from Ethicon, Inc., a subsidiary of Johnson & Johnson, for approximately $282.0 million in cash, subject to customary adjustments set forth in the purchase agreement related to working capital balances transferred to the Company. In the second half of 2024, the Company finalized and settled the working capital adjustment in the amount of $4.2 million, which resulted in a reduction to goodwill and also recognized a measurement period adjustment to recognize deferred tax liabilities of $1.1 million with a corresponding increase to goodwill as a result of a change to the estimated deferred tax rate applied and the book-to-tax difference associated with fixed assets acquired.
The addition of Acclarent’s ENT product portfolio, including sinus balloon dilation, eustachian tube balloon dilation, and surgical navigation systems technologies, and dedicated salesforce enhanced the Company’s position in the ENT specialty device market. Acclarent’s results of operations have been reported in the Company’s Codman Specialty Surgical reportable segment from the date of acquisition.
RESULTS OF OPERATIONS
Executive Summary
Net loss for the year ended December 31, 2025 was $(516.5) million, or $(6.74) per diluted share, compared to a net loss of $(6.9) million, or $(0.09) per diluted share for the year ended December 31, 2024. The net loss increased for the year ended December 31, 2025 as compared to prior periods, primarily driven by the impact of the goodwill impairment of $511.4 million recorded in the second quarter of the current year.
Income before income taxes includes the following special charges:
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
Acquisition, divestiture and integration-related charges (1) | $ | 3,597 | $ | 33,626 | |||||||
| Structural optimization charges | 47,994 | 24,194 | |||||||||
Boston recall / Braintree transition(2) | 56,202 | 45,034 | |||||||||
| EU medical device regulation | 41,928 | 44,570 | |||||||||
| Total | 149,721 | 147,424 | |||||||||
(1) This includes adjustments for contingent consideration liabilities. Refer to Note 15. Commitments and Contingencies of the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K).
(2) This primarily includes idle capacity charges, site transfer costs, quality remediation costs, right of use and fixed asset impairments.
The items reported above are reflected in the consolidated statements of operations as follows:
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Cost of goods sold | $ | 87,798 | $ | 72,461 | |||||||
| Research and development | 18,206 | 20,737 | |||||||||
| Selling, general and administrative | 42,449 | 53,922 | |||||||||
| Other expense | 1,268 | 304 | |||||||||
| Total | 149,721 | 147,424 | |||||||||
We typically define special charges as items for which the amounts and/or timing of such expenses may vary significantly from period to period, depending upon our acquisition, divestiture, integration and restructuring activities; items for which the amounts are non-cash in nature; and items which are not expected to recur at the same magnitude. We believe that given our ongoing strategy of seeking acquisitions, our continuing focus on rationalizing our existing manufacturing and distribution infrastructure and our continuing review of various product lines in relation to our current business strategy, some of the special charges discussed above could recur with similar materiality in the future.
44
We believe that the separate identification of these special charges provides important supplemental information to investors regarding financial and business trends relating to our financial condition and results of operations. Investors may find this information useful in assessing the comparability of our operating performance from period to period, against the business model objectives that management has established, and against other companies in our industry. We provide this information to investors so that they can analyze our operating results in the same way that management does and to use this information in their assessment of our core business and valuation of the Company.
Revenues and Gross Margin
The Company’s revenues and gross margin on product revenues were as follows:
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Segment Net Revenues | |||||||||||
| Codman Specialty Surgical | $ | 1,200,511 | $ | 1,143,636 | |||||||
| Tissue Technologies | 434,734 | 466,891 | |||||||||
| Total revenues | 1,635,245 | 1,610,527 | |||||||||
| Cost of goods sold | 803,625 | 728,466 | |||||||||
| Gross margin on total revenues | $ | 831,620 | $ | 882,061 | |||||||
| Gross margin as a percentage of total revenues | 50.9 | % | 54.8 | % | |||||||
Revenues and Gross Margin
For the year ended December 31, 2025, total revenues increased by $24.7 million, or 1.5%, to $1,635.2 million from $1,610.5 million during the prior year. This represents low single digit growth compared to the same period in the prior year, primarily driven by sales related to Acclarent, as well as impacts from quality and operational issues across both periods.
In the CSS segment, revenues were $1,200.5 million, an increase of $56.9 million, or 5.0% from the prior year period. Excluding the impact of foreign currency of $6.5 million, the increase is primarily due to the timing of the Acclarent acquisition completed in the second quarter in the prior year and CSF Management shipping holds experienced in the prior year.
In the TT segment, revenues were $434.7 million, a decrease of $32.2 million, or 6.9% from the prior year period, primarily attributable to the impact of quality and operational issues associated with MediHoney and decreases in private label revenues. This is partially offset by growth in Integra Skin, which recovered from prior year supply issues, and DuraSorb.
Gross margin was $831.6 million for the year ended December 31, 2025, a decrease of $50.4 million from $882.1 million for the prior year period. Gross margin as a percentage of revenue decreased to 50.9% in 2025 from 54.8% in 2024. For the year ended December 31, 2025, gross margins were impacted by quality and operational issues, which affected both revenue and expenses, as well as higher manufacturing costs and cost of tariffs. For the year ended December 31, 2024, gross margins were impacted by intangible impairment charges associated with the Boston recall, Acclarent inventory step up, and expenses associated with quality and operational issues.
Operating Expenses
The following is a summary of operating expenses as a percent of total revenues:
| Years Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Research and development | 6.1 | % | 7.2 | % | |||||||
| Selling, general and administrative | 42.8 | % | 44.5 | % | |||||||
| Goodwill impairment | 31.3 | % | — | % | |||||||
| Intangible asset amortization | 0.9 | % | 1.3 | % | |||||||
| Total operating expenses | 81.0 | % | 53.0 | % | |||||||
Total operating expenses, which consist of research and development, selling, general and administrative, and intangible asset amortization expenses, increased by $471.3 million or 55.2% to $1,325.0 million in 2025, compared to $853.7 million in the prior year, primarily driven by the goodwill impairment recorded in the second quarter of 2025.
45
Research and Development
Research and development expenses for the year ended December 31, 2025 decreased by $16.4 million as compared to the prior year, primarily attributable to cost management initiatives and reduced spend on EU MDR.
Selling, General and Administrative
Selling, general and administrative expenses for the year ended December 31, 2025 decreased by $17.3 million primarily due to adjustments to contingent consideration liabilities and cost management initiatives in the current year, as well as Acclarent acquisition costs incurred in the prior year.
Intangible Asset Amortization
Amortization expense (excluding amounts reported in cost of product revenues for technology-based intangible assets) in 2025 was $15.0 million compared to $21.3 million in 2024. The decrease is driven by the impairment of customer relationship intangible related to our Boston facility of $7.1 million, which was recorded in the first quarter of 2024.
We expect total annual amortization expense (which excludes amounts reported in cost of product revenues for technology-based intangible assets) to be approximately $14.9 million in 2026, $14.2 million in 2027, $10.7 million in 2028, $15.8 million in 2029, $15.1 million in 2030 and $43.7 million thereafter.
Non-Operating Income and Expenses
The following is a summary of non-operating income and expenses:
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Interest income | $ | 18,474 | $ | 20,040 | |||||||
| Interest expense | (86,255) | (70,632) | |||||||||
| Other (expense) income, net | (2,351) | 3,944 | |||||||||
| Total non-operating income and expense | $ | (70,132) | $ | (46,648) | |||||||
Interest Income
Interest income for the year ended December 31, 2025 decreased by $1.6 million as compared to the same period in the prior year primarily due to lower interest earned on cash balances.
Interest Expense
Interest expense for the year ended December 31, 2025 increased by $15.6 million as compared to the same period in the prior year primarily due to higher interest rates on the borrowings under the revolving credit facility component of the Senior Credit Facility as compared to the interest rates on the 2025 Notes.
Other (Expense) Income, Net
Other (expense) income, net for the year ended December 31, 2025 decreased by $6.3 million as compared to the same period in the prior year, primarily driven by foreign exchange impact related to EUR and CHF denominated balances.
Income Taxes
Our effective income tax rate was 8.3% and 61.9% of income before income taxes in 2025 and 2024, respectively. See Note 12. Income Taxes, in our consolidated financial statements for a reconciliation of the United States federal statutory rate to our effective tax rate. Our effective tax rate could vary from year to year depending on, among other factors, tax law changes, the geographic and business mix and taxable earnings and losses. We consider these factors and others, including our history of generating taxable earnings, in assessing our ability to realize deferred tax assets. We estimate our worldwide effective income tax rate for 2026 to be approximately 18%, estimated based on existing tax laws.
At December 31, 2025, the Company had $22.6 million of valuation allowance against the remaining $278.1 million of gross deferred tax assets recorded at December 31, 2025. Our deferred tax asset valuation allowance increased by $7.1 million in 2025, primarily driven by $3.7 million related to Swiss federal and local tax credits and $3.0 million related to U.S. state credits. The valuation allowance relates to deferred tax assets for which the Company does not believe it has satisfied the more likely than not threshold for realization.
46
At December 31, 2025, we had net operating loss carryforwards of $46.7 million for federal income tax purposes, $142.8 million for foreign income tax purposes and $85.4 million for state income tax purposes to offset future taxable income. The federal net operating loss carryforwards decreased in 2025 due to usage during the year. Of the total federal net operating loss carryforwards, $46.7 million expire through 2037. Regarding the foreign net operating loss carryforwards, $118.9 million expire through 2028 and $23.9 million have an indefinite carryforward period. The state net operating loss carryforwards expire through 2045.
As of December 31, 2025, the Company has not provided deferred income taxes on unrepatriated earnings from foreign subsidiaries as they are deemed to be indefinitely reinvested unless there is a manner under which to remit the earnings with no material tax cost.
GEOGRAPHIC PRODUCT REVENUES AND OPERATIONS
The Company attributes revenues to geographic areas based on the location of the customer. Total revenue by major geographic area consisted of the following:
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| United States | $ | 1,204,745 | $ | 1,192,675 | |||||||
| Europe | 162,666 | 158,496 | |||||||||
| Asia Pacific | 189,251 | 176,614 | |||||||||
| Rest of World | 78,583 | 82,742 | |||||||||
| Total Revenues | $ | 1,635,245 | $ | 1,610,527 | |||||||
We generate significant revenues outside the U.S., a portion of which are U.S. dollar-denominated transactions conducted with customers that generate revenue in currencies other than the U.S. dollar. As a result, currency fluctuations between the U.S. dollar and the currencies in which those customers do business could have an impact on the demand for our products in foreign countries. Local economic conditions, regulatory compliance or political considerations, the effectiveness of our sales representatives and distributors, local competition and changes in local medical practice all may combine to affect our sales into markets outside the U.S.
LIQUIDITY AND CAPITAL RESOURCES
Working Capital
At December 31, 2025 and December 31, 2024, working capital was $703.6 million and $159.6 million, respectively. Working capital consists of total current assets less total current liabilities as presented in the consolidated balance sheets. The increase in working capital as compared to the prior year is primarily driven by the repayment of 2025 Notes using our Senior Credit Facility, which decreased current liabilities.
Cash and Marketable Securities
The Company had cash and cash equivalents totaling approximately $235.0 million and $246.4 million at December 31, 2025 and 2024, respectively, which are valued based on Level 1 measurements in the fair value hierarchy. At December 31, 2025, our non-U.S. subsidiaries held approximately $193.0 million of cash and cash equivalents that are available for use outside the U.S. The Company asserts that it has the ability and intends to indefinitely reinvest the undistributed earnings from its foreign operations unless there is no material tax cost to remit the earnings into the U.S.
Short Term Investments
The Company had short term investments, primarily consisting of time deposits, which are valued based on Level 1 measurements in the fair value hierarchy, totaling approximately $28.7 million at December 31, 2025 and $27.2 million at December 31, 2024.
47
Cash Flows
| Years Ended December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Net cash provided by operating activities | $ | 50,384 | $ | 129,382 | |||||||
| Net cash used in investing activities | (108,063) | (390,808) | |||||||||
| Net cash provided by financing activities | 28,335 | 237,863 | |||||||||
| Effect of exchange rate fluctuations on cash | 18,017 | (6,464) | |||||||||
| Net decrease in cash and cash equivalents | $ | (11,327) | $ | (30,027) | |||||||
Cash Flows Provided by Operating Activities
Operating cash flows for the year ended December 31, 2025 decreased by $79.0 million compared to the same period in 2024. Within operating cash flows, net income less non-cash adjustments decreased for the year ended December 31, 2025 by approximately $76.3 million as compared to 2024, due to quality and operational issues, which affected both revenue and expenses, as well as higher manufacturing costs and cost of tariffs.
The changes in assets and liabilities, net of business acquisitions, decreased cash flows by $33.7 million in 2025, mainly attributable to increases in inventory.
The changes in assets and liabilities, net of business acquisitions, decreased cash flows by $31.0 million in 2024, mainly attributable to increases in inventory and prepaid and other current assets, offset by decreases in accounts receivable.
Cash Flows Used in Investing Activities
Uses of cash from investing activities for the year ended December 31, 2025 were $81.4 million paid for capital expenditures to support improvement initiatives at a number of our manufacturing facilities and other technology investments, $14.2 million related to the purchase of intangible assets for Durepair, $8.5 million related to the purchase of short-term investments, and $10.9 million related to settlement of our cross-currency swap designated as net investment hedge.
Sources of cash from investing activities for the year ended December 31, 2025 were $7.0 million for short term investments converted to cash.
Uses of cash from investing activities for the year ended December 31, 2024 were $277.8 million related to the Acclarent acquisition, $104.4 million paid for capital expenditures to support the investment in the new Braintree facility, as well as improvement initiatives at a number of our manufacturing facilities, and other technology investments, $49.0 million related to the purchase of short-term investments, $10.0 million related to the purchase of intangible assets for Durepair, and $4.1 million related to settlement of our cross-currency swap designated as net investment hedge.
Sources of cash from investing activities for the year ended December 31, 2024 were $54.5 million for short term investments converted to cash.
Cash Flows (Used in) Provided by Financing Activities
Uses of cash from financing activities for the year ended December 31, 2025 related to the repayments of the 2025 Notes of $575.0 million, as well as $94.9 million of repayments under our Senior Credit Facility and Securitization Facility. In addition, the company paid $16.5 million related to payments of Arkis and SIA contingent consideration, $4.1 million in debt issuance costs and $2.7 million in cash taxes for net equity settlements.
Sources of cash from financing activities for the year ended December 31, 2025 were $720.7 million proceeds from borrowings of long term indebtedness and $1.0 million related to the proceeds from employee stock purchases.
Uses of cash from financing activities for the year ended December 31, 2024 related to the repayments of $187.1 million under our Senior Credit Facility and Securitization Facility, $52.5 million related to the repurchase of treasury stock under the share repurchase agreements, $11.9 million related to payment of contingent consideration for the SIA acquisition, and $3.5 million in cash taxes paid for net equity settlements.
Sources of cash from financing activities for the year ended December 31, 2024 were $486.5 million proceeds from borrowings under our Senior Credit Facility and Securitization Facility and $6.4 million proceeds from the exercise of stock options.
48
Tariffs and Macroeconomic Environment
In April 2025, the U.S. government announced new tariffs on goods imported into the U.S. from dozens of countries, including China and the European Union member states. In response, governments have threatened or imposed reciprocal tariffs or taken other measures, and the United States is in the process of negotiating trade agreements with certain governments. In August 2025, the U.S. Court of Appeals for the Federal Circuit ruled against certain of the U.S. tariffs that have been implemented. The U.S. administration has appealed this ruling. In November 2025, the U.S. Supreme Court heard oral arguments on tariffs imposed under the International Emergency Economic Powers Act (“IEEPA”). In February 2026, the U.S. Supreme Court issued a 6–3 ruling invalidating the U.S. Administration’s tariff program implemented under the IEEPA, concluding that the IEEPA did not authorize the broad import duties previously imposed. The Court did not address the treatment of previously collected tariff payments, leaving refund processes subject to future administrative and judicial determinations. Following the ruling, the Administration announced a new global 10% tariff under Section 122 of the Trade Act of 1974, which permits temporary import surcharges of up to 15% for up to 150 days to address balance of payments deficits, with implementation effective almost immediately and subject to certain exemptions. We are evaluating the combined impact of these developments on our supply chain, cost structure, and financial outlook; however, the ultimate effects remain uncertain pending additional guidance from federal agencies. For context, the Company incurred $19.9 million in tariff costs in 2025, of which an estimated $16.0 million related to tariffs imposed under IEEPA authority. Additionally, in September 2025, the U.S. Department of Commerce initiated national security investigations into medical equipment, devices, and robotics. The tariff environment has continued to shift throughout the 2025 calendar year, with new measures being proposed, paused, implemented, and countered, contributing to broader trade policy uncertainty.
Tariffs have resulted in an increase in certain product costs and could have adverse impacts on, among other things, demand for our products and supply chains. Particularly, the U.S. import tariffs and reciprocal measures by China, are expected to increase the Company’s cost of goods sold. The Company anticipates that some of its suppliers will incur incremental tariff-related costs, which may be passed on to the Company. Approximately half of our global revenue is generated from products manufactured in the U.S. In China, which accounts for approximately 5 percent of our total revenue, roughly half of the products we sell are manufactured in the United States.
Any tariffs paid have been capitalized in inventory and are recognized in our cost of goods sold as those products are sold. During the year ended December 31, 2025, the Company paid approximately $19.9 million of tariffs on imported goods. Of this amount, $6.5 million was recognized in cost of goods sold in the consolidated statements of operations.
The overall macroeconomic and geopolitical environment, including tariffs or changes in trade policies, slower economic growth or recession, market volatility and inflation, and uncertainty regarding all of the foregoing, pose risks that could impact our business, results of operations, financial condition and cash flows. The extent and duration of the tariffs and the resulting impact on general economic conditions and on the business are uncertain and are expected to be impacted by various factors, such as negotiations between the U.S. and affected countries, the responses of other countries or regions, exemptions or exclusions that already exist or may be granted, availability and cost of alternative sources of our products and materials, and our ability to offset the effects of any tariffs that might be imposed. For additional information on the risks that tariffs pose to the Company, please see Part I, Item 1A. “Risk Factors” of this Annual Report on Form 10-K.
Credit Agreement, Convertible Senior Notes, Securitization and Related Hedging Activities
See Note 5. Debt, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for further details of our Amended and Restated Senior Credit Agreement, the 2025 Notes, and Securitization Facility and Note 6. Derivative Instruments to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for further details of our hedging activities. Our Senior Credit Facility has a maturity date of March 24, 2028 and we expect to seek to refinance all of our outstanding debt in 2026.
The Senior Credit Facility is subject to various financial and negative covenants and, at December 31, 2025, the Company was in compliance with all such covenants. Our Consolidated Total Leverage Ratio was 4.50, with the covenant requirement at 5.00 at the end of December 31, 2025. The covenant requirement will drop from 5.00 to 4.75 for the fiscal quarter ended September 30, 2026. Please refer to Note 5. Debt for the complete covenant table.
The 2025 Notes matured on August 15, 2025 and were settled upon maturity for $575.0 million in cash, excluding accrued interest, funded by borrowings on the revolving credit facility component of the Senior Credit Facility. No shares were issued to settle the 2025 Notes.
49
Share Repurchase Plan
See Note 8. Treasury Stock, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for further details of our share repurchase programs.
Dividend Policy
We have not paid any cash dividends on our common stock since our formation. Our Senior Credit Facility limits the amount of dividends that we may pay. Any future determinations to pay cash dividends on our common stock will be at the discretion of the Board of Directors and will depend upon our financial condition, results of operations, cash flows and other factors deemed relevant by the Board of Directors.
Capital Resources
We believe that our cash and available borrowings under the Senior Credit Facility are sufficient to finance our operations and capital expenditures for the next twelve months and foreseeable future. Our future capital requirements will depend on many factors, including the growth of our business, the timing and introduction of new products and investments, strategic plans and acquisitions, among others. Additional sources of liquidity available to us include short term borrowings and the issuance of long term debt and equity securities.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet financing arrangements during the year ended December 31, 2025 that have or are reasonably likely to have, a current or future effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to our interests.
Contractual Obligations and Commitments
We will continue to have cash requirements to support seasonal working capital needs and capital expenditures, to pay interest, to service debt, and to fund acquisitions. As part of our ongoing operations, we enter into contractual arrangements that obligate us to make future cash payments
Our primary obligations include principal and interest payments on the revolving credit facility and term loan component of the Senior Credit Facility and our Securitization Facility. See Note 5. Debt, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for details. The Company also leases some of our manufacturing facilities and office buildings which have required future minimum lease payments. See Note 11. Lease and Related Party Leases, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for a schedule of our future minimum lease payments. Amounts related to the Company’s other obligations, including employment agreements and purchase obligations were not material.
The Company has contingent consideration obligations related to prior years’ acquisitions and future pension contribution obligations. See Note 10. Retirement Benefit Plans, and Note 15. Commitments and Contingencies to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for details. The associated obligations are not fixed. The Company also has a liability for uncertain tax benefits including interest and penalties. See Note 12. Income Taxes to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for details. The Company cannot make a reliable estimate of the period in which the uncertain tax benefits may be realized.
CRITICAL ACCOUNTING POLICIES AND THE USE OF ESTIMATES
Our discussion and analysis of financial conditions and results of operations is based upon our consolidated financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States of America (“GAAP”). The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent liabilities, and the reported amounts of revenues and expenses. Significant estimates affecting amounts reported or disclosed in the consolidated financial statements include allowances for doubtful accounts receivable and sales returns and allowances; net realizable value of inventories; accounting for business combinations; valuation of goodwill and intangible assets including estimated projected cash flows, discount rates, and estimated useful lives used to value and test goodwill and intangible assets for impairment; income taxes and valuation allowances recorded against deferred tax assets; valuation of stock-based compensation; valuation of retirement benefit plan assets and liabilities; valuation of derivative instruments; and valuation of contingent liabilities. These estimates are based on historical experience and on various other assumptions that are believed to be reasonable under the current circumstances.
We believe that the following accounting policies, which form the basis for developing these estimates, are those that are most critical to the presentation of our consolidated financial statements and require the more difficult subjective and complex judgments, often because of the need to make estimates about the effect of matters that are inherently uncertain. Because of this uncertainty, actual results could differ from these estimates.
50
Inventories
Inventories, consisting of purchased materials, direct labor and manufacturing overhead, are stated at the lower of cost (determined by the first-in, first-out method) or net realizable value. At each balance sheet date, we evaluate ending inventories for excess quantities, obsolescence or shelf-life expiration. Our evaluation includes an analysis of historical sales levels by product, projections of future demand by product, the risk of technological or competitive obsolescence for our products, general market conditions, a review of the shelf-life expiration dates for our products, and the feasibility of reworking or using excess or obsolete products or components in the production or assembly of other products that are not obsolete or for which we do not have excess quantities in inventory. To the extent that we determine there are excess or obsolete quantities or quantities with a shelf life that is too near its expiration for us to reasonably expect that we can sell those products prior to their expiration, we adjust their carrying value to estimated net realizable value. If future demand or market conditions are lower than our projections, or if we are unable to rework excess or obsolete quantities into other products, we may record further adjustments to the carrying value of inventory through a charge to cost of product revenues in the period the revision is made.
The Company capitalizes inventory costs associated with certain products prior to regulatory approval, based on management’s judgment of probable economic benefit. The Company could be required to expense previously capitalized costs related to pre-approval inventory upon a change in such judgment, due to, among other potential factors, a denial or delay of approval by necessary regulatory bodies or a decision by management to discontinue the related development program.
Any tariffs paid have been capitalized in inventory and will be recognized in our cost of goods sold as those products are sold.
Refer to Note 2. Summary of Significant Accounting Policies to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for more information.
Business Combinations
The Company accounts for the acquisition of a business in accordance with ASC 805. Amounts paid to acquire a business are allocated to the assets acquired and liabilities assumed based on their respective estimated fair values as of the date of acquisition in accordance with the fair value hierarchy described in FASB Topic 820, Fair Value Measurement (“ASC 820”). Any excess of the purchase price over the fair value of the net assets acquired is recorded as goodwill. Transaction costs and costs to restructure the acquired company are expensed as incurred. Results of operations of acquired businesses are included in the Company’s results of operations as of the respective acquisition dates.
The Company determines the fair value of acquired intangible assets based on detailed valuations that use information and assumptions provided by management. Determining the fair value of these intangible assets acquired as part of a business combination requires the Company to make significant estimates. These estimates include the estimated annual net cash flows including application of forecasted revenue, the discount rate that appropriately reflects the risk inherent in each future cash flow stream, and an assessment of the asset’s life cycle, as well as other factors such as the consideration of legal, technical, regulatory, economic, and competitive risks. The fair value assigned to acquired intangible assets is determined by estimating the future cash flows of each project or technology and discounting the net cash flows back to their present values. The discount rate used is determined at the time of measurement in accordance with accepted valuation methodologies.
In our acquisition of Acclarent, the key areas of judgment relating to the valuation of the acquired definite-lived developed technology intangible assets were net revenue growth rates; cost of sales; operating expenses including selling and marketing costs, research and development costs, and general and administrative costs; discount rates; obsolescence curve; and intangible assets’ estimated useful lives. These assumptions were developed with the assistance of a third-party valuation expert.
In-process research and development (“IPR&D”) acquired in connection with the acquisition of a business in accordance with ASC 805 is initially recognized at fair value and characterized as an indefinite-lived intangible asset, irrespective of whether the acquired IPR&D has an alternative future use. The Company has not acquired any IPR&D in connection with the acquisition of a business during the years ended December 31, 2025 and 2024.
Research and development costs incurred after the acquisition are expensed as incurred. Upon receipt of regulatory approval, the indefinite-lived intangible asset is then accounted for as a finite-lived intangible asset and amortized on a straight-line basis or accelerated basis, as appropriate, over its estimated useful life. If the project is subsequently abandoned, the indefinite-lived intangible asset is charged to expense.
Due to the uncertainty associated with IPR&D, there is risk that actual results will differ materially from the original cash flow projections and that the research and development project will result in a successful commercial product. The risks associated with achieving commercialization include, but are not limited to, delay or failure to obtain regulatory approvals to conduct clinical trials, delay or failure to obtain required market clearances, delays or issues with patent issuance, or validity and litigation.
If the acquired net assets do not constitute a business under the acquisition method of accounting, the transaction is accounted for as an asset acquisition and no goodwill is recognized. In an asset acquisition, the amount allocated to acquired IPR&D with
51
no alternative future use is charged to expense at the acquisition date. Payments that would be recognized as contingent consideration in a business combination are recognized when probable in an asset acquisition.
Refer to Note 4. Acquisitions and Divestitures and Note 15. Commitments and Contingencies to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for more information.
Goodwill and Identifiable Intangible Assets
In accordance with FASB Topic 350, Intangibles—Goodwill and Other (“ASC 350”), goodwill is not subject to amortization but is tested for impairment at the reporting unit level annually in the third quarter. Additionally, the Company may perform interim tests of goodwill for impairment if an event occurs or circumstances change that could potentially reduce the fair value of a reporting unit below its carrying amount. The carrying value of each reporting unit is determined by assigning the assets and liabilities, including the existing goodwill and intangible assets, to those reporting units. An impairment loss is recognized when the reporting unit’s carrying amount exceeds its estimated fair value.
The Company tests for impairment by either performing a qualitative evaluation or a quantitative test. The qualitative evaluation is an assessment of factors, including reporting unit specific operating results as well as industry, market and general economic conditions, to determine whether it is more likely than not that the fair values of a reporting unit is less than its carrying amount, including goodwill. The Company may elect to bypass this qualitative evaluation for some or all of its reporting units and perform a quantitative test. The quantitative test uses a combination of both an income approach and a market approach to determine the fair value of the reporting unit. The income approach utilizes the estimated discounted cash flows for the reporting unit, while the market approach utilizes comparable publicly-traded companies’ revenue and earnings before interest, taxes, depreciation, and amortization (“EBITDA”) multiples. Estimates and assumptions used in the income approach to calculate projected future discounted cash flows included revenue growth rates, cost of sales, terminal growth rates, and a discount rate for each reporting unit. Discount rates are determined using a weighted average cost of capital for risk factors specific to each reporting unit and other market and industry data. The assumptions used are inherently subject to uncertainty and slight changes in these assumptions could have a significant impact on the concluded value. The estimates and assumptions applied represent a Level 3 measurement in the fair value hierarchy. Level 3 inputs are supported by limited or no market activity and reflect the Company’s assumptions in measuring fair value.
The key assumptions impacting the valuation included the following:
•The reporting unit’s financial projections, including revenue growth rates and cost of sales, which are based on management’s assessment of regional and macroeconomic variables, industry trends and market opportunities, and the Company’s strategic objectives and future growth plans.
•The projected terminal value for the reporting unit, which represents the present value of projected cash flows beyond the last period in the discounted cash flow analysis. The terminal value reflects the Company’s assumptions related to long-term growth rates and profitability, which are based on several factors, including local and macroeconomic variables, market opportunities, and future growth plans.
•The discount rate used to measure the present value of the projected future cash flows is set using a weighted-average cost of capital method that considers market and industry data as well as the Company’s specific risk factors that are likely to be considered by a market participant. The weighted-average cost of capital is the Company’s estimate of the overall after-tax rate of return required by equity and debt holders of a business enterprise.
During the second quarter of 2025, the Company performed a quantitative assessment of its Tissue Technologies, Neurosurgery, and Instruments and ENT reporting units in accordance with ASC 350 due to the decrease in the price per share of the Company’s common stock related to a number of factors including recent tariff changes that created broad economic uncertainty and the impact of quality, operational, and supply issues. The Company recognized an aggregate charge of $511.4 million in goodwill impairment expense in the consolidated statement of operations in the second quarter of 2025.
In the third quarter of 2025, the Company performed its annual test of its reporting units for impairment and completed a qualitative evaluation of its Tissue Technologies and Neurosurgery reporting units. The Instruments and ENT reporting unit had been deemed fully-impaired during the second quarter of 2025 and was excluded from this evaluation. After performing the qualitative analysis, the Company concluded that it was more likely than not that the fair values of the Tissue Technologies and Neurosurgery reporting units were greater than their carrying amounts. Therefore, it was not necessary to perform a quantitative impairment test.
If the price per share of the Company’s common stock, macro-economic market conditions, or related forecast revisions deteriorate, these events or changes in circumstances may indicate a decline in the fair values of the Tissue Technologies and Neurosurgery reporting units below their respective carrying amounts and require the Company to perform an interim test of goodwill for impairment which may potentially result in additional goodwill impairment expense in the future.
The Company had identifiable intangible assets, net of accumulated amortization, of $1.1 billion on its consolidated balance
52
sheet as of December 31, 2025.
Refer to Note 7. Goodwill and Other Intangibles, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for more information.
Income Taxes
Since we conduct operations on a global basis, our effective tax rate has and will depend upon the geographic distribution of our pre-tax earnings among locations with varying tax rates. Changes in the tax rates of the various jurisdictions in which we operate affect our profits. In addition, we maintain a reserve for uncertain tax benefits, changes to which could impact our effective tax rate in the period such changes are made. The effective tax rate can also be impacted by changes in valuation allowances of deferred tax assets, and tax law changes.
Our provision for income taxes may change period-to-period based on specific events, such as the settlement of income tax audits and changes in tax laws, as well as general factors, including the geographic mix of income before taxes, state and local taxes and the effects of the Company’s global income tax strategies. We maintain strategic management and operational activities in overseas subsidiaries. See Note 12. Income Taxes, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K), in our consolidated financial statements for disclosures related to foreign and domestic pretax income, foreign and domestic income tax expense (benefit) and the effect foreign taxes have on our overall effective tax rate.
We recognize a tax benefit from an uncertain tax position only if it is more likely than not to be sustained upon examination based on the technical merits of the position. The amount of the accrual for which an exposure exists is measured by determining the amount that has a greater than 50 percent likelihood of being realized upon ultimate settlement of the position. Components of the reserve are classified as a long-term liability in the consolidated balance sheets. We record interest and penalties accrued in relation to uncertain tax benefits as a component of income tax expense.
We believe that we have identified all reasonably identifiable exposures and that the reserve we have established for identifiable exposures is appropriate under the circumstances; however, it is possible that additional exposures exist and that exposures will be settled at amounts different from the amounts reserved. It is also possible that changes in facts and circumstances could cause us to either materially increase or reduce the carrying amount of our tax reserves.
Our deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and their basis for income tax purposes, and the temporary differences created by the tax effects of capital loss, net operating loss and tax credit carryforwards. We record valuation allowances when it is more likely than not that some portion or all of the deferred tax assets will not be realized. We could recognize no benefit from our deferred tax assets or we could recognize some or all of the future benefit depending on the amount and timing of taxable income we generate in the future.
As of December 31, 2025, the Company has not provided deferred income taxes on unrepatriated earnings from foreign subsidiaries as they are deemed to be indefinitely reinvested unless there is a manner under which to remit the earnings with no material tax cost. The current analysis indicates that we have sufficient U.S. liquidity, including borrowing capacity, to fund foreseeable U.S. cash needs without requiring the repatriation of foreign cash. One time or unusual items that may impact our ability or intent to keep the foreign earnings and cash indefinitely reinvested include significant U.S. acquisitions, loans from a foreign subsidiary, and changes in tax laws.
Refer to Note 12. Income Taxes, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for more information.
Recently Issued and Adopted Accounting Standards
Refer to Note 2. Summary of Significant Accounting Policies, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K), to the consolidated financial statements for recently adopted accounting pronouncements.
53
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are exposed to various market risks, including changes in foreign currency exchange rates and interest rates that could adversely affect our results of operations and financial condition. To manage the volatility relating to these typical business exposures, we may enter into various derivative transactions when appropriate. We do not hold or issue derivative instruments for trading or other speculative purposes.
Foreign Currency Exchange and Other Rate Risks
We operate on a global basis and are exposed to the risk that changes in foreign currency exchange rates could adversely affect our financial condition, results of operations and cash flows. We are primarily exposed to foreign currency exchange rate risk with respect to transactions and net assets denominated in Euros, British pounds, Swiss francs, Canadian dollars, Japanese yen, Israeli shekel, Australian dollars and Chinese yuan. We manage the foreign currency exposure centrally, on a combined basis, which allows us to net exposures and to take advantage of any natural offsets. To mitigate the impact of currency fluctuations on transactions denominated in nonfunctional currencies, we periodically enter into derivative financial instruments in the form of foreign currency exchange forward contracts with major financial institutions. We temporarily record realized and unrealized gains and losses on these contracts that qualify as cash flow hedges in other comprehensive income, and then recognize them in other income or expense when the hedged item affects net earnings.
From time to time, we enter into foreign currency forward exchange contracts to manage currency exposures for transactions denominated in a currency other than an entity’s functional currency. As a result, the impact of foreign currency gains/losses recognized in earnings are partially offset by gains/losses on the related foreign currency forward exchange contracts in the same reporting period. Refer to Note 6. Derivative Instruments, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for additional information.
We maintain written policies and procedures governing our risk management activities. With respect to derivatives, changes in hedged items are generally expected to be completely offset by changes in the fair value of hedge instruments. Consequently, foreign currency exchange contracts would not subject us to material risk due to exchange rate movements, because gains and losses on these contracts offset gains and losses on the assets, liabilities or transactions being hedged.
The results of operations discussed herein have not been materially affected by inflation.
Interest Rate Risk
Cash and Cash Equivalents - We are exposed to the risk of interest rate fluctuations on the interest income earned on our cash and cash equivalents. A hypothetical 100 basis points increase or decrease in interest rates applicable to our cash and cash equivalents outstanding at December 31, 2025 would impact the Company by approximately $2.3 million on an annual basis. We are subject to foreign currency exchange risk with respect to cash balances maintained in foreign currencies.
Short-Term Investments - We are exposed to the risk of interest rate fluctuations on the interest income earned on our short-term investments. A hypothetical 100 basis points movement in interest rates applicable to our short-term investments outstanding at December 31, 2025 would increase or decrease interest income by approximately $0.3 million on an annual basis.
Debt - Our interest rate risk relates primarily to U.S. dollar SOFR-indexed borrowings. We use interest rate swap derivative instruments to manage our earnings and cash flow exposure to changes in interest rates. These interest rate swaps fix the interest rate on a portion of our expected SOFR-indexed floating-rate borrowings. These interest rate swaps were designated as cash flow hedges as of December 31, 2025. The total notional amounts related to the Company’s interest rate swaps were $900.0 million, of which all are effective as of as of December 31, 2025. Based on our outstanding borrowings at December 31, 2025, a 100 basis points change in interest rates would have impacted interest expense on the unhedged portion of the debt by $9.6 million on an annualized basis. See Note 6. Derivative Instruments, to the Notes to Consolidated Financial Statements (Part IV, Item 15 of this Annual Report on Form 10-K) for further information regarding interest rate swaps.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTAL DATA
Financial statements specified by this Item, together with the report thereon of PricewaterhouseCoopers LLP, are presented following Item 15. Exhibits and Financial Statements of this Annual Report on Form 10-K.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURES
Not applicable.
54
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to provide reasonable assurance that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate, to allow for timely decisions regarding required disclosure. Disclosure controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and management is required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Management has designed our disclosure controls and procedures to provide reasonable assurance of achieving the desired control objectives.
As required by Exchange Act Rule 13a-15(b), we have carried out an evaluation, under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, of the effectiveness of the design and operation of our disclosure controls and procedures as of December 31, 2025. Based upon this evaluation, our principal executive officer and principal financial officer concluded that our disclosure controls and procedures were effective as of December 31, 2025 to provide such reasonable assurance.
Management’s Report on Internal Control Over Financial Reporting
Management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) under the Securities Exchange Act of 1934, as amended. Internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles in the United States of America (“GAAP”). We recognize that because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies and procedures may deteriorate.
To evaluate the effectiveness of our internal control over financial reporting, management used the criteria described in Internal Control — Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”). Based upon this evaluation, management concluded that our internal control over financial reporting was effective as of December 31, 2025.
The effectiveness of the Company’s internal control over financial reporting as of December 31, 2025 has been audited by PricewaterhouseCoopers LLP, an independent registered public accounting firm, as stated in their report which appears herein.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting (as defined in Rule 13a-15(f) under the Exchange Act) that occurred during the quarter ended December 31, 2025 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
Adoption of Executive Severance Plan
We are providing the following disclosure in lieu of filing a Current Report on Form 8-K relating to Item 5.02 (Departure of Directors or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers).
On February 24, 2026, the Compensation Committee of the Board of Directors of Integra LifeSciences Holdings Corporation (the “Company”) adopted the Integra LifeSciences Executive Separation Pay Plan (the “Executive Severance Plan”). The Executive Severance Plan provides severance benefits to the Company’s Executive Leadership Team, which includes eligible executives of the Company (each such individual, a “Participant”) upon a Qualifying Termination (as defined in the Executive Severance Plan). The current Participants include Lea Knight, Executive Vice President and Chief Financial Officer, Robert T. Davis, Jr., Executive Vice President, President – Tissue Technologies, Michael McBreen, Executive Vice President, President – Codman Specialty Surgical, and Harvinder Singh, Executive Vice President, President – International. Capitalized terms used but not otherwise defined herein have the meanings assigned to them in the Executive Severance Plan.
Severance benefits under the Executive Severance Plan include:
•A cash severance payment equal to twelve (12) Months of Base Pay (the “Severance Payment”) payable over 12 months (the “Separation Period”).
55
•A pro-rated bonus payment for the calendar year in which the relevant Qualifying Termination occurred in an amount equal to the actual bonus payment which would have been paid to the Participant had such Participant remained in continuous employment through the end of the calendar year of the bonus period and pro-rated based on the number of calendar days the Participant was employed by the Company during the calendar year of the Qualifying Termination, subject to (i) the Participant having remained continuously employed by the Company for a minimum of nine (9) months during the calendar year of the Participant’s Qualifying Termination and (ii) the funding of the Company, individual performance of the Employee and any additional eligibility criteria for such bonus payment.
•For Participants enrolled in Company-sponsored medical, dental and/or vision coverage, payment in an amount equal to the Company’s contribution towards such health coverage, as in effect for similarly situated active employees (as applicable, based on the Participant’s enrollment status and covered dependents, if any) covering a period through the earlier of (x) the end of the Separation Period, or (y) the date the Participant becomes eligible for group medical care coverage through other employment benefits for them and their eligible dependents through the continuation of coverage provisions.
•Eligibility for outplacement services during the Separation Period.
The Severance Payment will be paid in substantially equal installments during the Separation Period. The Executive Severance Plan provides that none of a termination of employment for unsatisfactory job performance, voluntary resignation or retirement, termination due to death, or disability, termination in connection with a divestiture where continued employment is offered, or certain other terminations or separations as set forth in the Executive Severance Plan will be considered a Qualifying Termination under the terms of the Executive Severance Plan.
Payment of benefits under the Executive Severance Plan is subject to: (i) the execution and non-revocation of a general release of claims, (ii) compliance with applicable post-employment restrictive covenants, (iii) applicable tax withholdings, and (iv) clawback in the event of a violation of the general release of claims.
This summary of the Executive Severance Plan does not purport to be complete and is subject to and qualified in its entirety by reference to the text of the Executive Severance Plan filed as Exhibit 10.12 hereto and incorporated herein by reference.
Rule 10b5-1 Trading Arrangements
During the three months ended December 31, 2025, no director or officer of the Company, nor the Company itself, adopted terminated
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
PART III
INCORPORATION BY REFERENCE
The information called for by Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities relating to equity compensation plans, Item 10. Directors, Executive Officers and Corporate Governance, Item 11. Executive Compensation, Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters, Item 13. Certain Relationships and Related Transactions, and Director Independence and Item 14. Principal Accountant Fees and Services is incorporated herein by reference to the Company’s definitive proxy statement for its Annual Meeting of Stockholders scheduled to be held on May 7, 2026, which definitive proxy statement is expected to be filed with the Commission not later than 120 days after the end of the fiscal year to which this report relates.
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENTS
(a) Documents filed as a part of this report:
1. Financial Statements.
56
The following financial statements are filed as a part of this report:
All other schedules not listed above have been omitted, because they are not applicable or are not required, or because the required information is included in the consolidated financial statements or notes thereto.
2. Exhibits required to be filed by Item 601 of Regulation S-K.
| 2.1(a) | ||||||||
| 2.1(b) | ||||||||
| 3.1(a) | ||||||||
| 3.1(b) | ||||||||
| 3.1(c) | ||||||||
| 3.1(d) | ||||||||
| 3.1(e) | ||||||||
| 3.2 | ||||||||
| 4.1 | ||||||||
| 4.2 | ||||||||
| 4.3* | ||||||||
| 4.4+ | ||||||||
57
| 10.1(a) | ||||||||
| 10.1(b) | ||||||||
| 10.2(a)* | ||||||||
| 10.2(b)* | ||||||||
| 10.3(a)* | ||||||||
| 10.3(b)* | ||||||||
| 10.3(c)* | ||||||||
| 10.3(d)* | ||||||||
| 10.3(e)* | ||||||||
| 10.3(f)*+ | ||||||||
| 10.3(g)*+ | ||||||||
| 10.3(h)*+ | ||||||||
| 10.3(i)* | ||||||||
| 10.3(j)* | ||||||||
| 10.3(k)* | ||||||||
| 10.3(l)* | ||||||||
| 10.3(m)* | ||||||||
| 10.3(n)* | ||||||||
58
| 10.3(o)* | ||||||||
| 10.3(p)*+ | ||||||||
| 10.4* | ||||||||
| 10.5* | ||||||||
| 10.6* | ||||||||
| 10.7* | ||||||||
| 10.8* | ||||||||
| 10.9* | ||||||||
| 10.10* | ||||||||
| 10.11(a)* | ||||||||
| 10.11(b)* | ||||||||
| 10.11(c)* | ||||||||
| 10.12*+ | ||||||||
| 10.13*+ | ||||||||
| 10.14(a) | ||||||||
| 10.14(b) | ||||||||
| 10.14(c) | ||||||||
59
| 10.14(d) | ||||||||
| 10.14(e) | ||||||||
| 10.14(f) | ||||||||
| 10.15 | ||||||||
| 10.16 | ||||||||
| 10.17 | ||||||||
| 10.18 | ||||||||
| 10.19 | ||||||||
| 10.20 | ||||||||
| 10.21 | ||||||||
| 10.22 | ||||||||
| 10.23 | ||||||||
| 10.24 | ||||||||
60
| 10.25 | ||||||||
| 10.26 | ||||||||
| 10.27 | ||||||||
| 10.28 | ||||||||
| 10.29 | ||||||||
| 10.30 | ||||||||
| 10.31 | ||||||||
| 10.32 | ||||||||
| 10.33 | ||||||||
| 10.34 | ||||||||
| 19.1(a) | ||||||||
| 19.1(b) | ||||||||
| 21.1+ | ||||||||
| 23.1+ | ||||||||
| 31.1+ | ||||||||
| 31.2+ | ||||||||
| 32.1+ | ||||||||
| 32.2+ | ||||||||
61
| 97.1 | ||||||||
| 101.INS+# | Inline XBRL Instance Document | |||||||
| 101.SCH+# | Inline XBRL Taxonomy Extension Schema Document | |||||||
| 101.CAL+# | Inline XBRL Taxonomy Extension Calculation Linkbase Document | |||||||
| 101.DEF+# | Inline XBRL Definition Linkbase Document | |||||||
| 101.LAB+# | Inline XBRL Taxonomy Extension Labels Linkbase Document | |||||||
| 101.PRE+# | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |||||||
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | |||||||
| * | Indicates a management contract or compensatory plan or arrangement. | ||||
| + | Indicates this document is filed as an exhibit herewith. | ||||
| # | The financial information of Integra LifeSciences Holdings Corporation Annual Report on Form 10-K for the year ended December 31, 2025 filed on February 26, 2026 formatted in XBRL (Extensible Business Reporting Language): (i) the Consolidated Statements of Operations, (ii) the Consolidated Statement of Comprehensive Income (Loss), (iii) the Consolidated Balance Sheets, (iv) Parenthetical Data to the Consolidated Balance Sheets, (v) the Consolidated Statements of Cash Flows, (vi) the Consolidated Statements of Changes in Stockholders’ Equity, and (vii) Notes to Consolidated Financial Statements, is furnished electronically herewith. | ||||
The Company’s Commission File Number for Reports on Form 10-K, Form 10-Q and Form 8-K is 000-26224.
ITEM 16. FORM 10-K SUMMARY
None.
62
SIGNATURES
Pursuant to the requirements of Section 13 of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| INTEGRA LIFESCIENCES HOLDINGS CORPORATION | |||||
| By: | /s/ Mojdeh Poul | ||||
| Mojdeh Poul | |||||
| President and Chief Executive Officer, and Director | |||||
| (Principal Executive Officer) | |||||
| By: | /s/ Lea Knight | ||||
| Lea Knight | |||||
| Executive Vice President and Chief Financial Officer | |||||
| (Principal Financial Officer) | |||||
| By: | /s/ Jeffrey A. Mosebrook | ||||
| Jeffrey A. Mosebrook | |||||
| Senior Vice President, Finance | |||||
| (Principal Accounting Officer) | |||||
Date: February 26, 2026
63
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons, on behalf of the registrant in the capacities indicated.
| Signature | Title | Date | ||||||||||||
| /s/ Mojdeh Poul | President and Chief Executive Officer, | February 26, 2026 | ||||||||||||
| Mojdeh Poul | and Director (Principal Executive Officer) | |||||||||||||
| /s/ Lea Knight | Executive Vice President and Chief Financial Officer | February 26, 2026 | ||||||||||||
| Lea Knight | (Principal Financial Officer) | |||||||||||||
| /s/ Jeffrey A. Mosebrook | Senior Vice President, Finance | February 26, 2026 | ||||||||||||
| Jeffrey A. Mosebrook | (Principal Accounting Officer) | |||||||||||||
| /s/ Stuart M. Essig, Ph.D. | Chairman of the Board | February 26, 2026 | ||||||||||||
| Stuart M. Essig, Ph.D. | ||||||||||||||
| /s/ Keith Bradley, Ph.D. | Director | February 26, 2026 | ||||||||||||
| Keith Bradley, Ph.D. | ||||||||||||||
| /s/ Shaundra Clay | Director | February 26, 2026 | ||||||||||||
| Shaundra Clay | ||||||||||||||
| /s/ Jeffrey A. Graves | Director | February 26, 2026 | ||||||||||||
| Jeffrey A. Graves | ||||||||||||||
| /s/ Barbara B. Hill | Director | February 26, 2026 | ||||||||||||
| Barbara B. Hill | ||||||||||||||
| /s/ Renee W. Lo | Director | February 26, 2026 | ||||||||||||
| Renee W. Lo | ||||||||||||||
| /s/ Christian S. Schade | Director | February 26, 2026 | ||||||||||||
| Christian S. Schade | ||||||||||||||
64
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of Integra LifeSciences Holdings Corporation
Opinions on the Financial Statements and Internal Control over Financial Reporting
We have audited the accompanying consolidated balance sheets of Integra LifeSciences Holdings Corporation and its subsidiaries (the "Company") as of December 31, 2025 and 2024, and the related consolidated statements of operations, of comprehensive income, of changes in stockholders’ equity and of cash flows for each of the three years in the period ended December 31, 2025, including the related notes (collectively referred to as the "consolidated financial statements"). We also have audited the Company's internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control - Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO).
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2025 in conformity with accounting principles generally accepted in the United States of America. Also in our opinion, the Company maintained, in all material respects, effective internal control over financial reporting as of December 31, 2025, based on criteria established in Internal Control - Integrated Framework (2013) issued by the COSO.
Basis for Opinions
The Company's management is responsible for these consolidated financial statements, for maintaining effective internal control over financial reporting, and for its assessment of the effectiveness of internal control over financial reporting, included in Management’s Report on Internal Control over Financial Reporting appearing under Item 9A. Our responsibility is to express opinions on the Company’s consolidated financial statements and on the Company's internal control over financial reporting based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud, and whether effective internal control over financial reporting was maintained in all material respects.
Our audits of the consolidated financial statements included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. Our audit of internal control over financial reporting included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audits also included performing such other procedures as we considered necessary in the circumstances. We believe that our audits provide a reasonable basis for our opinions.
Definition and Limitations of Internal Control over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
F-1
Critical Audit Matters
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that was communicated or required to be communicated to the audit committee and that (i) relates to accounts or disclosures that are material to the consolidated financial statements and (ii) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing a separate opinion on the critical audit matter or on the accounts or disclosures to which it relates.
Interim Goodwill Impairment Assessments – Tissue Technologies and Neurosurgery Reporting Units
As described in Notes 2 and 7 to the consolidated financial statements, the Company’s goodwill balance was $615.2 million as of December 31, 2025. Goodwill is not subject to amortization but is tested for impairment at the reporting unit level annually in the third quarter. Additionally, management may perform interim tests of goodwill for impairment if an event occurs or circumstances change that could potentially reduce the fair value below its carrying amount. During the second quarter of 2025, management performed a quantitative assessment of its Tissue Technologies, Neurosurgery, and Instruments and ENT reporting units, using a combination of both an income approach and a market approach, to determine the fair value of the reporting unit. The income approach utilizes the estimated discounted cash flows for the reporting unit, while the market approach utilizes comparable publicly-traded companies’ revenue and EBITDA multiples. Estimates and assumptions made by management used in the income approach included revenue growth rates, cost of sales, terminal growth rates, and a discount rate for each reporting unit. Management determined, after performing the quantitative analysis, that the fair value of the reporting units was less than the carrying amount and recognized an aggregate impairment charge of $511.4 million in the second quarter of 2025, a majority of which relates to the Tissue Technologies and Neurosurgery reporting units.
The principal considerations for our determination that performing procedures relating to the interim goodwill impairment assessments of the Tissue Technologies and Neurosurgery reporting units is a critical audit matter are (i) the significant judgment by management when developing the fair value estimate of the Tissue Technologies and Neurosurgery reporting units; (ii) a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating management’s significant assumptions related to revenue growth rates, cost of sales, terminal growth rate, and discount rate used in the income approach and comparable publicly-traded companies’ revenue and EBITDA multiples used in the market approach; and (iii) the audit effort involved the use of professionals with specialized skill and knowledge.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included testing the effectiveness of controls relating to management’s goodwill impairment assessments, including controls over the valuation of the Tissue Technologies and Neurosurgery reporting units. These procedures also included, among others (i) testing management’s process for developing the fair value estimate of the Tissue Technologies and Neurosurgery reporting units; (ii) evaluating the appropriateness of the income and market approaches used by management; (iii) testing the completeness and accuracy of underlying data used in the income and market approaches; and (iv) evaluating the reasonableness of the significant assumptions used by management related to the revenue growth rates, cost of sales, terminal growth rate, and discount rate used in the income approach and comparable publicly-traded companies’ revenue and EBITDA multiples used in the market approach. Evaluating management’s assumptions related to revenue growth rates and cost of sales involved evaluating whether the assumptions used by management were reasonable considering (i) the current and past performance of the Tissue Technologies and Neurosurgery reporting units; (ii) the consistency with external market and industry data; and (iii) whether the assumptions were consistent with evidence obtained in other areas of the audit. Professionals with specialized skill and knowledge were used to assist in evaluating (i) the appropriateness of the income and market approaches and (ii) the reasonableness of the terminal growth rate, discount rate, and comparable publicly-traded companies’ revenue and EBITDA multiples assumptions.
/s/ PricewaterhouseCoopers LLP
February 26, 2026
We have served as the Company’s auditor since 1989.
F-2
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS
| (Dollars in thousands, except per share amounts) | Years Ended December 31, | ||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Total Revenue, net | $ | $ | $ | ||||||||||||||
| Costs and Expenses: | |||||||||||||||||
| Cost of goods sold | |||||||||||||||||
| Research and development | |||||||||||||||||
| Selling, general and administrative | |||||||||||||||||
| Intangible asset amortization | |||||||||||||||||
| Goodwill impairment charge | |||||||||||||||||
| Total costs and expenses | |||||||||||||||||
| Operating (loss) income | ( | ||||||||||||||||
| Interest income | |||||||||||||||||
| Interest expense | ( | ( | ( | ||||||||||||||
| Other (expense) income, net | ( | ||||||||||||||||
| (Loss) income before income taxes | ( | ( | |||||||||||||||
| (Benefit) provision for income taxes | ( | ( | |||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | ||||||||||||
| Net (loss) income per share | |||||||||||||||||
| Basic | $ | ( | $ | ( | $ | ||||||||||||
| Diluted | $ | ( | $ | ( | $ | ||||||||||||
Weighted average common shares outstanding (Note 13. Net Income Per Share): | |||||||||||||||||
| Basic | |||||||||||||||||
| Diluted | |||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
CONSOLIDATED STATEMENTS OF COMPREHENSIVE INCOME
| (Dollars in thousands) | Years Ended December 31, | ||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | ||||||||||||
| Other comprehensive loss, before tax: | |||||||||||||||||
| Change in foreign currency translation adjustments | ( | ( | ( | ||||||||||||||
| Unrealized (loss) gain on derivatives | |||||||||||||||||
| Unrealized derivative (loss) gain arising during period | ( | ( | |||||||||||||||
| Less: Reclassification adjustments for (loss) gain included in net income | ( | ( | |||||||||||||||
| Unrealized (loss) gain on derivatives | ( | ( | |||||||||||||||
| Defined benefit pension plan - net gain (loss) arising during period | ( | ||||||||||||||||
| Total other comprehensive (loss) gain, before tax | ( | ( | ( | ||||||||||||||
| Income tax benefit (expense) related to items in other comprehensive gain (loss) | ( | ||||||||||||||||
| Total other comprehensive (loss) gain, net of tax | ( | ( | ( | ||||||||||||||
| Comprehensive (loss) income, net of tax | $ | ( | $ | ( | $ | ||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
CONSOLIDATED BALANCE SHEETS
| (Dollars in thousands, except per share amounts) | December 31, | ||||||||||
| 2025 | 2024 | ||||||||||
| ASSETS | |||||||||||
| Current assets: | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
| Short-term investments | |||||||||||
Trade accounts receivable, net of allowances of $ | |||||||||||
| Inventories, net | |||||||||||
| Prepaid expenses | |||||||||||
| Other current assets | |||||||||||
| Total current assets | |||||||||||
| Property, plant and equipment, net | |||||||||||
| Right of use asset - operating leases | |||||||||||
| Intangible assets, net | |||||||||||
| Goodwill | |||||||||||
| Deferred tax assets, net | |||||||||||
| Other assets | |||||||||||
| Total assets | $ | $ | |||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |||||||||||
| Current liabilities: | |||||||||||
| Current portion of borrowings under senior credit facility | $ | $ | |||||||||
| Short-term borrowings under securitization facility | |||||||||||
| Current portion of lease liability - operating leases | |||||||||||
| Convertible securities | |||||||||||
| Accounts payable, trade | |||||||||||
| Contract liabilities | |||||||||||
| Income tax payable | |||||||||||
| Accrued compensation | |||||||||||
| Accrued expenses and other current liabilities | |||||||||||
| Total current liabilities | |||||||||||
| Long-term borrowings under senior credit facility | |||||||||||
| Long-term borrowings under securitization facility | |||||||||||
| Lease liability - operating leases | |||||||||||
| Deferred tax liabilities | |||||||||||
| Other liabilities | |||||||||||
| Total liabilities | |||||||||||
| Stockholders’ equity: | |||||||||||
Preferred Stock; | |||||||||||
Common stock; $ | |||||||||||
| Additional paid-in capital | |||||||||||
Treasury stock, at cost; | ( | ( | |||||||||
| Accumulated other comprehensive (loss) | ( | ( | |||||||||
| Retained earnings | |||||||||||
| Total stockholders’ equity | |||||||||||
| Total liabilities and stockholders’ equity | $ | $ | |||||||||
F-5
The accompanying notes are an integral part of these consolidated financial statements.
F-6
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
| (Dollars in thousands) | Years Ended December 31, | ||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| OPERATING ACTIVITIES: | |||||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | ||||||||||||
| Adjustments to reconcile net income to net cash provided by operating activities: | |||||||||||||||||
| Depreciation and amortization | |||||||||||||||||
| Non-cash impairment charges | |||||||||||||||||
| Deferred income tax benefit | ( | ( | ( | ||||||||||||||
| Share-based compensation | |||||||||||||||||
| Amortization of debt issuance costs and expenses associated with debt refinancing | |||||||||||||||||
| Non-cash lease expense | ( | ||||||||||||||||
| Lease incentive | |||||||||||||||||
| Loss on disposal of property and equipment and construction in-progress | |||||||||||||||||
| Change in fair value of contingent consideration and others | ( | ( | |||||||||||||||
| Changes in assets and liabilities: | |||||||||||||||||
| Accounts receivable | ( | ||||||||||||||||
| Inventories | ( | ( | ( | ||||||||||||||
| Prepaid expenses and other current assets | ( | ( | |||||||||||||||
| Other non-current assets | ( | ( | |||||||||||||||
| Accounts payable, accrued expenses and other current liabilities | ( | ( | |||||||||||||||
| Contract liabilities | |||||||||||||||||
| Other non-current liabilities | ( | ( | ( | ||||||||||||||
| Net cash provided by operating activities | |||||||||||||||||
| INVESTING ACTIVITIES: | |||||||||||||||||
| Purchases of property and equipment | ( | ( | ( | ||||||||||||||
| Purchases of intangible assets | ( | ( | |||||||||||||||
| Purchases of short-term investments | ( | ( | ( | ||||||||||||||
| Proceeds from maturities of short-term investments | |||||||||||||||||
| Cash paid for business acquisitions, net of cash acquired | ( | ||||||||||||||||
| Net (payment) proceeds on swaps designated as net investment hedges | ( | ( | |||||||||||||||
| Net cash used in investing activities | ( | ( | ( | ||||||||||||||
| FINANCING ACTIVITIES: | |||||||||||||||||
| Proceeds from borrowings of long-term indebtedness | |||||||||||||||||
| Payments on debt | ( | ( | ( | ||||||||||||||
| Repayments of convertible debt | ( | ||||||||||||||||
| Payment of debt issuance costs | ( | ( | |||||||||||||||
| Purchase of treasury stock | ( | ( | ( | ||||||||||||||
| Payments for contingent considerations | ( | ( | |||||||||||||||
| Proceeds from exercise of stock options and employee stock purchase plan | |||||||||||||||||
| Cash taxes paid in net equity settlement | ( | ( | ( | ||||||||||||||
| Net cash provided by (used in) financing activities | ( | ||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | ( | ||||||||||||||||
| Net decrease in cash and cash equivalents | ( | ( | ( | ||||||||||||||
| Cash and cash equivalents at beginning of period | |||||||||||||||||
| Cash and cash equivalents at end of period | $ | $ | $ | ||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-7
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS' EQUITY
(Dollars in thousands)
| Common Stock | Treasury Stock | Additional Paid-In Capital | Accumulated Other Comprehensive Income (loss) | Retained Earnings | Total Equity | ||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||||||||||||
Balance, January 1, 2023 | $ | ( | $ | ( | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income, net of tax | — | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock through employee stock purchase plan | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock for vesting of share-based awards, net of shares withheld for taxes and forfeitures | ( | — | — | ( | |||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Accelerated shares repurchased | — | — | ( | ( | — | — | ( | ||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2023 | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive loss, net of tax | — | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock through employee stock purchase plan | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock for vesting of share-based awards, net of shares withheld for taxes and forfeitures | — | — | |||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Accelerated shares repurchased | — | — | ( | ( | ( | — | — | ( | |||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2024 | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||||
| Other comprehensive loss, net of tax | — | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||
| Issuance of common stock through employee stock purchase plan | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||
| Issuance of common stock for vesting of share-based awards, net of shares withheld for taxes and forfeitures | ( | — | — | ( | |||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||
| Other Adjustments | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||
Balance, December 31, 2025 | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-8
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. BUSINESS
Integra LifeSciences Holdings Corporation (the “Company”) was incorporated in Delaware in 1989. The Company is a worldwide leader in medical technology. The Company was founded with the acquisition of an engineered collagen technology platform used to repair and regenerate tissue. Since then, Integra has developed numerous product lines from this technology for applications ranging from burn and deep tissue wounds to the repair of dura mater in the brain, as well as nerves and tendons. The Company has expanded its base regenerative technology business to include surgical instruments, neurosurgical products, Ear, Nose, Throat (“ENT”) products, and advanced wound care through global acquisitions and product development to meet the evolving needs of its customers and enhance patient care. The Company sells its products directly through various salesforces and through a variety of other distribution channels.
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
BASIS OF PRESENTATION
PRINCIPLES OF CONSOLIDATION
USE OF ESTIMATES
CASH AND CASH EQUIVALENTS
The Company considers all short-term, highly liquid investments purchased with original maturities of three months or less to be cash equivalents. These investments are carried at cost, which approximates fair value.
SHORT-TERM INVESTMENTS
The Company has short-term investments, which primarily consisted of time deposits with original maturities between three months and one year. The short-term investments, which are valued based on Level 1 measurements in the fair value hierarchy, totaled approximately $28.7 million at December 31, 2025 compared to $27.2 million at December 31, 2024. Interest and dividends are recorded in income when earned.
TRADE ACCOUNTS RECEIVABLE AND ALLOWANCES FOR DOUBTFUL ACCOUNTS RECEIVABLE
Trade accounts receivable are recorded at the invoiced amount and do not bear interest. The Company grants credit to customers in the normal course of business but generally does not require collateral or any other security to support its receivables.
The Company evaluates the collectability of accounts receivable based on a combination of factors. The Company recognizes a provision for doubtful accounts that reflects the Company’s estimate of expected credit losses for trade accounts receivable. In circumstances where a specific customer is unable to meet its financial obligations to the Company, a provision to the allowances for doubtful accounts is recorded against amounts due to reduce the net recognized receivable to the amount that is reasonably expected to be collected. For all other customers, the Company evaluates measurement of all expected credit losses for trade receivables held at the reporting date based on historical experience, current conditions, and reasonable and supportable forecasts.
F - 9
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company’s exposure to credit losses may increase if its customers are adversely affected by changes in healthcare laws, coverage and reimbursement, economic pressures or uncertainty associated with local or global economic recessions.
The below table shows the roll forward of the allowance for doubtful accounts for the years ended December 31, 2025, 2024, and 2023:
| Balance at Beginning of Period | Charged to Costs and Expenses | Acquisition | Deductions | Balance at End of Period | |||||||||||||||||||||||||
| Dollars in thousands | |||||||||||||||||||||||||||||
| Year Ended: | |||||||||||||||||||||||||||||
| December 31, 2025 | $ | ( | $ | ||||||||||||||||||||||||||
| December 31, 2024 | $ | ( | $ | ||||||||||||||||||||||||||
| December 31, 2023 | $ | ( | $ | ||||||||||||||||||||||||||
Deductions primarily relates to allowance for doubtful accounts written off during the year, net of recoveries and other adjustments.
INVENTORIES
| December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Finished goods | $ | $ | |||||||||
| Work in process | |||||||||||
| Raw materials | |||||||||||
| Total inventories, net | $ | $ | |||||||||
At each balance sheet date, the Company evaluates inventories for excess quantities, obsolescence or shelf life expiration. This evaluation includes analysis of historical sales levels by product, projections of future demand, the risk of technological or competitive obsolescence for products, general market conditions, a review of the shelf life expiration dates for products, as well as the feasibility of reworking or using excess or obsolete products or components in the production or assembly of other products that are not obsolete or for which there are not excess quantities in inventory. To the extent that management determines there are excess or obsolete inventory or quantities with a shelf life that is too near its expiration for the Company to reasonably expect that it can sell those products prior to their expiration, the Company adjusts the carrying value to estimated net realizable value.
Any tariffs paid have been capitalized in inventory and is recognized in our cost of goods sold as those products are sold. For the years ended December 31, 2025 and 2024, respectively, the Company paid approximately $19.9 million and $0.5 million of tariffs on imported goods. Of this amount, $6.5 million and $0.5 million, respectively, was recognized in cost of goods sold in the consolidated statements of operations.
F-10
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
PROPERTY, PLANT AND EQUIPMENT
Property, plant and equipment are stated at historical cost less accumulated depreciation and any impairment charges. The Company provides for depreciation using the straight-line method over the estimated useful lives of the assets. Leasehold improvements are amortized over the lesser of the lease term or the useful life. The cost of major additions and improvements is capitalized, while maintenance and repair costs that do not improve or extend the lives of the respective assets are charged to operations as incurred.
The cost of computer software developed or obtained for internal use is accounted for in accordance with FASB Topic 350-40, Intangibles - Goodwill and Other - Internal-Use Software (“ASC 350-40”).
Property, plant and equipment balances and corresponding lives were as follows:
| December 31, | |||||||||||||||||
| Dollars in thousands | 2025 | 2024 | Useful Lives | ||||||||||||||
| Land | $ | $ | |||||||||||||||
| Buildings and building improvements | |||||||||||||||||
| Leasehold improvements | |||||||||||||||||
| Machinery and equipment | |||||||||||||||||
| Information systems and hardware | |||||||||||||||||
| Furniture, fixtures, and office equipment | |||||||||||||||||
| Construction-in-progress | |||||||||||||||||
| Total | |||||||||||||||||
| Less: Accumulated depreciation | ( | ( | |||||||||||||||
| Property, plant and equipment, net | $ | $ | |||||||||||||||
Depreciation expense associated with property, plant and equipment was $44.8 million, $42.4 million, and $40.9 million for the years ended December 31, 2025, 2024, and 2023, respectively.
CAPITALIZED INTEREST
ACQUISITIONS
The Company accounts for the acquisition of a business in accordance with FASB Topic 805, Business Combinations (“ASC 805”). Amounts paid to acquire a business are allocated to the assets acquired and liabilities assumed based on their respective estimated fair values as of the date of acquisition in accordance with the fair value hierarchy described in FASB Topic 820, Fair Value Measurement (“ASC 820”). Any excess of the purchase price over the fair value of the net assets acquired is recorded as goodwill. Transaction costs and costs to restructure the acquired company are expensed as incurred. Results of operations of acquired companies are included in the Company’s results of operations as of the respective acquisition dates.
Contingent consideration is recorded at fair value as measured on the date of acquisition. The value recorded is based on estimates of future financial projections under various potential scenarios using either a Monte Carlo simulation or the probability-weighted income approach derived from revenue estimates and probability assessment with respect to the likelihood of achieving contingent obligations. Contingent payments related to acquisitions consist of development, regulatory, and commercial milestone payments, in addition to sales-based payments, and are valued using discounted cash flow techniques. Each quarter until such contingent amounts are earned, the fair value of the liability is remeasured at each reporting period and adjusted as a component of operating expenses based on changes to the underlying assumptions. The change in the fair value of sales-based payments is based upon future revenue estimates and increases or decreases as revenue estimates or expectation of timing of payment charges. The estimates used to determine the fair value of the contingent consideration liability are subject to significant judgment and actual results could differ from the amounts originally recorded.
F-11
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company determines the fair value of acquired intangible assets based on detailed valuations that use certain information and assumptions provided by management. Determining the fair value of these intangible assets acquired as part of a business combination requires the Company to make significant estimates. These estimates include the estimated annual net cash flows including application of forecasted revenue growth rates, cost of sales, the discount rate that appropriately reflects the risk inherent in each future cash flow stream, the obsolescence curve, and an assessment of the asset’s life cycle, as well as other factors such as the consideration of legal, technical, regulatory, economic, and competitive risks. The fair value assigned to acquired intangible assets is determined by estimating the future cash flows of each project or technology and discounting the net cash flows back to their present values. The discount rate used is determined at the time of measurement in accordance with accepted valuation methodologies.
In-process research and development (“IPR&D”) acquired in connection with the acquisition of a business in accordance with ASC 805 is initially recognized at fair value and characterized as an indefinite-lived intangible asset, irrespective of whether the acquired IPR&D has an alternative future use. The Company has not acquired any IPR&D in connection with the acquisition of a business during the years ended December 31, 2025 and 2024.
Research and development costs incurred after the acquisition are expensed as incurred. Upon receipt of regulatory approval, the indefinite-lived intangible asset is then accounted for as a finite-lived intangible asset and amortized on a straight-line basis or accelerated basis, as appropriate, over its estimated useful life. If the project is subsequently abandoned, the indefinite-lived intangible asset is charged to expense.
Due to the uncertainty associated with research and development projects, there are risks that actual results will differ materially from the original cash flow projections or that the research and development project will not result in a successful commercial product. The risks associated with achieving commercialization include, but are not limited to, delay or failure to obtain regulatory approvals to conduct clinical trials, delay or failure to obtain required market clearances, delays or issues with patent issuance, or validity and litigation.
Refer to Note 4. Acquisitions and Divestitures for more information.
GOODWILL AND OTHER INTANGIBLE ASSETS
The excess of the cost over the fair value of net assets of acquired businesses is recorded as goodwill. In accordance with FASB Topic 350, Intangibles - Goodwill and Other (“ASC 350”), goodwill is not subject to amortization but is tested for impairment at the reporting unit level annually in the third quarter. Additionally, the Company may perform interim tests of goodwill for impairment if an event occurs or circumstances change that could potentially reduce the fair value of a reporting unit below its carrying amount. The carrying value of each reporting unit is determined by assigning the assets and liabilities, including the existing goodwill and intangible assets, to those reporting units. Refer to Note 7. Goodwill and Other Intangibles for more information.
The Company has two reportable segments with three underlying reporting units. Refer to Note 16. Segment and Geographic Information for more information on reportable segments.
Other intangible assets include patents, trademarks, purchased technology, and supplier and customer relationships. Identifiable intangible assets are initially recorded at fair market value at the time of acquisition generally using an income or cost approach. Developed technologies and other definite-lived intangible assets are amortized over their estimated useful lives either using the straight-line method or, if reliably determinable, based on the pattern of which the economic benefit of the asset is expected to be utilized. The Company capitalizes costs incurred to renew or extend the term of recognized intangible assets, if applicable, and amortizes those costs over their expected useful lives.
F-12
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
In accordance with ASC 350, the Company tests its indefinite-lived intangible assets for impairment annually in the third quarter. Additionally, the Company may perform interim tests if an event occurs or circumstances change that could potentially reduce the fair value of an indefinite lived intangible asset below its carrying amount. The Company tests indefinite-lived intangible assets for impairment by either performing a qualitative evaluation or a quantitative test. The qualitative evaluation is an assessment of factors, including specific operating results as well as industry, market and general economic conditions, to determine whether it is more likely than not that the fair values of the intangible asset is less than its carrying amount. The Company may elect to bypass this qualitative evaluation and perform a quantitative test. The quantitative test uses an income approach to determine the fair value of the indefinite-lived intangible asset. The income approach utilizes the estimated discounted cash flows for the indefinite-lived intangible asset. Estimates and assumptions used in the income approach to calculate projected future discounted cash flows included revenue growth rates, cost of sales, terminal growth rate, and a discount rate for each indefinite-lived intangible asset. Discount rates are determined using a weighted average cost of capital that considers market and industry data as well as the Company’s specific risk factors that are likely to be considered by a market participant. The weighted-average cost of capital is the Company’s estimate of the overall after-tax rate of return required by equity and debt holders of a business enterprise. The assumptions used are inherently subject to uncertainty and slight changes in these assumptions could have a significant impact on the concluded value. The estimates and assumptions applied represent a Level 3 measurement in the fair value hierarchy described in ASC 820. Level 3 inputs are supported by limited or no market activity and reflect the Company’s assumptions in measuring fair value.
Developed technologies and other definite-lived intangible assets are amortized over their estimated useful lives either using the straight-line method or, if reliably determinable, based on the pattern of which the economic benefit of the asset is expected to be utilized. Definite-lived intangible assets tested periodically for impairment in accordance with FASB Topic 360, Property, Plant and Equipment (“ASC 360”) when events or changes in circumstances indicate that an asset’s carrying value may not be recoverable. The evaluation for recoverability involves comparing the carrying amount of the definite-lived intangible asset to the Company’s expectations of the future undiscounted cash flows derived from the definite-lived intangible asset. In the event the carrying value of the definite-lived intangible asset exceeds the undiscounted future cash flows expected to be derived from the definite-lived intangible asset over its remaining estimated useful life, the definite-lived intangible asset is considered not recoverable and the definite-lived intangible asset is tested for impairment. An impairment loss is measured as the excess of the asset’s carrying value over its fair value, calculated using discounted future cash flows. The computed impairment loss is recognized in the period that the impairment occurs.
Refer to Note 7. Goodwill and Other Intangibles for more information.
LONG-LIVED ASSETS
INTEGRA FOUNDATION
F-13
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
DERIVATIVES
The Company develops, manufactures, and sells medical devices globally and its earnings and cash flows are exposed to market risk from changes in interest rates and currency exchange rates. The Company addresses these risks through a risk management program that includes the use of derivative financial instruments and operates the program pursuant to documented corporate risk management policies. All derivative financial instruments are recognized in the financial statements at fair value in accordance with the authoritative guidance. Under the guidance, for those instruments that are designated and qualify as hedging instruments, the hedging instrument must be designated as a fair value hedge, cash flow hedge, or a hedge of a net investment in a foreign operation, based on the exposure being hedged. The accounting for changes in the fair value of a derivative instrument depends on whether it has been designated and qualifies as part of a hedging relationship and, further, on the type of hedging relationship. The Company’s derivative instruments do not subject its earnings or cash flows to material risk, and gains and losses on these derivatives generally offset losses and gains on the item being hedged. The Company has not entered into derivative transactions for speculative purposes. From time to time, the Company may enter into derivatives that are not designated as hedging instruments in order to protect itself from currency volatility due to intercompany balances.
All derivative instruments are recognized at their fair values as either assets or liabilities on the balance sheet. The Company determines the fair value of its derivative instruments using the fair value hierarchy described in ASC 820, by considering the estimated amount the Company would receive to sell or transfer these instruments at the reporting date and by taking into account expected forward interest rates, currency exchange rates, the creditworthiness of the counterparty for assets, and its creditworthiness for liabilities. In certain instances, the Company utilizes a discounted cash flow model to measure fair value. Generally, the Company uses inputs that include quoted prices for similar assets or liabilities in active markets, other observable inputs for the asset or liability and inputs derived principally from, or corroborated by, observable market data by correlation or other means. The Company has classified all of its derivative assets and liabilities within Level 2 of the fair value hierarchy because observable inputs are available for substantially the full term of its derivative instruments. The Company classifies derivatives designated as hedges in the same category as the item being hedged for cash flow presentation purposes.
Refer to Note 6. Derivative Instruments for more information.
FOREIGN CURRENCY
INCOME TAXES
Income taxes are accounted for by using the asset and liability method. Deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax basis. A valuation allowance is provided when it is more likely than not that some portion or all of the deferred tax assets will not be realized. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period when the change is enacted.
The Company recognizes a tax benefit from an uncertain tax position only if it is more likely than not to be sustained upon examination based on the technical merits of the position. Reserves are established for positions that don't meet this recognition threshold. The reserve is measured as the largest amount of benefit determined on a cumulative probability basis that the Company believes is more likely than not to be realized upon ultimate settlement of the position. These reserves are classified as long-term liabilities in the consolidated balance sheets of the Company, unless the reserves are expected to be paid in cash during the next twelve months, in which case they are classified as current liabilities. The Company also records interest and penalties accrued in relation to uncertain tax benefits as a component of income tax expense.
While the Company believes it has identified all reasonable exposures and the reserve it has established is appropriate under the circumstances, it is possible that additional exposures exist and that exposures may be settled at amounts different than the amounts reserved. It is also possible that changes in facts and circumstances could cause the Company to either materially increase or reduce the carrying amount of its tax reserve.
F-14
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Refer to Note 12. Income Taxes for more information.
REVENUE RECOGNITION
Revenue is recognized upon the transfer of control of promised products or services to the customers in an amount that reflects the consideration the Company expects to receive in exchange for those products and services in accordance with FASB Topic 606, Revenue from Contracts with Customers (“ASC 606”).
Total revenue, net, includes product sales, product royalties and other revenues, such as fees received from services.
For products shipped with FOB shipping point terms, the control of the product passes to the customer at the time of shipment. For shipments in which the control of the product is transferred when the customer receives the product, the Company recognizes revenue upon receipt by the customer. Certain products that the Company produces for private label customers have no alternative use and the Company has a right of payment for performance to date. Revenues from those products are recognized over the period that the Company manufactures these products, which is typically one month to three months . The Company uses the input method to measure the manufacturing activities completed to date, which depicts the progress of the Company’s performance obligation of transferring control of goods being manufactured for private label customers.
A portion of the Company’s product revenue is generated from consigned inventory maintained at hospitals and distributors, and also from inventory physically held by field sales representatives. For these types of products sales, the Company retains control until the product has been used or implanted, at which time revenue is recognized.
Revenues from sale of products and services are evidenced by either a contract with the customer or a valid purchase order and an invoice which includes all relevant terms of sale. For product sales, invoices are generally issued upon the transfer of control (or upon the completion of the manufacturing in the case of the private label transactions recognized over time) and are typically payable 30 days after the invoice date. The Company performs a review of each specific customer’s creditworthiness and ability to pay prior to acceptance as a customer. Further, the Company performs periodic reviews of its customers’ creditworthiness prospectively.
The Company also maintains a provision for estimated returns and allowances in the same period that the related revenue is recorded. This reserve is based upon an analysis of actual credit memos issued for pricing issues or returned goods over an extended period, as well as assumptions about outstanding accounts receivable and judgment in interpreting the data.
Refer to Note 3. Revenue from Contracts with Customers for more information.
RESEARCH AND DEVELOPMENT
EMPLOYEE TERMINATION BENEFITS
As of December 31, 2025, the Company did not have a written severance plan, but had a history of providing consistent benefits for employees in the case of involuntary termination. In situations outside the U.S., there are minimum statutory termination benefits requirements by country that must be paid to the affected employees. The Company records employee severance costs associated with these restructuring activities in accordance with the authoritative guidance for non-retirement post-employment benefits. Charges associated with these activities are recorded when the payment of benefits is probable and can be reasonably estimated. In situations where the Company pays out termination benefits in excess of past practices or statutory minimum amounts based on management’s discretion, the Company records these termination costs once communication is made to the affected employees.
F-15
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company incurred employee termination costs on restructuring activities in the consolidated statement of operations for the years ended December 31, 2025 and 2024. The following table summarizes the restructuring related accrual balances included within accrued expenses and other current liabilities in the consolidated balance sheet for the years ended December 31, 2025 and 2024.
| Years Ended December 31, | ||||||||||||||
| (Dollars in thousands) | 2025 | 2024 | ||||||||||||
| Balance, beginning of the year | $ | $ | ||||||||||||
| Charges: | ||||||||||||||
| Payments and other adjustments | ( | ( | ||||||||||||
| Balance, end of the year | $ | $ | ||||||||||||
STOCK-BASED COMPENSATION
Relevant authoritative guidance requires companies to recognize the expense related to the fair value of their stock-based compensation awards. Stock-based compensation expense for stock option awards are based on the grant date fair value using the binomial distribution model. The Company recognizes compensation expense for stock option awards, restricted stock awards, performance stock awards and contract stock awards over the requisite service period of the award. All excess tax benefits and tax deficiencies from stock-based compensation are included in the provision for income taxes in the consolidated statement of operations.
Refer to Note 9. Stock Based Compensation for more information.
RETIREMENT BENEFIT PLANS
Defined Benefit Pension Plans
The Company maintains defined benefit pension plans that cover certain employees in France, Japan, Germany and Switzerland. Various factors are considered in determining the pension liability, including the number of employees expected to be paid their salary levels and years of service, the expected return on plan assets, the discount rate used to determine the benefit obligations, the timing of benefit payments and other actuarial assumptions.
Retirement benefit plan assumptions are reassessed on an annual basis or more frequently if changes in circumstances indicate a re-evaluation of assumptions are required. The key benefit plan assumptions are the discount rate and expected rate of return on plan assets. The discount rate is based on average rates on bonds that match the expected cash outflows of the benefit plans. The expected rate of return is based on historical and expected returns on the various categories of plan assets.
The Company uses the corridor approach in measuring the amount of net periodic benefit pension cost to recognize each period. The corridor approach defers all actuarial gains and losses resulting from variances between actual results and actuarial assumptions. Those unrecognized gains and losses are amortized when the net gains and losses exceed 10% of the greater of the market-related value of plan assets or the projected benefit obligation at the beginning of the year. The amount in excess of the corridor is amortized over the average remaining service period to retirement date of active plan participants.
Deferred Compensation Plan
The Company maintains a deferred compensation plan in which certain employees of the Company may defer the payment and taxation of up to 75 % of their base salary and up to 100 % of bonus amounts and other eligible cash compensation.
This deferred compensation is invested in funds offered under the Plan and is valued based on Level 1 measurements in the fair value hierarchy. The purpose of the plan is to retain key employees by providing them with an opportunity to defer a portion of their compensation as elected by the participant in accordance with the plan. Any amounts set aside to defray the liabilities assumed by the Company will remain the general assets of the Company until such amounts are distributed to the participants. Assets of the Company’s deferred compensation plan are included in other current assets and recorded at fair value based on their quoted market prices. Offsetting liabilities relating to the deferred compensation plan are included in other liabilities.
Refer to Note 10. Retirement Benefit Plans for more information.
CONCENTRATION OF CREDIT RISK
Financial instruments, which potentially subject the Company to concentrations of credit risk, consist principally of cash and cash equivalents, which are held at major financial institutions, investment-grade marketable debt securities, and trade receivables.
F-16
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
RECENT ACCOUNTING PRONOUNCEMENTS
In December 2023, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures (“ASU 2023-09”) which enhances the transparency of income tax disclosures by expanding annual disclosure requirements related to the rate reconciliation and income taxes paid. The amendments are effective for fiscal years beginning after December 15, 2024. The amendments should be applied on a prospective basis. Retrospective application to all prior periods presented in the financial statements is permitted. The Company has adopted ASU 2023-09 for the annual period ended on December 31, 2025 on a prospective basis.
In November 2024, the FASB issued ASU 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses (“ASU 2024-03”), and in January 2025, the FASB issued ASU 2025-01, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Clarifying the Effective Date (“ASU 2025-01”). ASU 2024-03 requires additional disclosure of the nature of expenses included in the income statement. The standard requires disclosures about specific types of expenses included in the expense captions presented in the income statement. This ASU is effective for fiscal years beginning after December 15, 2026, and interim periods beginning after December 15, 2027, with early adoption permitted. The requirements should be applied on a prospective basis while retrospective application is permitted. The Company is currently evaluating the impact that the adoption of this guidance will have on its disclosures.
In September 2025, the FASB issued ASU 2025-06, Intangibles - Goodwill and Other - Internal-Use Software (Subtopic 350-40): Targeted Improvements to the Accounting for Internal-Use Software (“ASU 2025-06”), which removes all references to software development project stages and requires entities to start capitalizing software costs when both of the following occur: (i) management has authorized and committed to funding the software project and (ii) it is probable that the project will be completed and the software will be used to perform the function intended. This ASU is effective for fiscal years beginning after December 15, 2027, and interim periods within those fiscal years, with early adoption permitted as of the beginning of a fiscal year. The amendment may be applied prospectively, retrospectively, or via a modified prospective transition method. The Company is currently evaluating the impact that the adoption of this guidance will have on its consolidated financial statements.
In November 2025, the FASB issued ASU 2025-09, Derivatives and Hedging (Topic 815): Hedge Accounting Improvements (“ASU 2025-09”), which amends existing guidance to clarify and enhance the hedge accounting guidance in FASB Topic 815 and better align hedge accounting with the economics of an entity’s risk management strategies to enable entities to achieve and maintain hedge accounting for highly effective economic hedges. This ASU is effective for fiscal years beginning after December 15, 2026, and interim periods within those fiscal years, with early adoption permitted. The amendment may be applied prospectively for all hedging relationships. The Company is currently evaluating the impact that the adoption of this guidance will have on its consolidated financial statements.
In December 2025, the FASB issued ASU 2025-10, Government Grants (Topic 832): Accounting for Government Grants by Business Entities (“ASU 2025-10”), which establishes authoritative guidance on the recognition, measurement, presentation, and disclosure for government grants received by business entities. This amendment defines a government grant, establishes when and how a grant related to an asset or income is recognized and measured, and includes presentation and disclosure requirements. This ASU is effective for fiscal years beginning after December 15, 2028, and interim periods within those fiscal years, with early adoption permitted. The amendment may be applied using a modified prospective, modified retrospective, or fully retrospective transition method. The Company is currently evaluating the impact that the adoption of this guidance will have on its consolidated financial statements.
There are no other recently issued accounting pronouncements that are expected to have any significant effect on the Company’s financial position, results of operations, or cash flows.
SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION
Cash paid for interest during the years ended December 31, 2025, 2024, and 2023 was $81.4 million (net of $5.0 million that was capitalized into construction in progress), $64.4 million (net of $4.5 million that was capitalized into construction in progress), and $44.3 million (net of $2.4 million that was capitalized into construction in progress), respectively.
Cash paid for income taxes, net of refunds, for the years ended December 31, 2025, 2024, and 2023 were $17.2 million, $22.7 million, and $23.6 million, respectively.
F-17
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The following table outlines cash paid for income tax, net of refunds, by taxing jurisdiction:
| Dollars in thousands | December 31, | |||||||
| 2025 | ||||||||
| Federal | $ | |||||||
| State | ||||||||
| Foreign | ||||||||
| Total | $ | |||||||
From the above adjustments, income taxes paid (net of refunds) exceed the 5% of taxes paid in the following jurisdictions:
| Dollars in thousands | ||||||||
| Foreign | ||||||||
| Germany | $ | |||||||
| Ireland | ||||||||
| Israel | ||||||||
| Switzerland | ||||||||
| Total | $ | |||||||
NON-CASH INVESTING AND FINANCING ACTIVITIES
Property and equipment purchases included in liabilities at December 31, 2025, 2024, and 2023 were $7.5 million, $12.3 million, and $10.0 million, respectively.
3. REVENUES FROM CONTRACTS WITH CUSTOMERS
Summary of Accounting Policies on Revenue Recognition
Revenue is recognized upon the transfer of control of promised products or services to the customers in an amount that reflects the consideration the Company expects to receive in exchange for those products and services.
Performance Obligations
The Company’s performance obligations consist mainly of transferring control of goods and services identified in the contracts, purchase orders, or invoices. The Company has no significant multi-element contracts with customers.
Significant Estimates
Usage-based royalties and licenses are estimated based on the provisions of contracts with customers and recognized in the same period that the royalty-based products are sold by the Company’s strategic partners. The Company estimates and recognizes royalty revenue based upon communication with licensees, historical information, and expected sales trends. Differences between actual reported licensee sales and those that were estimated are adjusted in the period in which they become known, which is typically the following quarter. Historically, such adjustments have not been significant.
The Company estimates returns, price concessions, and discount allowances using the expected value method based on historical trends and other known factors. Rebate allowances are estimated using the most likely method based on each customer contract.
The Company’s return policy, as set forth in its product catalogs and sales invoices, requires review and authorization in advance prior to the return of product. Upon the authorization, a credit will be issued for the goods returned within a set amount of days from the shipment, which is generally 90 days.
The Company disregards the effects of a financing component if the Company expects, at contract inception, that the period between the transfer and customer payment for the goods or services will be one year or less. The Company has no significant revenues recognized on payments expected to be received more than one year after the transfer of control of products or services to customers.
F-18
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Contract Assets and Liabilities
Revenues recognized from the Company’s private label business that are not invoiced to the customers as a result of recognizing revenue over time are recorded as a contract asset included in the prepaid expenses and other current assets account in the consolidated balance sheet. Upon invoicing to the customer, the balance is recorded in trade receivable, net in the consolidated balance sheet.
Other operating revenues may include fees received under service agreements. Non-refundable fees received under multiple-period service agreements are recognized as revenue as the Company satisfies the performance obligations to the other party. A portion of the transaction price allocated to the performance obligations to be satisfied in the future periods is recognized as contract liability.
The following table summarized the changes in the contract asset and liability balances for the year ended December 31, 2025:
| Dollars in thousands | Total | ||||
| Contract Asset | |||||
Contract asset, January 1, 2025 | $ | ||||
| Transferred to trade receivable from contract asset included in beginning of the year contract asset | ( | ||||
| Contract asset, net of transferred to trade receivables on contracts during the period | |||||
Contract asset, December 31, 2025 | $ | ||||
| Contract Liability | |||||
Contract liability, January 1, 2025 | $ | ||||
| Recognition of revenue included in beginning of year contract liability | ( | ||||
| Contract liability, net of revenue recognized on contracts during the period | |||||
| Foreign currency translation | |||||
Contract liability, December 31, 2025 | $ | ||||
At December 31, 2025, the short-term portion of the contract liability of $11.5 million, representing 50 % of unsatisfied or partially unsatisfied performance obligations, is expected to be recognized as revenue within 12 months and is included in current liabilities in the consolidated balance sheet. The long-term portion of $11.5 million, representing the remaining balance to be recognized thereafter, is included in other liabilities in the consolidated balance sheet.
As of December 31, 2025, the Company is expected to recognize revenue from unsatisfied or partially satisfied performance obligations of approximately $11.5 million in 2026, $6.9 million in 2027, $3.4 million in 2028, $0.9 million in 2029 and $0.3 million in 2030.
Shipping and Handling Fees
Product Warranties
Certain of the Company’s medical devices, including monitoring systems and neurosurgical systems, are designed to operate over long periods of time. These products are sold with warranties which may extend for up to two years from the date of purchase. The warranties are not considered a separate performance obligation. The Company estimates its product warranties using the expected value method based on historical trends and other known factors. The Company includes them in accrued expenses and other current liabilities in the consolidated balance sheet.
Taxes Collected from Customers
F-19
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Disaggregated Revenue
The following table presents revenues disaggregated by the major sources of revenues for years-ended December 31, 2025, 2024, and 2023 (dollar amounts in thousands):
| Year Ended December 31, 2025 | Year Ended December 31, 2024 | Year Ended December 31, 2023 | |||||||||
| Neurosurgery | $ | $ | $ | ||||||||
| Instruments | |||||||||||
ENT(1)(2) | |||||||||||
| Total Codman Specialty Surgical | |||||||||||
| Wound Reconstruction and Care | |||||||||||
| Private Label | |||||||||||
| Total Tissue Technologies | |||||||||||
| Total revenue | $ | $ | $ | ||||||||
(1) See Note 4. Acquisitions and Divestitures for details surrounding the acquisition of Acclarent on April 1, 2024.
(2) Prior period revenues included within our instruments business have been reclassified under the ENT business.
See Note 16. Segment and Geographic Information for details of revenues based on the location of the customer.
4. ACQUISITIONS AND DIVESTITURES
Durepair® Acquisition
On October 2, 2024, the Company completed the acquisition of the product rights for the Durepair® Regeneration Matrix (“Durepair”), a non-synthetic dura substitute for repair of the dura mater during neurosurgical procedures, from Medtronic plc for total cash consideration of $45.0 million. The Company made a cash payment of $10.0 million upon the closing of the acquisition in October 2024, $15.0 million on the first anniversary of the acquisition in October 2025, and will make an additional cash payment of $20.0 million upon the second anniversary of the acquisition in October 2026. The additional cash payment to be made in October 2026 is included at its present value in accrued expenses and other current liabilities as of December 31, 2025.
The acquisition of the product rights for Durepair, which consist of certain patents and trademarks, regulatory approvals, and other records, has been accounted for as an asset acquisition in accordance with FASB Topic 805, Business Combinations (“ASC 805”) as the acquisition does not include an assembled workforce and substantially all of the fair value of the assets acquired is concentrated in a single identifiable intangible asset.
Acclarent, Inc. Acquisition
On April 1, 2024, the Company completed the acquisition of all of the outstanding capital stock of Acclarent, Inc. (“Acclarent”), a developer and marketer of medical devices used in ear, nose, throat (“ENT”) procedures, from Ethicon, Inc., a subsidiary of Johnson & Johnson, for approximately $282.0 million in cash, subject to customary adjustments set forth in the purchase agreement related to working capital balances transferred to the Company. In the second half of 2024, the Company finalized and settled the working capital adjustment in the amount of $4.2 million, which resulted in a reduction to goodwill and also recognized a measurement period adjustment to recognize deferred tax liabilities of $1.1 million with a corresponding increase to goodwill as a result of a change to the estimated deferred tax rate applied and the book-to-tax difference associated with fixed assets acquired.
The addition of Acclarent’s ENT product portfolio, including sinus balloon dilation, eustachian tube balloon dilation, and surgical navigation systems technologies, and dedicated salesforce enhanced the Company’s position in the ENT specialty device market. Acclarent’s results of operations have been reported in the Company’s Codman Specialty Surgical reportable segment from the date of acquisition.
Assets Acquired and Liabilities Assumed at Fair Value
The Acclarent acquisition has been accounted for using the acquisition method of accounting in accordance with ASC 805. This method requires that assets acquired and liabilities assumed in a business combination are recognized at their fair values as of the acquisition date.
F-20
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The following table summarizes the fair values of the assets acquired and liabilities assumed at the acquisition date:
| Dollars in thousands | Estimated Fair Value | Estimated Useful Life | |||||||||
| Current assets: | |||||||||||
| Cash | $ | ||||||||||
Trade accounts receivable, net of allowances of $ | |||||||||||
| Inventories, net | |||||||||||
| Prepaid expenses | |||||||||||
| Other current assets | |||||||||||
| Total current assets | $ | ||||||||||
| Property, plant and equipment, net | |||||||||||
| Right of use asset - operating leases | |||||||||||
| Intangible assets, net | |||||||||||
| Completed technology | |||||||||||
| Trademarks/brand names | |||||||||||
| All other | |||||||||||
| Goodwill | |||||||||||
| Deferred tax assets | |||||||||||
| Total assets acquired | $ | ||||||||||
| Current liabilities: | |||||||||||
| Accounts payable, trade | $ | ||||||||||
| Contract liabilities | |||||||||||
| Accrued compensation | |||||||||||
| Accrued expenses and other current liabilities | |||||||||||
| Current portion of lease liability - operating leases | |||||||||||
| Total current liabilities | $ | ||||||||||
| Lease liability - operating leases | |||||||||||
| Deferred tax liabilities | |||||||||||
| Total liabilities assumed | $ | ||||||||||
| Net assets acquired | $ | ||||||||||
The carrying value of trade accounts receivable, prepaid expenses, other current assets, accounts payable, contract liabilities, accrued compensation, accrued expenses and other current liabilities, as well as certain other current and non-current assets and liabilities, generally represented the fair value at the date of acquisition.
Intangible Assets
The estimated fair value of the intangible assets acquired was determined using the multi-period, excess earnings method of the income approach, which estimates value based on the present value of future economic benefits attributable to the intangible assets. The significant assumptions used in developing the valuation included the estimated annual net cash flows including application of revenue growth rates, cost of sales, the discount rate that appropriately reflects the risk inherent in each future cash flow stream, obsolescence rate, and an assessment of the asset’s life cycle, as well as other factors. The assumptions used in the financial forecasts were based on historical data, supplemented by current and anticipated growth rates, management plans, and market-comparable information. Fair value determinations require considerable judgment and are sensitive to changes in underlying assumptions and factors. The intangible assets acquired have a weighted average useful life of 11 years.
The Company used a discount rate of 12.2 % to arrive at the present value for the acquired intangible assets to reflect the rate of return a market participant would expect to earn and incremental commercial uncertainty in the cash flow projections. No assurances can be given that the underlying assumptions used to prepare the discounted cash flow analysis will not change. For these and other reasons, actual results may vary significantly from estimated results.
F-21
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Goodwill
Goodwill is the excess of the consideration transferred over the net assets recognized and represents the expected synergies of the combined company and assembled workforce. Goodwill has been allocated to the Codman Specialty Surgical segment, as shown in Note 7. Goodwill and Other Intangibles. Goodwill recognized as a result of this acquisition is non-deductible for income tax purposes.
Deferred Tax Liabilities
Deferred tax liabilities result from identifiable intangible assets’ fair value adjustments. These adjustments create excess book basis over tax basis which is tax-effected by the statutory tax rates of applicable jurisdictions.
5. DEBT
Amendment to the Seventh Amended and Restated Credit Agreement
On June 6, 2025, in response to the risks and uncertainties surrounding the Company’s future results of operations due to tariffs, the Company entered into the June 2025 Amendment of the Senior Credit Facility with a syndicate of lending banks with Bank of America, N.A., as Administrative Agent. The June 2025 Amendment did not increase the Company’s total indebtedness or extend the maturity date of the Senior Credit Facility.
Under the terms of the June 2025 Amendment, the Company’s Consolidated Total Leverage Ratio (defined, as of any date of determination, as the ratio of (a) Consolidated Funded Indebtedness as of such date (as defined in the Senior Credit Facility) less cash that is not subject to any restriction on the use or investment thereof to (b) Consolidated EBITDA (as defined in the Senior Credit Facility)) for the period of four consecutive fiscal quarters ending on such date was modified to the following:
| Fiscal Quarter Ending | Maximum Consolidated Total Leverage Ratio | |||||||
| June 30, 2025 through June 30, 2026 | ||||||||
| September 30, 2026 | ||||||||
| December 31, 2026 | ||||||||
| March 31, 2027 and the last day of each fiscal quarter thereafter | ||||||||
In addition to the foregoing, from the date of the June 2025 Amendment through the fiscal quarter ending December 31, 2026 (the “Covenant Relief Period”), the Amendment, among other things, also: (i) temporarily establishes, during the Covenant Relief Period, a revised applicable rate schedule; (ii) temporarily limits, during the Covenant Relief Period, the Company’s ability to make certain investments; (iii) temporarily restricts, during the Covenant Relief Period, the Company’s ability to incur incremental indebtedness under the Senior Credit Facility, create certain liens, make certain restricted payments and incur or guarantee indebtedness of excluded subsidiaries; and (iv) temporarily prohibits, during the Covenant Relief Period, the Company from selling, transferring or exclusively licensing material intellectual property to a subsidiary of the Company that is not a loan party under the Senior Credit Facility or designating any subsidiary that owns or exclusively licenses any material intellectual property as an excluded subsidiary under the Senior Credit Facility.
Borrowings under the Senior Credit Facility bear interest under the applicable rate schedule, at the Company’s option, at a rate equal to the following:
i.Term SOFR in effect from time to time plus 0.10 % plus the applicable rate (ranging from 1.00 % to 2.13 % during the Covenant Relief Period and from 1.00 % to 1.75 % thereafter), or
ii.The highest of:
1.the weighted average overnight Federal funds rate, as published by the Federal Reserve Bank of New York, plus 0.50 %;
2.the prime lending rate of Bank of America, N.A.; or
3.the one-month Term SOFR plus 1.00 %
The applicable rates are based on the Company’s Consolidated Total Leverage Ratio for the period of four consecutive fiscal quarters ending on such date.
The Company will pay an annual commitment fee (ranging from 0.15 % to 0.33 % during the Covenant Relief Period and from 0.15 % to 0.30 % thereafter), based on the Company’s Consolidated Total Leverage Ratio, on the amount available for borrowing under the revolving credit facility component of the Senior Credit Facility.
F-22
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
In connection with the June 2025 Amendment, the Company incurred $3.9 million in fees to creditors and third parties.
On March 24, 2023, the Company entered into the Senior Credit Facility with a syndicate of lending banks with Bank of America, N.A., as Administrative Agent. The Senior Credit Facility extended the maturity date to March 24, 2028, amended the contractual repayments of the term loan component, and amended the interest rate from LIBOR to SOFR-indexed interest.
The Company continues to have the aggregate principal amount of up to approximately $2.1 billion available to it through the following facilities: (i) a $775.0 million term loan facility, and (ii) a $1.3 billion revolving credit facility, which includes a $60.0 million sublimit for the issuance of standby letters of credit and a $60.0 million sublimit for swingline loans. The terms of the Senior Credit Facility limit the amount of dividends the Company may pay.
The Senior Credit Facility is collateralized by substantially all of the assets of the Company’s U.S. subsidiaries, excluding intangible assets. The Senior Credit Facility is subject to various financial and negative covenants and, at December 31, 2025, the Company was in compliance with all such covenants.
At December 31, 2025 and 2024 there was $1,045.0 million and $365.0 million, respectively, outstanding under the revolving credit facility component of the Senior Credit Facility. At December 31, 2025 and 2024, there was $726.6 million and $760.5 million outstanding under the term loan component of the Senior Credit Facility at a weighted average interest rate of 5.7 % and 6.0 %, respectively. As of December 31, 2025 and 2024 there was $38.8 million and $33.9 million, respectively, of the term loan component of the Senior Credit Facility classified as current on the condensed consolidated balance sheet.
The fair value of the revolving credit facility component and term loan component of the Senior Credit Facility at December 31, 2025 was $1,043.3 million and $725.5 million, respectively. The fair value was determined by using a discounted cash flow model based on current market interest rates available to the Company. These inputs are corroborated by observable market data for similar liabilities and therefore classified within Level 2 of the fair value hierarchy. Level 2 inputs represent inputs that are observable for the asset or liability, either directly or indirectly, and are other than active market observable inputs that reflect unadjusted quoted prices for identical assets or liabilities.
As of December 31, 2025 and 2024, letters of credit outstanding totaled $3.0 million and $1.7 million, respectively. There were no amounts drawn under the letters of credit outstanding as of December 31, 2025.
Contractual repayments of the term loan component of the Senior Credit Facility are due as follows:
| Year Ended December 31, 2025 | Principal Repayment | |||||||
| Dollars in thousands | ||||||||
| 2026 | $ | |||||||
| 2027 | ||||||||
| 2028 | ||||||||
| $ | ||||||||
Future interest payments on the term loan component of the Senior Credit Facility based on current interest rates are expected to approximate $40.5 million in 2026, $38.1 million in 2027 and $8.3 million in 2028. Interest is calculated on the term loan component of the Senior Credit Facility based on SOFR plus the certain amounts set forth in the Senior Credit Facility. As the revolving credit facility component of the Senior Credit Facility and Securitization Facility (defined below) can be repaid at any time, no interest has been included in the calculation.
Any outstanding borrowings on the revolving credit facility component of the Senior Credit Facility are due on March 24, 2028.
F-23
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Convertible Senior Notes
The Company’s 0.5 % Convertible Senior Notes due 2025 (the “2025 Notes”) issued in February 2020 pursuant to an indenture, dated as of February 7, 2020 (the “Original Indenture”), between the Company and Citibank, N.A., as trustee, matured on August 15, 2025. The 2025 Notes were settled upon maturity for $575.0 million in cash, excluding accrued interest, funded by borrowings on the revolving credit facility component of the Senior Credit Facility. No shares were issued to settle the 2025 Notes.
In connection with the issuance of the 2025 Notes, the Company entered into call transactions and warrant transactions, primarily with affiliates of the initial purchasers of the 2025 Notes (the “hedge participants”). The cost of the call transactions was $104.2 million for the 2025 Notes. The Company received $44.5 million of proceeds from the warrant transactions for the 2025 Notes. The call transactions involved purchasing call options from the hedge participants, and the warrant transactions involved selling call options to the hedge participants with a higher strike price than the purchased call options. The initial strike price of the call transactions was $73.67 , subject to anti-dilution adjustments substantially similar to those in the 2025 Notes. The initial strike price of the warrant transactions was $113.34 for the 2025 Notes, subject to customary anti-dilution adjustments. The call transactions entered into with the hedge participants expired in August 2025 and the warrant transactions entered into with the hedge participants expire ratably over the period from November 2025 through February 2026.
Securitization Facility
The Company maintains an accounts receivable securitization facility (the “Securitization Facility”) under which accounts receivable of certain domestic subsidiaries are sold on a non-recourse basis to a special purpose entity (“SPE”), which is a bankruptcy-remote, consolidated subsidiary of the Company. Accordingly, the assets of the SPE are not available to satisfy the obligations of the Company or any of its subsidiaries. From time to time, the SPE may finance such accounts receivable with a revolving loan facility secured by a pledge of such accounts receivable. The amount of outstanding borrowings on the Securitization Facility at any one time is limited to $150.0 million. The Securitization Facility Agreement (“Securitization Agreement”) governing the Securitization Facility contains certain covenants and termination events. An occurrence of an event of default or a termination event under this Securitization Agreement may give rise to the right of its counterparty to terminate this facility. As of December 31, 2025, the Company was in compliance with the covenants and none of the termination events had occurred and the Securitization Facility is classified as current borrowings under the securitization facility on the consolidated balance sheet, as it is due within one year.
On October 22, 2025, the Company entered into an amendment (the “October 2025 Amendment”) of the Securitization Facility which modified a financial covenant related to the maximum Delinquency Ratio (as defined in the Securitization Agreement) over the remaining term of the Securitization Agreement as amended by the December 2023 Amendment. The October 2025 Amendment did not affect the borrowing capacity or extend the maturity date of the Securitization Facility. The Company incurred approximately $0.1 million of new issuance costs associated with the October 2025 Amendment which will be amortized over the remaining term of the Securitization Agreement as amended by the December 2023 Amendment.
On December 15, 2023, the Company entered into an amendment (the “December 2023 Amendment”) of the Securitization Facility which extended the maturity date from May 28, 2024 to December 15, 2026. The Company incurred approximately $0.3 million of new issuance costs associated with the December 2023 Amendment which will be amortized over 3 years, the length of the Securitization Agreement as amended by the December 2023 Amendment. Due to the increase in borrowing capacity, the remaining $0.1 million of unamortized costs from the previous agreement will also be amortized over the length of the amended agreement, three years . In addition, on April 17, 2023 the Company entered into an amendment (the “April 2023 Amendment”) of the Securitization Facility and amended the interest rate from LIBOR to SOFR-indexed rate. The December 2023 Amendment and April 2023 Amendment did not increase the Company’s total indebtedness.
At December 31, 2025 and 2024, the Company had $87.8 million and $108.1 million, respectively, of outstanding borrowings under its Securitization Facility with an interest rate of 4.8 % and 5.4 %, respectively. The fair value of the outstanding borrowing of the Securitization Facility at December 31, 2025 was $86.7 million. These fair values were determined by using a discounted cash flow model based on current market interest rates available to the Company. These inputs are corroborated by observable market data for similar liabilities and therefore classified within Level 2 of the fair value hierarchy. Level 2 inputs represent inputs that are observable for the asset or liability, either directly or indirectly, and are other than active market observable inputs that reflect unadjusted quoted prices for identical assets or liabilities.
Debt Issuance Costs
Debt issuance costs associated with the Senior Credit Facility (other than the revolving credit facility component) and the 2025 Notes are presented as a reduction to the carrying value of the related debt. Debt issuance costs associated with the revolving credit facility component of the Senior Credit Facility are capitalized within other long-term assets on the consolidated balance sheet.
F-24
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Estimated Fair Value of Debt Measurements
The carrying amounts and the estimated fair values of debt as of December 31, 2025 and 2024 are as follows:
| December 31, 2025 | December 31, 2024 | ||||||||||||||||
| Fair Value Measurement | Carrying Value | Estimated Fair Value | Carrying Value | Estimated Fair Value | |||||||||||||
| Dollars in thousands | |||||||||||||||||
| Senior credit facility - term loan | Level 2 | $ | $ | $ | $ | ||||||||||||
| Senior credit facility - revolving component | Level 2 | ||||||||||||||||
| 2025 Notes | Level 1 | ||||||||||||||||
| Securitization | Level 2 | ||||||||||||||||
| Subtotal | $ | $ | $ | $ | |||||||||||||
| Debt issuance costs | ( | ( | |||||||||||||||
| Total debt | $ | $ | $ | $ | |||||||||||||
6. DERIVATIVE INSTRUMENTS
Interest Rate Hedging
The Company’s interest rate risk relates to U.S. dollar denominated variable interest rate borrowings. The Company uses interest rate swap derivative instruments to manage earnings and cash flow exposure resulting from changes in interest rates. These interest rate swaps apply a fixed interest rate on a portion of the Company’s expected SOFR-indexed borrowings. Additionally, the Company entered into a basis swap where the Company receives Term SOFR and pays daily compounded SOFR to convert the portfolio of swaps from daily compounded SOFR to Term SOFR.
The Company held the following interest rate swaps as of December 31, 2025 and 2024 (dollar amounts in thousands):
| December 31, 2025 | December 31, 2024 | December 31, 2025 | December 31, 2024 | |||||||||||||||||||||||||||||||||||||||||||||||
| Hedged Item | Notional Amount | Designation Date | Effective Date | Termination Date | Fixed Interest Rate | Estimated Fair Value | ||||||||||||||||||||||||||||||||||||||||||||
| Asset (Liability) | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | October 10, 2018 | July 1, 2020 | June 30, 2025 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | October 10, 2018 | July 1, 2020 | June 30, 2025 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | October 10, 2018 | July 1, 2020 | June 30, 2025 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | December 18, 2018 | December 30, 2022 | December 31, 2027 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | December 18, 2018 | December 30, 2022 | December 31, 2027 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | December 15, 2020 | July 31, 2025 | December 31, 2027 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| 1-month Term SOFR Loan | December 15, 2020 | July 1, 2025 | December 31, 2027 | % | ||||||||||||||||||||||||||||||||||||||||||||||
Basis Swap (1) | March 31, 2023 | March 24, 2023 | December 31, 2027 | N/A | ( | 0 | ( | |||||||||||||||||||||||||||||||||||||||||||
| $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||
(1) The notional of the basis swap amortizes to match the total notional of the interest rate swap portfolio over time | ||||||||||||||||||||||||||||||||||||||||||||||||||
The interest rate swaps were carried on the consolidated balance sheet at fair value and changes in the fair values were recorded as unrealized gains or losses in accumulated other comprehensive income (“AOCI”). For the years ended December 31, 2025 and 2024, the Company recorded a loss of $(8.9 ) million and a gain of $22.7 million, respectively, in AOCI related to the change in fair value of the interest rate swaps.
F-25
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
For the years ended December 31, 2025 and 2024, the Company recorded gains of $12.6 million and $17.5 million, respectively, in the consolidated statements of operations related to the interest rate differential of the interest rate swaps. The estimated gain that is expected to be reclassified to interest expense from AOCI as of December 31, 2025 within the next twelve months is $15.0 million.
The Company has designated these derivative instruments as cash flow hedges. The Company assesses the effectiveness of these derivative instruments and has recorded the changes in the fair value of the derivative instrument designated as a cash flow hedge as unrealized gains or losses in AOCI, net of tax, until the hedged item affected earnings, at which point any gain or loss was reclassified to earnings. If the hedged cash flow does not occur, or if it becomes probable that it will not occur, the Company will reclassify the remaining amount of any gain or loss on the related cash flow hedge recorded in AOCI to interest expense at that time.
Foreign Currency Hedging
From time to time, the Company enters into foreign currency hedge contracts intended to protect the U.S. dollar value of certain forecasted foreign currency denominated transactions. The Company assesses the effectiveness of the contracts that are designated as hedging instruments. The changes in fair value of foreign currency cash flow hedges are recorded in AOCI, net of tax. Those amounts are subsequently reclassified to earnings from AOCI as impacted by the hedged item when the hedged item affects earnings. If the hedged forecasted transaction does not occur or if it becomes probable that it will not occur, the Company will reclassify the amount of any gain or loss on the related cash flow hedge to earnings at that time. For contracts not designated as hedging instruments, the changes in fair value of the contracts are recognized in other income, net in the consolidated statements of operation, along with the offsetting foreign currency gain or loss on the underlying assets or liabilities.
The success of the Company’s hedging anticipated currency exchange gains or losses to the extent that there are differences between forecasted and actual activities during periods of currency volatility. In addition, changes in currency exchange rates related to any unhedged transactions may affect earnings and cash flows.
Cross-Currency Rate Swaps
The objective of these cross-currency swaps is to reduce volatility of earnings and cash flows associated with changes in the foreign currency exchange rate on intercompany loans. Under the terms of these contracts, which have been designated as cash flow hedges, the Company will make interest payments in Swiss francs (“CHFs”) and receive interest in U.S. dollars. Upon the maturity of these contracts, the Company will pay the principal amount of the loans in CHFs and receive U.S. dollars from the counterparties.
In December 2020, the Company entered into cross-currency swap agreements to convert a notional amount of $471.6 million equivalent to 420.1 million of a CHF-denominated intercompany loan into U.S. dollars. The CHF-denominated intercompany loan was the result of an intra-entity transfer of certain intellectual property rights to a subsidiary in Switzerland completed during the fourth quarter of 2020. The intercompany loan requires quarterly principal payments of CHF 5.8 million plus accrued interest. As a result, the aggregate notional amount of the related cross-currency swaps will decrease by a corresponding amount. In February 2025, the Company amended the CHF-denominated intercompany loan to extend the maturity to December 2030. Concurrently, the Company amended the cross-currency swap agreement, with a notional amount of $368.4 million, equivalent to CHF 328.1 million, to extend the maturity to December 2030.
In November 2025, the Company entered into cross-currency swap agreements to convert an aggregate notional amount of $170.0 million equivalent to 137.6 million of two CHF-denominated intercompany loans into U.S. dollars. The CHF-denominated intercompany loans were the result of an intra-entity transfer of certain intellectual property rights to a subsidiary in Switzerland completed during the fourth quarter of 2025. The intercompany loan of CHF 80.9 million which matures in September 2028 requires quarterly interest payments and annual principal payments of CHF 8.1 million in September 2026 and September 2027; the intercompany loan of CHF 56.6 million which matures in September 2030 requires quarterly interest payments and a principal payment of CHF 8.1 million in September 2029. As the principal outstanding decreases on the intercompany loans, the aggregate notional amount of the related cross-currency swaps will decrease by a corresponding amount.
F-26
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company held the following cross-currency rate swaps as of December 31, 2025 and 2024 (dollar amounts in thousands):
| December 31, 2025 | December 31, 2024 | December 31, 2025 | December 31, 2024 | ||||||||||||||||||||||||||||||||||||||
| Effective Date | Termination Date | Fixed Rate | Aggregate Notional Amount | Fair Value Asset (Liability) | |||||||||||||||||||||||||||||||||||||
| Pay CHF | December 21, 2020 | December 22, 2025 | CHF | $ | $ | ( | |||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||
| Pay CHF | February 20, 2025 | December 20, 2030 | CHF | $ | ( | ||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||
| Pay CHF | November 10, 2025 | September 30, 2030 | CHF | $ | ( | ||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||
| Pay CHF | November 10, 2025 | September 29, 2028 | CHF | $ | ( | ||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||
| Total | $ | ( | $ | ( | |||||||||||||||||||||||||||||||||||||
The cross-currency swaps designated as cash flow hedges are carried on the consolidated balance sheet at fair value, and changes in the fair values are recorded as unrealized gains or losses in AOCI. For the years ended December 31, 2025 and 2024 the Company recorded a loss of $(49.0 ) million and a gain of $41.0 million, respectively, in AOCI related to change in fair value of the cross-currency swaps.
For the years ended December 31, 2025 and 2024, the Company recorded a loss of $(51.3 ) million and a gain of $32.2 million, respectively, in other income, net related to the change in fair value related to the foreign currency rate translation to offset the gains and losses, respectively, recognized on the intercompany loans.
For the years ended December 31, 2025 and 2024, the Company recorded gains of $8.7 million and $4.7 million, respectively, respectively, in other income, net included in the consolidated statements of operations related to the interest rate differential of the cross-currency swaps.
The estimated loss that is expected to be reclassified to other income (expense), net from AOCI as of December 31, 2025 within the next twelve months is $10.0 million.
Net Investment Hedges
The Company manages certain foreign exchange risks through a variety of strategies, including hedging. The Company is exposed to foreign exchange risk from its international operations through foreign currency purchases, net investments in foreign subsidiaries, and foreign currency assets and liabilities created in the normal course of business.
In February 2025, the Company entered into a cross-currency swap agreement with a notional amount of CHF 67.8 million equivalent to $75.0 million, where the Company agreed with third-parties to sell CHF in exchange for U.S. dollars at a specified rate at the maturity of the contract. The new cross-currency swap agreement was designated as a net investment hedge to partially offset the effects of foreign currency on foreign subsidiaries.
In July 2025, the Company entered into a cross-currency swap agreement with a notional amount of CHF 59.7 million, equivalent to $75.0 million, where the Company agreed with third-parties to sell CHF in exchange for U.S. dollars at a specified rate at the maturity of the contract. The new cross-currency swap agreement was designated as a net investment hedge to partially offset the effects of foreign currency on foreign subsidiaries.
F-27
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company held the following cross-currency rate swaps designated as net investment hedges as of December 31, 2025 and 2024 (dollar amounts in thousands):
| December 31, 2025 | December 31, 2024 | December 31, 2025 | December 31, 2024 | ||||||||||||||||||||||||||||||||||||||||||||
| Effective Date | Termination Date | Fixed Rate | Aggregate Notional Amount | Fair Value Asset (Liability) | |||||||||||||||||||||||||||||||||||||||||||
| Pay EUR | October 3, 2018 | September 30, 2025 | EUR | $ | |||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Pay CHF | May 26, 2022 | December 16, 2028 | CHF | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Pay CHF | November 17, 2023 | December 17, 2029 | CHF | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Pay CHF | May 6, 2024 | December 18, 2030 | CHF | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Pay CHF | February 21, 2025 | December 15, 2031 | CHF | ( | |||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Pay CHF | July 25, 2025 | December 15, 2032 | CHF | ( | |||||||||||||||||||||||||||||||||||||||||||
| Receive U.S.$ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Total | $ | ( | $ | ( | |||||||||||||||||||||||||||||||||||||||||||
The cross-currency swaps were carried on the consolidated balance sheet at fair value and changes in the fair values were recorded as unrealized gains or losses in AOCI. For the years ended December 31, 2025 and 2024, the Company recorded a loss of $(49.5 ) million and a gain of $24.5 million, respectively, in AOCI related to the change in fair value of the cross-currency swaps.
For the years ended December 31, 2025 and 2024, the Company recorded gains of $12.5 million and $10.1 million, respectively, in interest income included in the consolidated statements of operations related to the interest rate differential of the cross-currency swaps.
The estimated gain that is expected to be reclassified to interest income from AOCI as of December 31, 2025 within the next twelve months is $11.4 million.
Foreign Currency Forward Contracts
The Company has entered into forward contracts designated as cash flow hedges for forecasted purchases in foreign currencies, primarily CHF-denominated intercompany purchases. To the extent these forward contracts meet hedge accounting criteria, changes in their fair value are included in accumulated other comprehensive loss. These changes in fair value will be recognized into earnings as a component of cost of sales when the forecasted-transaction occurs. These contracts typically settle at various dates within twelve months of execution. As of December 31, 2025 the notional amount of foreign currency forward contracts was CHF 10.4 million.
For the year ended December 31, 2025 and 2024 the Company recorded a gain of $1.9 million and a loss of $(1.1 ) million, respectively, in AOCI related to the change in fair value of the foreign currency forward contracts.
For the year ended December 31, 2025 and 2024 the Company recorded a gain of $0.6 million and a loss of $(0.4 ) million, respectively, in other income and cost of goods sold included in the consolidated statements of operations related to the amortization of forward points and rate translation on the foreign currency forward contracts.
On February 19, 2026, the Company entered into foreign currency forwards with a notional amount of 8.0 million CHF to mitigate the exchange rate risk of CHF-denominated intercompany purchases. These contracts settle at various dates within twelve months of execution.
F-28
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Counterparty Credit Risk
The Company manages its concentration of counterparty credit risk on its derivative instruments by limiting acceptable counterparties to a group of major financial institutions with investment grade credit ratings, and by actively monitoring their credit ratings and outstanding positions on an ongoing basis. Therefore, the Company considers the credit risk of the counterparties to be low. Furthermore, none of the Company’s derivative transactions are subject to collateral or other security arrangements, and none contain provisions that depend upon the Company’s credit ratings from any credit rating agency.
Fair Value of Derivative Instruments
The Company has classified all of its derivative instruments within Level 2 of the fair value hierarchy because observable inputs are available for the derivative instruments. The fair values of the interest rate swaps and cross-currency swaps were developed using a market approach based on publicly available market yield curves and the terms of the swap. The Company performs ongoing assessments of counterparty credit risk.
F-29
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Effects of Derivative Instruments on Financial Position and Results of Operations
The following table summarizes the fair value for derivatives designated as hedging instruments in the condensed consolidated balance sheets as of December 31, 2025 and 2024:
| Fair Value as of December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
Location on Balance Sheet (1): | |||||||||||
| Derivatives designated as hedges — Assets: | |||||||||||
| Prepaid expenses and other current assets | |||||||||||
| Cash Flow Hedges | |||||||||||
Interest rate swap | $ | $ | |||||||||
| Cross-currency swap | |||||||||||
| Foreign Currency Forward Contracts | |||||||||||
| Net Investment Hedges | |||||||||||
| Cross-currency swap | |||||||||||
| Other assets | |||||||||||
| Cash Flow Hedges | |||||||||||
Interest rate swap | |||||||||||
| Total derivatives designated as hedges — Assets | $ | $ | |||||||||
| Derivatives designated as hedges — Liabilities | |||||||||||
| Accrued expenses and other current liabilities | |||||||||||
| Cash Flow Hedges | |||||||||||
Interest rate swap | $ | $ | |||||||||
| Cross-currency swap | |||||||||||
| Foreign Currency Forward Contracts | |||||||||||
| Net Investment Hedges | |||||||||||
| Cross-currency swap | |||||||||||
| Other liabilities | |||||||||||
| Cash Flow Hedges | |||||||||||
Interest rate swap | |||||||||||
| Cross-currency swap | |||||||||||
| Net Investment Hedges | |||||||||||
| Cross-currency swap | |||||||||||
| Total derivatives designated as hedges — Liabilities | $ | $ | |||||||||
(1)The Company classifies derivative assets and liabilities as current based on the cash flows expected to be incurred within the following 12 months.
The following presents the effect of derivative instruments designated as cash flow hedges and net investment hedges on the accompanying condensed consolidated statement of operations during the years ended December 31, 2025 and 2024:
F-30
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
| Dollars in thousands | Balance in AOCI Beginning of Year | Amount of Gain (Loss) Recognized in AOCI | Amount of Gain (Loss) Reclassified from AOCI into Earnings | Balance in AOCI End of Year | Location in Statements of Operations | ||||||||||||||||||||||||
| Year Ended December 31, 2025 | |||||||||||||||||||||||||||||
| Cash Flow Hedges | |||||||||||||||||||||||||||||
| Interest rate swap | $ | $ | ( | $ | $ | Interest expense | |||||||||||||||||||||||
| Cross-currency swap | ( | ( | ( | ( | Other income, net | ||||||||||||||||||||||||
| Forward Currency Forward Contracts | ( | Cost of sales and other income, net | |||||||||||||||||||||||||||
| Net Investment Hedges | — | ||||||||||||||||||||||||||||
| Cross-currency swap | ( | ( | ( | Interest income | |||||||||||||||||||||||||
| $ | $ | ( | $ | ( | $ | ( | |||||||||||||||||||||||
| Year Ended December 31, 2024 | |||||||||||||||||||||||||||||
| Cash Flow Hedges | |||||||||||||||||||||||||||||
| Interest rate swap | $ | $ | $ | $ | Interest expense | ||||||||||||||||||||||||
| Cross-currency swap | ( | ( | Other income, net | ||||||||||||||||||||||||||
| Forward Currency Forward Contracts | ( | ( | ( | Cost of sales and other income, net | |||||||||||||||||||||||||
| Net Investment Hedges | |||||||||||||||||||||||||||||
| Cross-currency swap | ( | ( | Interest income | ||||||||||||||||||||||||||
| $ | ( | $ | $ | $ | |||||||||||||||||||||||||
Derivative Instruments Not Designated as Hedges:
From time to time, the Company enters into foreign currency forward contracts to mitigate risk from the fluctuations in foreign currency exchange rates associated with intercompany balances in Chinese yuan (“CNH”), CHF, and EUR. These contracts typically settle at various dates within twelve months of execution. As of December 31, 2025, the notional amounts totaled CNH 90.0 million and CHF 14.0 million, equivalent to $12.7 million, and $17.8 million, respectively.
In 2021, the Company entered into a foreign currency swap, with a notional amount of JPY 800.0 million, equivalent to $7.3 million, to mitigate the risk from fluctuations in foreign currency exchange rates associated with an intercompany loan denominated in Japanese yen. In a foreign currency swap transaction, the Company agrees with another party to exchange, at specified intervals, the difference between one currency and another currency at a fixed exchange rate, generally set at inception, calculated by reference to an agreed upon notional amount. The notional amount of each currency is exchanged at the inception and termination of the currency swap by each party. The Company subsequently paid down a portion of this swap, bringing the notional amount down to JPY 400.0 million, equivalent to $3.6 million as of December 31, 2025.
The fair value of the derivative instruments not designated as hedges was $1.0 million and $1.7 million as of December 31, 2025 and 2024, respectively.
The following table summarizes the (gains) and losses on derivative instruments not designated as hedges on the condensed consolidated statements of income, which was included in other income:
| Dollars in thousands | December 31, | |||||||||||||
| 2025 | 2024 | |||||||||||||
| Foreign currency forward contracts | $ | ( | $ | |||||||||||
| Foreign currency swaps | ( | |||||||||||||
| Total | $ | ( | $ | |||||||||||
7. GOODWILL AND OTHER INTANGIBLE ASSETS
Goodwill
In accordance with FASB Topic 350, Intangibles—Goodwill and Other (“ASC 350”), goodwill is not subject to amortization but is tested for impairment at the reporting unit level annually in the third quarter. Additionally, the Company may perform
F-31
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
interim tests of goodwill for impairment if an event occurs or circumstances change that could potentially reduce the fair value of a reporting unit below its carrying amount. The carrying value of each reporting unit is determined by assigning the assets and liabilities, including the existing goodwill and intangible assets, to those reporting units. An impairment loss is recognized when the reporting unit’s carrying amount exceeds its estimated fair value.
The Company tests for impairment by either performing a qualitative evaluation or a quantitative test. The qualitative evaluation is an assessment of factors, including reporting unit specific operating results as well as industry, market and general economic conditions, to determine whether it is more likely than not that the fair values of a reporting unit is less than its carrying amount, including goodwill. The Company may elect to bypass this qualitative evaluation for some or all of its reporting units and perform a quantitative test. The quantitative test uses a combination of both an income approach and a market approach to determine the fair value of the reporting unit. The income approach utilizes the estimated discounted cash flows for the reporting unit, while the market approach utilizes comparable publicly-traded companies’ revenue and earnings before interest, taxes, depreciation, and amortization (“EBITDA”) multiples. Estimates and assumptions used in the income approach to calculate projected future discounted cash flows included revenue growth rates, cost of sales, terminal growth rates, and a discount rate for each reporting unit. Discount rates are determined using a weighted average cost of capital for risk factors specific to each reporting unit and other market and industry data. The assumptions used are inherently subject to uncertainty and slight changes in these assumptions could have a significant impact on the concluded value. The estimates and assumptions applied represent a Level 3 measurement in the fair value hierarchy. Level 3 inputs are supported by limited or no market activity and reflect the Company’s assumptions in measuring fair value.
The key assumptions impacting the valuation included the following:
•The reporting unit’s financial projections, including revenue growth rates and cost of sales, which are based on management’s assessment of regional and macroeconomic variables, industry trends and market opportunities, and the Company’s strategic objectives and future growth plans.
•The projected terminal value for the reporting unit, which represents the present value of projected cash flows beyond the last period in the discounted cash flow analysis. The terminal value reflects the Company’s assumptions related to long-term growth rates and profitability, which are based on several factors, including local and macroeconomic variables, market opportunities, and future growth plans.
•The discount rate used to measure the present value of the projected future cash flows is set using a weighted-average cost of capital method that considers market and industry data as well as the Company’s specific risk factors that are likely to be considered by a market participant. The weighted-average cost of capital is the Company’s estimate of the overall after-tax rate of return required by equity and debt holders of a business enterprise.
During the second quarter of 2025, the Company performed a quantitative assessment of its Tissue Technologies, Neurosurgery, and Instruments and ENT reporting units in accordance with ASC 350 due to the decrease in the price per share of the Company’s common stock related to a number of factors including recent tariff changes that have created broad economic uncertainty and the impact of quality, operational, and supply issues. The Company recognized an aggregate charge of $511.4 million in goodwill impairment expense in the consolidated statement of operations in the second quarter of 2025. The quantitative test for the Tissue Technologies reporting unit utilized a terminal growth rate of 2.5 % and a discount rate of 13.5 % in the income approach. The Company determined, after performing the quantitative analysis, that the fair value of the Tissue Technologies reporting unit was less than its carrying amount and recognized an impairment of $123.3 million. The quantitative test for the Neurosurgery reporting unit utilized a terminal growth rate of 2.5 % and a discount rate of 13.5 % in the income approach. The Company determined, after performing the quantitative analysis, that the fair value of the Neurosurgery reporting unit was less than its carrying amount and recognized an impairment of $249.0 million. The quantitative test for the Instruments and ENT reporting unit utilized a terminal growth rate of 2.5 % and a discount rate of 12.0 % in the income approach. The Company determined, after performing the quantitative analysis, that the fair value of the Instruments and ENT reporting unit was less than its carrying amount and recognized an impairment of $139.1 million.
In the third quarter of 2025, the Company performed its annual test of its reporting units for impairment and completed a qualitative evaluation of its Tissue Technologies and Neurosurgery reporting units. The Instruments and ENT reporting unit had been deemed fully-impaired during the second quarter of 2025 and was excluded from this evaluation. After performing the qualitative analysis, the Company concluded that it was more likely than not that the fair values of the Tissue Technologies and Neurosurgery reporting units were greater than their carrying amounts. Therefore, it was not necessary to perform a quantitative impairment test.
The Company did not recognize any goodwill impairment charges during 2024.
F-32
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Changes in the carrying amount of goodwill in 2025 and 2024 were as follows:
| Dollars in thousands | Codman Specialty Surgical | Tissue Technologies | Total | ||||||||||||||
Goodwill at January 1, 2024 | |||||||||||||||||
| Acclarent Acquisition | |||||||||||||||||
| Foreign currency translation | ( | ( | ( | ||||||||||||||
Balance at December 31, 2024 | $ | $ | $ | ||||||||||||||
| Impairment | ( | ( | ( | ||||||||||||||
| Foreign currency translation | |||||||||||||||||
Balance at December 31, 2025 | $ | $ | $ | ||||||||||||||
Other Intangible Assets
The components of the Company’s identifiable intangible assets were as follows:
| December 31, 2025 | |||||||||||||||||||||||
| Dollars in thousands | Weighted Average Life | Cost | Accumulated Amortization | Net | |||||||||||||||||||
| Completed technology | $ | $ | ( | $ | |||||||||||||||||||
| Customer relationships | ( | ||||||||||||||||||||||
| Trademarks/brand names | ( | ||||||||||||||||||||||
| Codman trade name | Indefinite | — | |||||||||||||||||||||
| Supplier relationships | ( | ||||||||||||||||||||||
| All other | ( | ||||||||||||||||||||||
| $ | $ | ( | $ | ||||||||||||||||||||
| December 31, 2024 | |||||||||||||||||||||||
| Dollars in thousands | Weighted Average Life | Cost | Accumulated Amortization | Net | |||||||||||||||||||
| Completed technology | $ | $ | ( | $ | |||||||||||||||||||
| Customer relationships | ( | ||||||||||||||||||||||
| Trademarks/brand names | ( | ||||||||||||||||||||||
| Codman trade name | Indefinite | — | |||||||||||||||||||||
| Supplier relationships | ( | ||||||||||||||||||||||
| All other | ( | ||||||||||||||||||||||
| $ | $ | ( | $ | ||||||||||||||||||||
Intangible Assets with Indefinite Lives
The Company does not amortize intangible assets with indefinite lives but tests its intangible assets with indefinite lives for impairment annually in the third quarter in accordance with ASC 350. Additionally, the Company performs interim tests of its intangible assets with indefinite lives for impairment if an event occurs or circumstances change that could potentially reduce the fair value of an indefinite lived intangible asset below its carrying amount. The Company tests for impairment by either performing a qualitative evaluation or a quantitative test. The qualitative evaluation is an assessment of factors, including specific operating results as well as industry, market and general economic conditions, to determine whether it is more likely than not that the fair values of the intangible asset is less than its carrying amount. The Company may elect to bypass this qualitative evaluation and perform a quantitative test.
F-33
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
During the second quarter of 2025 the Company performed a quantitative assessment of its Codman tradename intangible asset in accordance with ASC 350 due to the decrease in the price per share of the Company’s common stock related to a number of factors including recent tariff changes that have created broad economic uncertainty and the impact of quality, operational, and supply issues. In performing this test, the Company utilized a discount rate of 14.5 %. The assumptions used in evaluating the Codman tradename for impairment are subject to change and are tracked against historical results by management. Based on the results of the quantitative test, the Company recorded no impairment to the Codman tradename intangible asset.
In the third quarter of 2025, the Company performed its annual test of its intangible assets with indefinite lives for impairment and completed a qualitative evaluation of its Codman tradename intangible asset. After performing the qualitative analysis, the Company concluded that it was more likely than not that the fair value of the Codman tradename intangible asset was greater than its carrying amount. Therefore, it was not necessary to perform a quantitative impairment test.
The Company did not recognize any impairment charges on its intangible assets with indefinite lives during 2024.
Intangible Assets with Definite Lives
Developed technologies and other definite-lived intangible assets are amortized over their estimated useful lives either using the straight-line method or, if reliably determinable, based on the pattern of which the economic benefit of the asset is expected to be utilized. Definite-lived intangible assets are periodically evaluated for impairment in accordance with FASB Topic 360, Property, Plant and Equipment (“ASC 360”) whenever events or changes in circumstances indicate that a definite-lived intangible asset’s carrying value may not be recoverable.
Total amortization of intangible assets (including amounts reported in cost of product revenues for technology-based intangible assets) in 2025 was $107.1 million compared to $105.2 million in 2024. Of this amount, $92.1 million and $84.0 million, respectively, was related to amortization of technology based intangibles and included in cost of goods sold.
Based on quarter-end exchange rates, amortization expense (including amounts reported in cost of goods sold) is expected to be approximately $107.6 million in 2026, $106.6 million in 2027, $103.0 million in 2028, $97.7 million in 2029, $91.6 million in 2030 and $447.7 million thereafter.
In the first quarter of 2024, due to third-party audit findings and an update to the estimated timeframe to resume the commercial distribution of products manufactured in the Company’s manufacturing facility located in Boston, Massachusetts (the “Boston facility”), the Company elected to perform quantitative impairment testing on certain definite-lived intangible assets including completed technology and customer relationships in accordance with ASC 360. The Company recorded an impairment charge related to the definite-lived intangible asset associated with the customer relationships of $7.1 million in intangible asset amortization in the consolidated statement of operations. With respect to the definite-lived intangible assets associated with the completed technology of SurgiMend® and PriMatrix®, the Company determined that the carrying amount of these definite-lived intangible assets were recoverable and, therefore, the intangible assets were not deemed to be impaired. In the second quarter of 2024, the Company approved a plan to transition the manufacturing of SurgiMend and PriMatrix from the Boston facility to the Company’s manufacturing facility in Braintree, Massachusetts (the “Braintree facility”). The Company considered the impact to the update to the estimated timeframe to resume the commercial distribution of products manufactured in the Boston facility on the assumptions used in the quantitative assessment of the definite-lived intangible assets completed in the first quarter of 2024, which did not require further evaluation for impairment. The carrying values of SurgiMend and PriMatrix are $25.6 million and $20.0 million, respectively, as of December 31, 2025.
Potential Impact of Risks and Uncertainties on Management’s Estimates
F-34
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
8. TREASURY STOCK
As of December 31, 2025 and 2024, the Company held 14.4 million and 14.4 million shares of treasury stock outstanding with a cost of $689.2 million and $691.4 million, respectively, at a weighted average cost per share of $47.86
In 2024, the Company entered into a $50 million accelerated share repurchase (“the 2024 ASR”) and received 1.3 million shares of common stock at inception of the 2024 ASR, which represented approximately 70 % of the expected total shares under the 2024 ASR. The early exercise provision was exercised by the 2024 ASR counterparty. The Company received an additional 0.4 million shares determined using the volume-weighted average price of the Company’s common stock during the term of the 2024 ASR.
On July 18, 2023, the Board of Directors authorized a $225.0 million share repurchase program. On December 31, 2025, the Company’s share repurchase program expired. At the time of expiration, approximately $50.0 million remained available for repurchase. The Company did not adopt a new share repurchase program upon the expiration, and no shares were repurchased during the year ended December 31, 2025.
9. STOCK-BASED COMPENSATION
Stock-based compensation expense - all related to employees and members of the Board of Directors - recognized under the authoritative guidance was as follows:
| Years Ended December 31, | |||||||||||||||||
| Dollars in thousands | 2025 | 2024 | 2023 | ||||||||||||||
| Cost of goods sold | $ | $ | $ | ||||||||||||||
| Research and development | |||||||||||||||||
| Selling, general and administrative | |||||||||||||||||
| Total stock-based compensation expense | $ | $ | $ | ||||||||||||||
| Total estimated tax benefit related to stock-based compensation expense | |||||||||||||||||
| Net effect on net income | $ | $ | $ | ||||||||||||||
EQUITY AWARD PLANS
As of December 31, 2025, the Company had stock options, restricted stock awards, performance stock awards, contract stock awards and restricted stock unit awards outstanding under the Integra LifeSciences Holdings Corporation Fifth Amended and Restated 2003 Equity Incentive Plan (the “2003 Plan”).
In May 2010 and May 2017, the stockholders of the Company approved amendments to the 2003 Plan to increase by 3.5 million and 1.7 million, respectively, the number of shares of common stock that may be issued under the 2003 Plan. The Company has reserved 14.7 million shares under the 2003 Plan. The 2003 Plan permits the Company to grant incentive and non-qualified stock options, stock appreciation rights, restricted stock, contract stock, performance stock, or dividend equivalent rights to designated directors, officers, employees and associates of the Company.
Stock options issued under the 2003 Plan become exercisable over specified periods, generally within four years from the date of grant for officers and employees, and within one year from the date of the grant for members of the Board of Directors. The awards generally expire eight years from the grant date for employees and from to ten years for directors and certain executive officers, except in certain instances that result in accelerated vesting due to death, disability, retirement age or change in control provisions within their grant agreements. Restricted stock issued under the 2003 Plan vests ratably over specified periods, generally three years after the date of grant. The vesting of performance stock issued under the 2003 Plan is subject to service and performance conditions.
Stock Options
The Company values stock option grants using the binomial distribution model. Management believes that the binomial distribution model is preferable to the Black-Scholes model because it is a more flexible model that gives consideration to the impact of non-transferability and vesting provisions in valuing employee stock options.
In determining the value of stock options granted, the Company considered that it has never paid cash dividends and does not currently intend to pay cash dividends, and thus has assumed a 0.0 % dividend yield. Expected volatilities are based on the historical volatility of the Company’s stock price. The expected life of stock options is estimated based on historical data on exercise of stock options, post-vesting forfeitures and other factors to estimate the expected term of the stock options granted. The risk-free interest rates are derived from the U.S. Treasury yield curve in effect on the date of grant for instruments with a remaining term similar to the expected life of the options. The Company accounts for forfeitures as they occur.
F-35
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company granted stock options at two separate dates during the year ended December 31, 2025 and applied a range of assumptions for the volatility and risk free interest rate employed in the weighted average methodology in the calculation of option fair value. For the years ended December 31, 2024 and December 31, 2023, all assumptions employed a weighted average methodology in the calculation of option fair value. The following table outlines these assumptions:
| Years Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Dividend yield | |||||||||||||||||
| Expected volatility | |||||||||||||||||
| Risk free interest rate | |||||||||||||||||
| Expected life of option from grant date | |||||||||||||||||
Weighted average grant date fair value of options granted | $ | $ | $ | ||||||||||||||
The following table summarizes the Company’s stock option activity:
| Shares | Weighted Average Exercise Price | Weighted Average Contractual Term in Years | Aggregate Intrinsic Value | ||||||||||||||||||||
| Stock Options | (In thousands) | (In thousands) | |||||||||||||||||||||
Outstanding at January 1, 2025 | $ | $ | |||||||||||||||||||||
| Granted | — | — | |||||||||||||||||||||
| Exercised | — | — | |||||||||||||||||||||
| Forfeited or Expired | ( | — | — | ||||||||||||||||||||
Outstanding at December 31, 2025 | $ | $ | |||||||||||||||||||||
Exercisable at December 31, 2025 | $ | $ | |||||||||||||||||||||
The Company recognized $1.7 million, $2.2 million, and $1.4 million in expense related to stock options during the years ended December 31, 2025, 2024, and 2023, respectively. There were no options exercised for the year ended December 31, 2025. The intrinsic value of options exercised for the years ended December 31, 2024, and 2023 were $0.7 million, and $1.8 million, respectively. Cash received from option exercises and employee stock purchase plan was $1.0 million, $6.4 million, and $4.3 million, for the years ended December 31, 2025, 2024, and 2023, respectively. For the years ended December 31, 2025 and 2024, the realized tax expense related to the exercise of stock options was $0.8 million and $0.5 million, respectively. A realized tax benefit $0.1 million was recognized for the year ended December 31, 2023.
As of December 31, 2025, there was approximately $4.5 million of total unrecognized compensation costs related to unvested stock options. These costs are expected to be recognized over a weighted-average period of approximately three years .
F-36
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Awards of Restricted Stock, Performance Stock and Contract Stock
The following table summarizes the Company’s awards of restricted stock, performance stock and contract stock for the year ended December 31, 2025:
| Restricted Stock Awards | Performance Stock and Contract Stock Awards | ||||||||||||||||||||||
| Shares | Weighted Average Grant Date Fair Value Per Share | Shares | Weighted Average Grant Date Fair Value Per Share | ||||||||||||||||||||
| (In thousands) | (In thousands) | ||||||||||||||||||||||
Unvested, January 1, 2025 | $ | $ | |||||||||||||||||||||
| Granted | |||||||||||||||||||||||
| Adjustments for performance achievement related to award target | ( | ||||||||||||||||||||||
| Cancellations | ( | ( | |||||||||||||||||||||
| Vested | ( | ( | |||||||||||||||||||||
Unvested, December 31, 2025 | $ | $ | |||||||||||||||||||||
The Company recognized $16.9 million, $22.1 million and $18.7 million in expense related to such awards during the years ended December 31, 2025, 2024, and 2023, respectively. The total fair market value of shares vested and released in 2025, 2024, and 2023 was $9.8 million, $11.3 million and $18.2 million, respectively. Vested awards include shares that have been fully earned but had not been delivered as of December 31, 2025.
Performance stock awards have performance features associated with them. Performance stock, restricted stock and contract stock awards generally have requisite service periods of three years. The fair value of these awards is being expensed on a straight-line basis over the vesting period. As of December 31, 2025, there were no performance stock units (“PSUs”) subject to vest and be released based on 2025 performance achievement.
As of December 31, 2025, there was approximately $27.7 million of total unrecognized compensation costs related to unvested restricted stock, performance stock and contract stock awards. These costs are expected to be recognized over a weighted-average period of approximately two years .
At December 31, 2025, there were approximately 4.6 million shares available for grant under the 2003 Plan.
The Company capitalized share based compensation costs of $0.8 million, $0.7 million, and $0.6 million for the years ended December 31, 2025, 2024, and 2023, into inventory, respectively. Such share-based compensation was recognized as cost of goods sold when related inventory was sold.
CEO Separation
On February 27, 2024, the Company announced that Mr. De Witte would retire from his position as President and Chief Executive Officer and director of the Company following the completion of a succession process and entered into a letter agreement with Mr. De Witte to modify his current employment agreement and put forth the form of a post-employment consulting agreement. The Company applied modification accounting to the outstanding equity-based awards granted to Mr. De Witte as of that date, which revalued and accelerated stock-based compensation associated with equity-based awards granted to him over his expected service period to the Company. Pursuant to this letter agreement, Mr. De Witte’s unvested equity-based awards will continue to vest during his continued service period to the Company and vested stock options were modified such that they will remain exercisable until the earlier of (a) the stated term of the stock options and (b) six months following his cessation of continued service to the Company. As a result of the modifications, the Company recorded a total of $1.9 million in accelerated stock-based compensation expenses for the year ended December 31, 2024.
EMPLOYEE STOCK PURCHASE PLAN
The purpose of the Employee Stock Purchase Plan (the “ESPP”) is to provide eligible employees of the Company with the opportunity to acquire shares of common stock at periodic intervals by means of accumulated payroll deductions. The ESPP is a non-compensatory plan. Under the ESPP, a total of 3.0 million shares of common stock are reserved for issuance. These shares will be made available either from the Company’s authorized but unissued shares of common stock or from shares of common stock reacquired by the Company as treasury stock. At December 31, 2025, 1.8 million shares remain available for purchase
F-37
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
10. RETIREMENT BENEFIT PLANS
DEFINED BENEFIT PLANS
The Company has various defined benefit plans which covers certain employees in France, Japan, Germany and Switzerland.
| Year ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Service cost | $ | $ | $ | ||||||||||||||
| Interest cost | |||||||||||||||||
| Expected return on plan assets | ( | ( | ( | ||||||||||||||
| Amortization of prior service cost (credit) | ( | ( | ( | ||||||||||||||
| Settlements | ( | ||||||||||||||||
| Actuarial losses | ( | ( | |||||||||||||||
| Net periodic benefit cost | $ | $ | $ | ||||||||||||||
The following weighted average assumptions were used to develop net periodic pension benefit costs and the actuarial present values of projected pension benefit obligations for the years ended December 31, 2025 and 2024, respectively:
| As of December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| Discount rate | % | % | % | ||||||||||||||
| Expected return on plan assets | % | % | % | ||||||||||||||
| Rate of compensation increase | % | % | % | ||||||||||||||
| Interest crediting rate for cash balance plans | % | % | % | ||||||||||||||
The Company’s discount rates are determined by considering current yield curves representing high quality, long-term fixed income instruments. The resulting discount rates are consistent with the duration of plan liabilities. In 2025 and 2024, the discount rates were prescribed as the current yield on corporate bonds with an average credit rating of AA or AAA of equivalent currency and term to the liabilities. The expected returns on plan assets represent the average rate of return expected to be earned on plan assets over the period the benefits included in the benefit obligation are to be paid. In developing the expected rates of return, the Company considers returns of historical market data as well as actual returns on the plan assets. Using this reference information, the long-term return expectations for each asset category are developed according to the allocation among those investment categories.
The assessment is determined using projections from external financial sources, long-term historical averages, actual returns by asset class and the various asset class allocations by market.
F-38
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The following sets forth the change in projected benefit obligations and the change in plan assets for the years ended December 31, 2025 and 2024 and a reconciliation of the funded status at December 31, 2025 and 2024, respectively (amounts in thousands):
| Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Change In Projected Benefit Obligations | |||||||||||
| Projected benefit obligations, beginning of year | $ | $ | |||||||||
| Interest cost | |||||||||||
| Service cost | |||||||||||
| Actuarial loss | |||||||||||
| Plan amendments | ( | ||||||||||
| Plan settlements | ( | ( | |||||||||
| Employee contribution | |||||||||||
| Premiums paid | ( | ( | |||||||||
| Benefit payment | ( | ( | |||||||||
| Net transfer in/(out) | |||||||||||
| Effect of foreign currency exchange rates | ( | ||||||||||
| Projected benefit obligations, end of year | $ | $ | |||||||||
| Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Change In Plan Assets | |||||||||||
| Plan assets at fair value, beginning of year | $ | $ | |||||||||
| Actual return on plan assets | |||||||||||
| Employer contributions | |||||||||||
| Employee contributions | |||||||||||
| Plan settlements | ( | ( | |||||||||
| Benefits paid | ( | ( | |||||||||
| Premiums paid | ( | ( | |||||||||
| Net transfer in/(out) | |||||||||||
| Effect of foreign currency exchange rates | ( | ||||||||||
| Plan assets at fair value, end of year | $ | $ | |||||||||
| Year Ended December 31, | |||||||||||
| 2025 | 2024 | ||||||||||
| Reconciliation Of Funded Status | |||||||||||
| Fair value of plan assets | $ | $ | |||||||||
| Benefit obligations | |||||||||||
| Unfunded benefit obligations | $ | $ | |||||||||
The unfunded benefit obligations are included in other liabilities in the consolidated balance sheets at December 31, 2025 and 2024, respectively.
During the periods ended December 31, 2025 and 2024, the Company had a net gains of $1.0 million and $1.3 million, respectively, recognized within accumulated other comprehensive loss that has not been recognized as a component of net periodic benefit cost. The combined accumulated benefit obligations for the defined benefit plans was $71.7 million and $60.1 million as of December 31, 2025 and 2024, respectively.
Unrecognized gains and losses are amortized over the average remaining future service for each plan. For plans with no active employees, they are amortized over the average life expectancy. The amortization of gains and losses is determined by using a 10% corridor of the greater of the market value of assets or the accumulated benefit obligation. Total unamortized gains and losses in excess of the corridor are amortized over the average remaining future service.
F-39
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Prior service costs/benefits for the pension plans are amortized over the average remaining future service of plan participants at the time of the plan amendment.
The net plan assets of the pension plans are invested in common trusts. Common trusts are classified as Level 2 in fair value hierarchy. The fair value of common trusts is valued at net asset value based on the fair values of the underlying investments of the trusts as determined by the sponsor of the trusts. The investment strategy of the Company’s defined benefit plans is both to meet the liabilities of the plans as they fall due and to maximize the return on invested assets within appropriate risk profile.
The benefit plans in France and Germany had no
As of December 31, 2025, no plan assets are expected to be returned to the Company in the next twelve months.
The following table is the summary of expected future benefit payments (in thousands):
| 2026 | $ | ||||
| 2027 | $ | ||||
| 2028 | $ | ||||
| 2029 | $ | ||||
| 2030 | $ | ||||
| Next five years | $ | ||||
As of December 31, 2025, contributions expected to be paid to the plan in 2026 is $3.8 million.
DEFINED CONTRIBUTION PLANS
The Company also has various defined contribution savings plans offered to our U.S. and non-U.S. employees. The Company matches a certain percentage of each employee’s contributions as per the provisions of the plans. Total contributions by the Company to the plans were $12.1 million, $10.4 million and $10.4 million for the years ended December 31, 2025, 2024, and 2023, respectively.
DEFERRED COMPENSATION PLAN
The Company maintains a Deferred Compensation Plan in which certain employees of the Company may defer the payment and taxation of up to 75 % of their base salary and up to 100 % of bonus amounts and other eligible cash compensation.
This deferred compensation is invested in funds offered under this plan and is valued based on Level 1 measurements in the fair value hierarchy. Assets of the Company’s deferred compensation plan are included in other current assets and recorded at fair value based on their quoted market prices. The fair value of these assets at December 31, 2025 and 2024 was $7.9 million and $6.7 million. Offsetting liabilities relating to the deferred compensation plan are included in other liabilities.
11. LEASES AND RELATED PARTY LEASES
The Company leases administrative, manufacturing, research and distribution facilities and vehicles through operating lease agreements. The Company has no material finance leases as of December 31, 2025. Many of the Company’s leases include both lease (e.g., fixed payments including rent) and non-lease components (e.g., common-area or other maintenance costs). For vehicles, the Company has elected the practical expedient to group lease and non-lease components.
Most facility leases include one or more options to renew. The exercise of lease renewal options is typically at the Company’s sole discretion, therefore, the majority of renewals to extend the lease terms are not included in the Right of Use (“ROU”) assets and lease liabilities as they are not reasonably certain of exercise. The Company regularly evaluates renewal options and when they are reasonably certain of exercise, the renewal period is included in the lease term.
As most of the Company’s leases do not provide an implicit rate, the Company uses a collateralized incremental borrowing rate based on the information available at the lease commencement date in determining the present value of the lease payments.
Total operating lease expense for the year ended December 31, 2025 and 2024, was $23.9 million and $24.3 million, respectively, which includes $0.3
F-40
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Supplemental balance sheet information related to operating leases at December 31, 2025 were as follows:
| December 31, 2025 | December 31, 2024 | ||||||||||
| (In thousands, except lease term and discount rate) | |||||||||||
| ROU assets | $ | $ | |||||||||
| Current lease liabilities | |||||||||||
| Non-current lease liabilities | |||||||||||
| Total lease liabilities | $ | $ | |||||||||
| Weighted average remaining lease term (in years): | |||||||||||
| Leased facilities | |||||||||||
| Leased vehicles | |||||||||||
| Leased equipment | |||||||||||
| Weighted average discount rate: | |||||||||||
| Leased facilities | % | % | |||||||||
| Leased vehicles | % | % | |||||||||
| Leased equipment | % | % | |||||||||
Supplemental cash flow information related to leases was as follows:
| December 31, 2025 | December 31, 2024 | ||||||||||
| (In thousands) | |||||||||||
| Cash paid for amounts included in the measurement of lease liabilities: | |||||||||||
| Operating cash flows from operating leases | $ | $ | |||||||||
| ROU assets obtained in exchange for lease liabilities, net of modifications: | |||||||||||
| Operating leases | $ | $ | |||||||||
Future minimum lease payments under operating leases at December 31, 2025 were as follows:
| Related Parties | Third Parties | Total | |||||||||||||||
| (In thousands) | |||||||||||||||||
| 2026 | $ | $ | $ | ||||||||||||||
| 2027 | |||||||||||||||||
| 2028 | |||||||||||||||||
| 2029 | |||||||||||||||||
| 2030 | |||||||||||||||||
| 2031 | |||||||||||||||||
| Thereafter | |||||||||||||||||
| Total minimum lease payments | $ | $ | $ | ||||||||||||||
| Less: Imputed interest | |||||||||||||||||
| Total lease liabilities | |||||||||||||||||
| Less: Current lease liabilities | |||||||||||||||||
| Long-term lease liabilities | $ | ||||||||||||||||
There were no material future minimum lease payments under finance leases at December 31, 2025.
F-41
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Related Party Leases
The Company leases its manufacturing facility in Plainsboro, New Jersey, from a general partnership that is 50 % owned by a principal stockholder of the Company. The term of the current lease agreement is through October 31, 2029 at an annual rate of approximately $0.3 million. The current lease agreement also provides (i) a 5-year renewal option for the Company to extend the lease from November 1, 2029 through October 31, 2034 at the fair market rental rate of the premises, and (ii) another 5-year renewal option to extend the lease from November 1, 2034 through October 31, 2039 at the fair market rental rate of the premises.
Lease Impairment Charge
The Company approved a plan to transition the commercial distribution of PriMatrix® and SurgiMend® from the Boston facility to the Company’s manufacturing facility in Braintree, Massachusetts and permanently cease use of the Boston facility. As a result, in the second quarter of 2024, the Company recorded a $4.6 million impairment charge in the Tissue Technologies reportable segment, as the carrying amounts of the operating lease right-of-use asset and fixed assets related to the Boston facility exceeded their fair values based on the Company’s estimates of future discounted cash flows through the end of the lease term and the end of their remaining useful lives, respectively. The $4.6 million impairment charge was comprised of a $1.7 million impairment of an operating lease right-of-use asset and a $2.9 million write-off of fixed assets, which was recorded as a component of in the consolidated statements of operations.
12. INCOME TAXES
| Years Ended December 31, | |||||||||||||||||
| Dollars in thousands | 2025 | 2024 | 2023 | ||||||||||||||
| United States operations | $ | ( | $ | ( | $ | ( | |||||||||||
| Foreign operations | ( | ||||||||||||||||
| Total | $ | ( | $ | ( | $ | ||||||||||||
A reconciliation of the U.S. Federal statutory rate to the Company’s effective tax rate is as follows:
| Year Ended December 31, 2025 | |||||||||||
| Amount | Percent | ||||||||||
| Federal statutory rate | $ | ( | % | ||||||||
State income taxes, net of federal tax benefit (1) | $ | ( | % | ||||||||
| Foreign tax effects: | |||||||||||
| Switzerland: | |||||||||||
| Swiss IP Transfer | $ | ( | % | ||||||||
| Federal statutory tax rate difference | $ | ( | % | ||||||||
| Cantonal/communal rate difference | $ | ( | % | ||||||||
| Other | $ | ( | % | ||||||||
| Other foreign jurisdictions | $ | ( | % | ||||||||
| Non-taxable or non-deductible items: | |||||||||||
| Goodwill impairment | $ | ( | % | ||||||||
| Other | $ | ( | % | ||||||||
| Cross-border tax laws: | |||||||||||
| Global intangible low-taxed income | $ | ( | % | ||||||||
| Subpart F income | $ | ( | % | ||||||||
| Tax credits: | |||||||||||
| Foreign tax credit | $ | ( | % | ||||||||
| Research and development credit | $ | ( | % | ||||||||
| Valuation allowance | $ | ( | % | ||||||||
| Worldwide changes in prior year unrecognized tax benefits | $ | ( | % | ||||||||
| Effective Tax Rate | $ | ( | % | ||||||||
F-42
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
(1) State Taxes in California, Florida, Illinois, New Jersey, New York, and Tennessee made up the majority (greater than 50 percent) of the tax effect in this category.
| Years Ended December 31, | ||||||||||||||
| 2024 | 2023 | |||||||||||||
| Federal statutory rate | % | % | ||||||||||||
| Increase (decrease) in income taxes resulting from: | ||||||||||||||
| State income taxes, net of federal tax benefit | % | % | ||||||||||||
| Benefit derived from foreign operations | % | ( | % | |||||||||||
| Nondeductible meals and entertainment | ( | % | % | |||||||||||
| Intercompany profit in inventory | % | % | ||||||||||||
| Research and development credit | % | ( | % | |||||||||||
| Nondeductible executive compensation & stock compensation shortfall | ( | % | % | |||||||||||
Transaction and deal related costs | ( | % | % | |||||||||||
| Changes in valuation allowances | ( | % | % | |||||||||||
| Return to provision | % | ( | % | |||||||||||
| Other | ( | % | % | |||||||||||
| Effective tax rate | % | % | ||||||||||||
Our effective tax rate was 8.3 % and 61.9 % of income before income taxes for the years ended December 31, 2025 and December 31, 2024, respectively.
In 2025, the Company’s effective tax rate was primarily driven by the goodwill impairment charge, as a portion of the charge is non-deductible for tax, the transfer of certain intellectual property, and the inclusion of Global Intangible Low-Taxed Income (“GILTI”) and the global minimum tax in certain foreign jurisdictions; offset by federal, state, and international tax benefits generated from operating losses and favorable prior year tax return positions.
In 2024, the Company’s effective tax rate was driven by federal, state, and international tax benefits generated from operating losses in certain jurisdictions, including a $4.4 million benefit from federal and state research tax credits, offset by the inclusion of GILTI.
In 2023, the Company’s effective tax rate was primarily driven by the inclusion of GILTI, offset by a $5.8 million income tax benefit related to a four-year tax credit received by a Swiss subsidiary, which can be used to offset cantonal and communal income and capital taxes during tax years 2024 through 2027. Any unused balance at the end of the 2027 tax period will be forfeited.
During 2025, the Company’s foreign operations generated a $30.7 million decrease in income tax expense when compared to the same period in 2024 due to geographic and business mix of taxable earnings and losses and the transfer of certain intellectual property, among other factors. The 2025 foreign effective tax rate is 12.6 % compared to 12.3 % in 2024.
Changes to income tax laws and regulations, in any of the tax jurisdictions in which the Company operates, could impact the effective tax rate. Various governments, both U.S. and non-U.S., are increasingly focused on tax reform and revenue-raising legislation. On July 4, 2025, the One Big Beautiful Bill Act (“OBBBA”) was enacted in the U.S. The OBBBA includes a number of significant provisions, including the permanent extension of certain expiring provisions of the 2017 Tax Cuts and Jobs Act. Additionally, the OBBBA contains changes to the capitalization of research and development expenses, accelerated fixed asset depreciation, and limitations on deductions for interest expense, among other provisions. The Company has evaluated the impact of the OBBBA on its financial statements and estimates the financial impact for the year ended December 31, 2025 to be immaterial.
Further, legislation in foreign jurisdictions may be enacted, in continued response to the ongoing base erosion and profit-sharing (“BEPS”) project led by the Organization for Economic Cooperation and Development (“OECD”). The OECD released model rules related to a new 15% global minimum tax regime (“Pillar 2”). A number of the jurisdictions that the Company operates in have adopted some form of the model rules, which became effective beginning in 2024. The Pillar 2 rules are complex and provide for delays for implementing the tax during the early transition years, if certain conditions are met.
F-43
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The United States has not adopted Pillar 2, and as of December 31, 2025, the G7 countries announced an agreement to exempt U.S. companies from certain elements of the Pillar 2 framework. If this exemption (the “Pillar Two Side-by-Side Package”) is ultimately enacted into law in relevant jurisdictions, it is expected to be favorable for the Company. However, Pillar 2 remains in effect in other countries, and there is significant uncertainty regarding the implementation of the G7 agreement, the interpretation and consistent application of existing Pillar 2 rules, their interaction with national tax laws, and their consistency with current tax treaty obligations.
The Company is calculating an immaterial expense related to Pillar 2 tax liability for the year ending December 31, 2025. Related changes in U.S. and non-U.S. jurisdictions could have an adverse effect on the Company’s effective tax rate. The Company will continue to monitor legislative activity across its U.S. and non-U.S. jurisdictions.
The provision for income taxes consisted of the following:
| Years Ended December 31, | |||||||||||||||||
| Dollars in thousands | 2025 | 2024 | 2023 | ||||||||||||||
| Current: | |||||||||||||||||
| Federal | $ | $ | ( | $ | |||||||||||||
| State | |||||||||||||||||
| Foreign | |||||||||||||||||
| Total current | $ | $ | $ | ||||||||||||||
| Deferred: | |||||||||||||||||
| Federal | ( | ( | ( | ||||||||||||||
| State | ( | ( | |||||||||||||||
| Foreign | ( | ||||||||||||||||
| Total deferred | $ | ( | $ | ( | $ | ( | |||||||||||
| Provision for income taxes | $ | ( | $ | ( | $ | ||||||||||||
The income tax effects of significant temporary differences that give rise to deferred tax assets and liabilities, shown before jurisdictional netting, are presented below:
F-44
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
| December 31, | |||||||||||
| Dollars in thousands | 2025 | 2024 | |||||||||
| Assets: | |||||||||||
| Doubtful accounts | $ | $ | |||||||||
| Inventory related items | |||||||||||
| Tax credits | |||||||||||
| Accrued vacation | |||||||||||
| Accrued bonus | |||||||||||
| Stock compensation | |||||||||||
| Deferred revenue | |||||||||||
| Net operating loss carryforwards | |||||||||||
| Capitalization of research and development expenses | |||||||||||
| Unrealized foreign exchange loss | |||||||||||
| Leases | |||||||||||
| Other | |||||||||||
| Total deferred tax assets | |||||||||||
| Less valuation allowance | ( | ( | |||||||||
| Deferred tax assets after valuation allowance | $ | $ | |||||||||
| Liabilities: | |||||||||||
| Intangible and fixed assets | ( | ( | |||||||||
| Unrealized foreign exchange gain | ( | ( | |||||||||
| Leases | ( | ( | |||||||||
| Other | ( | ( | |||||||||
| Total deferred tax liabilities | $ | ( | $ | ( | |||||||
| Total net deferred tax (liabilities) assets | $ | $ | ( | ||||||||
The 2017 U.S. Tax Cuts and Jobs Act (the “2017 Tax Act”) contained a provision which requires, for tax purposes, the capitalization and amortization of research and development expenses; effective for years beginning after December 31, 2021. The recently enacted OBBBA allows companies to deduct research and development expenses, reversing the related provision of the 2017 Tax Act. However, the Company continued to capitalize and amortize its research and development expenses in 2025. The Company’s deferred tax assets increased by $5.3 million and $10.4 million at December 31, 2025 and December 31, 2024 respectively within the table above, due to the capitalization of research and development expenses in relation to the 2017 Tax Act.
At December 31, 2025, the Company had net operating loss carryforwards of $46.7 million for federal income tax purposes, $142.8 million for foreign income tax purposes and $85.4 million for state income tax purposes to offset future taxable income. For the federal net operating loss carryforwards, $46.7 million will expire through 2037. For foreign net operating loss carryforwards, $118.9 million will expire through 2028, while the remaining $23.9 million have an indefinite carry forward period. The state net operating loss carryforwards expire through 2045.
The valuation allowance relates to deferred tax assets for certain items that will be deductible for income tax purposes under very limited circumstances and for which the Company believes it will not satisfy the more likely than not threshold for realization of the associated tax benefit. In the event that the Company determines that it would be able to realize more or less than the recorded amount of net deferred tax assets, an adjustment to the deferred tax asset valuation allowance would be recorded in the period such a determination is made.
The valuation allowance at December 31, 2025 increased by $7.1 million, as compared to 2024, primarily driven by a $3.7 million increase related to the Swiss federal and local tax credits and a $3.0 million increase related to U.S. state tax credits. The valuation allowance for 2024 had increased by $3.0 million, as compared to 2023, primarily driven by an increase related to the expiring Swiss federal tax credit.
F-45
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
| Balance at Beginning of Period | Charged to Costs and Expenses | Other | Deductions | Balance at End of Period | |||||||||||||||||||||||||
| Description | |||||||||||||||||||||||||||||
| Dollars in thousands | |||||||||||||||||||||||||||||
Year ended December 31, 2025 | |||||||||||||||||||||||||||||
Deferred tax assets valuation allowance | |||||||||||||||||||||||||||||
Year ended December 31, 2024 | |||||||||||||||||||||||||||||
| Deferred tax assets valuation allowance | ( | ( | |||||||||||||||||||||||||||
Year ended December 31, 2023 | |||||||||||||||||||||||||||||
| Deferred tax assets valuation allowance | |||||||||||||||||||||||||||||
As of December 31, 2025, the Company has not provided deferred income taxes on unrepatriated earnings from foreign subsidiaries as they are deemed to be indefinitely reinvested unless there is a manner under which to remit the earnings with no material tax cost. The Company will repatriate foreign earnings when there is no need for reinvestment overseas and no material tax cost to bring the earnings back to the United States. Reinvestment considerations would include future acquisitions, transactions, and capital expenditure plans.
A reconciliation of the beginning and ending amount of uncertain tax benefits is as follows:
| Years Ended December 31, | |||||||||||||||||
| Dollars in thousands | 2025 | 2024 | 2023 | ||||||||||||||
| (In thousands) | |||||||||||||||||
| Balance, beginning of year | $ | $ | $ | ||||||||||||||
| Gross increases: | |||||||||||||||||
| Current year tax positions | |||||||||||||||||
| Prior years' tax positions | |||||||||||||||||
| Lapse of statute | ( | ( | ( | ||||||||||||||
| Balance, end of year | $ | $ | $ | ||||||||||||||
Approximately $1.6 million of the balance at December 31, 2025 relates to uncertain tax positions that, if recognized, would affect the annual effective tax rate. The Company has no uncertain tax positions at December 31, 2025 related to tax positions for which it is reasonably possible that the amounts could be reduced during the twelve months following December 31, 2025.
The Company recognizes interest and penalties relating to uncertain tax positions in income tax expense. The Company recognized a minimal expense for the years ended December 31, 2025, 2024, and 2023. The Company had minimal interest and penalties accrued for the years ended December 31, 2025, 2024, and 2023.
F-46
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
13. NET INCOME PER SHARE
Basic and diluted net income per share was as follows:
| Years Ended December 31, | |||||||||||||||||
| Dollars in thousands, except per share amounts | 2025 | 2024 | 2023 | ||||||||||||||
| Basic net (loss) income per share: | |||||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | ||||||||||||
| Weighted average common shares outstanding | |||||||||||||||||
| Basic net (loss) income per common share | $ | ( | $ | ( | $ | ||||||||||||
| Diluted net (loss) income per share: | |||||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | ||||||||||||
| Weighted average common shares outstanding — Basic | |||||||||||||||||
| Effect of dilutive securities: | |||||||||||||||||
| Stock options and restricted stock | |||||||||||||||||
| Weighted average common shares for diluted earnings per share | |||||||||||||||||
| Diluted net (loss) income per common share | $ | ( | $ | ( | $ | ||||||||||||
14. ACCUMULATED OTHER COMPREHENSIVE (LOSS) INCOME
Comprehensive (loss) income for the years ended December 31, 2025, 2024 and 2023:
| Dollars in thousands | 2025 | 2024 | 2023 | |||||||||||||||||
| Net (loss) income | $ | ( | $ | ( | $ | |||||||||||||||
| Foreign currency translation adjustment, net of tax | ( | ( | ||||||||||||||||||
| Change in unrealized (gain) loss on derivatives, net of tax | ( | ( | ||||||||||||||||||
| Pension liability adjustment, net of tax | ( | |||||||||||||||||||
| Comprehensive (loss) income, net | $ | ( | $ | ( | $ | |||||||||||||||
Changes in accumulated other comprehensive loss by component between December 31, 2025 and 2024 are presented in the table below, net of tax:
| Dollars in thousands | Gains and Losses on Derivatives | Defined Benefit Pension Items | Foreign Currency Items | Total | ||||||||||||||||||||||
Balance at December 31, 2024 | $ | $ | $ | ( | $ | ( | ||||||||||||||||||||
| Other comprehensive (loss) gain | ( | ( | ||||||||||||||||||||||||
| Less: Amounts reclassified from accumulated other comprehensive income, net | ( | ( | ||||||||||||||||||||||||
| Net current-period other comprehensive (loss) gain | ( | ( | ||||||||||||||||||||||||
Balance at December 31, 2025 | $ | $ | $ | ( | $ | ( | ||||||||||||||||||||
For the year ended December 31, 2025, the Company reclassified a loss of $(47.0 ) million and a gain $1.1 million from AOCI to other income and cost of goods sold, respectively. Additionally, the Company reclassified a gain of $19.3 million from AOCI to interest income.
F-47
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
15. COMMITMENTS AND CONTINGENCIES
In consideration for certain technology, manufacturing, distribution, and selling rights and licenses granted to the Company, the Company has agreed to pay royalties on sales of certain products that it sells. The royalty payments that the Company made under these agreements were not significant for any of the periods presented.
In the ordinary course of its business, the Company is involved in, from time to time, various legal actions, including any matters described below, involving product liability, employment, intellectual property and commercial disputes, shareholder related matters, environmental proceedings, tax disputes, and governmental proceedings and investigations, some of which have been settled by the Company. In the opinion of management, such matters are either adequately covered by insurance or otherwise indemnified, or are not expected, individually or in the aggregate, to result in a material, adverse effect on the Company’s financial condition. However, it is possible that the Company’s results of operations, financial position and cash flows in a particular period could be materially affected by these contingencies.
The Company accrues for loss contingencies when it is deemed probable that a loss has been incurred and that loss is estimable. If the reasonable estimate of a probable loss is a range, and no amount within the range is a better estimate than any other, the minimum amount of the range is recorded. In most cases, significant judgment is required to estimate the amount and timing of a loss to be recorded and actual results may differ from these estimates. The amounts accrued are based on the full amount of the estimated loss before considering insurance proceeds and do not include an estimate for legal fees expected to be incurred in connection with the loss contingency. The Company consistently accrues legal fees expected to be incurred in connection with loss contingencies as those fees are incurred by outside counsel as a period cost.
On December 21, 2023, Fortis Advisors, LLC (representative of the security holders of ACell, Inc. (“ACell”)) filed for arbitration against Integra LifeSciences before the Court of International Arbitration of the International Chamber of Commerce claiming breach of contract related to the earnout consideration from the 2021 acquisition of ACell. Refer to the “Contingent Consideration” subheading of this Note for additional information on the ACell contingent consideration. The arbitration was held in September 2025 and the tribunal’s decision is pending. The Company believes that it has strong defenses to the allegations in the arbitration and intends to defend the matter vigorously.
On September 12, 2023, a securities class action complaint, captioned Pembroke Pines Firefighters & Police Officers Pension Fund v. Integra LifeSciences Holdings Corporation, No. 23-cv-20321 (D.N.J.), was filed by a purported stockholder of the Company in the United States District Court for the District of New Jersey (the “Pembroke Litigation”) against the Company and certain of the Company’s current and former executive officers. The Pembroke Litigation, filed on behalf of a putative class of stockholders who purchased or acquired the Company’s common stock between March 11, 2019 and May 22, 2023, inclusive, alleges violations of Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended, and Rule 10b-5 promulgated thereunder, on the basis of purportedly materially false and misleading statements and omissions relating to certain quality systems issues identified by the FDA at the Company’s Boston facility, the Company’s efforts to remediate those issues, and the Company’s forecasts for certain products in its Tissue Technologies segment. The complaint seeks, among other things, compensatory damages, attorneys’ fees, expert fees, and other costs. The Company believes that it has strong defenses to the allegations in the Pembroke Litigation, and intends to continue to defend the matter vigorously. On July 1, 2025, the class action complaint was dismissed without prejudice. The plaintiffs filed a Second Amended Complaint on August 14, 2025. The Company has filed a motion to dismiss the Second Amended Complaint on October 14, 2025.
On February 21, 2025, a derivative lawsuit captioned Grabowsky v. Integra LifeSciences Holding Corp. et al, No. 3:25-cv-01399 (D.N.J.) was filed in the United States District Court for the District of New Jersey. The action purports to assert derivative claims on behalf of the Company against its current Board of Directors and certain of its current or former officers and directors. The action asserts claims that the individual defendants breached their fiduciary duties and harmed the Company by making false and misleading statements and omissions relating to certain quality systems issues identified by the U.S. Food and Drug Administration (“FDA”) at the Company’s Boston facility, the Company’s efforts to remediate those issues, and the Company’s forecasts for certain products in its Tissue Technologies segment. On March 28, 2025, the derivative lawsuit was voluntarily dismissed by the plaintiff. On April 1, 2025 the lawsuit was dismissed with prejudice.
On May 13, 2025 and May 16, 2025, derivative lawsuits captioned Leverett v. Integra LifeSciences Holding Corp. et al, No. 3:2025-cv-04214 (D.N.J.) and Simpkins v. Integra LifeSciences Holding Corp. et al, No. 3:2025-cv-04446 (D.N.J.) were filed in the United States District Court for the District of New Jersey. The actions purport to assert derivative claims on behalf of the Company against its current Board of Directors and certain of its current or former officers and directors. The actions assert claims that the individual defendants breached their fiduciary duties and harmed the Company by making false and misleading statements and omissions relating to certain quality systems issues identified by the U.S. Food and Drug Administration (“FDA”) at the Company’s Boston, Massachusetts manufacturing facility, the Company’s efforts to remediate those issues, and the Company’s forecasts for certain products in its Tissue Technologies segment. The Company believes that it has strong defenses to the allegations in the lawsuits and intends to defend the matters vigorously.
F-48
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Contingent Consideration
Contingent consideration is recorded at fair value as measured on the date of acquisition. The value recorded is based on estimates of future financial projections under various potential scenarios using either a Monte Carlo simulation or the probability-weighted income approach derived from revenue estimates and probability assessment with respect to the likelihood of achieving contingent obligations. Contingent payments related to acquisitions consist of development, regulatory, and commercial milestone payments, in addition to sales-based payments, and are valued using discounted cash flow techniques that are classified within Level 3 of the fair value hierarchy because they are measured at fair value using significant unobservable inputs, including management’s forecast of future revenues for the acquired businesses as well as management’s estimates of the likelihood of achieving the other specified criteria. Each quarter until such contingent amounts are earned, the fair value of the liability is remeasured at each reporting period and adjusted as a component of operating expenses based on changes to the underlying assumptions. The change in the fair value of sales-based payments is based upon future revenue estimates and increases or decreases as revenue estimates or expectation of timing of payment charges. The estimates used to determine the fair value of the contingent consideration liability are subject to significant judgment and actual results are likely to differ from the amounts originally recorded.
The Company determined the fair value of contingent consideration during the twelve-month period ended December 31, 2025 and 2024 to reflect the change in fair value during the period.
A reconciliation of the opening balances to the closing balances of these Level 3 measurements for the years ended December 31, 2025 and 2024 is as follows (in thousands):
| Contingent Consideration Liability Related to Acquisition of: | ||||||||||||||||||||||||||||||||
Arkis (1) | Derma Sciences (2) | ACell (2) | Surgical Innovations Associates (SIA), Inc. (2) | Total | ||||||||||||||||||||||||||||
Balance as of January 1, 2025 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Payments | ( | ( | ( | |||||||||||||||||||||||||||||
| Change in fair value of contingent consideration liabilities | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||
Balance as of December 31, 2025 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Short-Term | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Long-Term | ||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
F-49
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
| Contingent Consideration Liability Related to Acquisition of: | ||||||||||||||||||||||||||||||||
Arkis (1) | Derma Sciences (2) | ACell (2) | Surgical Innovations Associates (SIA), Inc. (2) | Total | ||||||||||||||||||||||||||||
Balance as of January 1, 2024 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Payments | ( | ( | ||||||||||||||||||||||||||||||
| Change in fair value of contingent consideration liabilities | ( | ( | ( | ( | ||||||||||||||||||||||||||||
Balance as of December 31, 2024 | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Short-Term | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
| Long-Term | ||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||
(1) Location in financial statements: Research and development
(2) Location in financial statements: Selling, general and administrative
Arkis BioSciences Inc.
As part of the acquisition of Arkis BioSciences Inc. (“Arkis”), the Company is required to pay the former shareholders of Arkis up to $25.5 million based on the timing of certain development milestones of $10.0 million and commercial sales milestones of $15.5 million, respectively. The Company used a probability weighted income approach to calculate the fair value of the contingent consideration that considered the possible outcomes of scenarios related to each specified milestone. The Company estimated the fair value of the contingent consideration to be $13.1 million at the acquisition date. In the first quarter of 2025, the Company paid out a development milestone related to design verification procedures for $5.0 million.
Derma Sciences
The Company assumed contingent consideration incurred by Derma Sciences, Inc. (“Derma Sciences”) related to its acquisitions of BioD and the intellectual property related to MediHoney™ products. The Company accounted for the contingent liabilities by recording their fair value on the date of the acquisition based on a probability weighted income approach. The Company has already paid $33.3 million related to the aforementioned contingent liabilities. One contingent milestone remains which relates to net sales of MediHoney™ products exceeding certain amounts defined in the agreement between the Company and Derma Sciences. The potential maximum undiscounted payment amounts to $3.0 million.
ACell, Inc.
As part of the acquisition of ACell, the Company is required to make payments to the former shareholders of ACell up to $100 million in total for years 2022, 2023, and 2025 based on the achievement by the Company of certain revenue-based performance milestones. The Company estimated the fair value of the contingent consideration to be $23.9 million at the acquisition date. The 2022, 2023 and 2025 milestones were not achieved.
Surgical Innovations Associates, Inc.
As part of the acquisition of Surgical Innovations Associates, Inc. (“SIA”), the Company is required to pay to the former shareholders of SIA up to $90.0 million for two separate payments, which are dependent on (1) achieving certain revenue-based performance milestones in 2023, 2024, and 2025 (up to $50.0 million in additional payments), as well as (2) the approval by the FDA of the pre-market approval ("PMA") application for DuraSorb for certain uses by certain timing targets (up to $40.0 million in additional payments). The Company estimated the fair value of the contingent consideration for the revenue based milestone to be $32.6 million at the acquisition date and $25.0 million for the PMA approval milestone at the acquisition date. In the second quarter of 2025, the Company paid out $18.1 million related to the 2024 performance year. Similarly, in the second quarter of 2024, the Company paid out $12.4 million related to the 2023 performance year. The Company calculated the remaining milestone payment for the 2025 performance year based on actual sales for the year ended December 31, 2025. The Company used probabilities of achieving the conditions to calculate the fair value of the contingent consideration for the PMA approval milestone. The liability was reduced in 2025 due to risk and uncertainty around timing of receiving the PMA approval.
F-50
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
16. SEGMENT AND GEOGRAPHIC INFORMATION
The Company is organized primarily on the basis of products and operates two global reportable segments. Resources are allocated and performance is assessed by the Company’s President and Chief Executive Officer, which the Company has determined to be the Chief Operating Decision Maker (“CODM”).
The two reportable segments and their activities are described below.
•The Codman Specialty Surgical segment operations consist of (i) the Neurosurgery business, which sells a full line of products for neurosurgery and neuro critical care such as tissue ablation equipment, dural repair products, cerebral spinal fluid management devices, intracranial monitoring equipment, and cranial stabilization equipment; (ii) the Instruments business, which sells more than 40,000 instrument patterns and surgical and lighting products to hospitals, surgery centers, dental, podiatry, and veterinary offices; and (iii) the ENT business, which includes instrumentation, balloon technologies for sinus dilation and eustachian tube dilation, as well as surgical navigation systems.
•The Tissue Technologies segment operations consists of Wound Reconstruction and Care business, which sells offerings such as skin and wound repair, plastics and surgical reconstruction products and nerve and tendon repair products. The Tissue Technologies segment also includes the Company’s private label business.
The Corporate and Other category includes (i) various executive, finance, human resource, information systems and legal functions, (ii) brand management, and (iii) share-based compensation costs, which are not allocated to the reportable segments.
The accounting policies of both segments are the same as those described in Note 2. Summary of Significant Accounting Policies.
For both segments, the CODM uses segment revenue and segment operating income to assess the performance for each segment and in the annual budgeting and forecasting process. The CODM considers budget-to-actual variances on a quarterly basis for segment revenue and segment operating income when making decisions about allocating capital and personnel to the segments.
The operating results of the reportable segments as presented are not comparable to one another because (i) certain operating segments are more dependent than others on corporate functions for unallocated general and administrative and/or operational manufacturing functions and (ii) the Company does not allocate certain manufacturing costs and general and administrative costs to the reportable segments.
F-51
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Net sales and profit by each reportable segment for the years ended December 31, 2025, 2024, and 2023 are as follows:
| Dollars in thousands | Codman Specialty Surgical | Tissue Technologies | Corporate and Other | Total | |||||||||||||||||||
| 2025 Total revenue, net | $ | $ | $ | $ | |||||||||||||||||||
| Cost of goods sold | |||||||||||||||||||||||
| Research and development | |||||||||||||||||||||||
| Selling, general and administrative | |||||||||||||||||||||||
| Intangible asset amortization | |||||||||||||||||||||||
| Goodwill impairment | |||||||||||||||||||||||
| 2025 Total cost and expenses | |||||||||||||||||||||||
| 2025 Operating income (loss) | $ | $ | ( | $ | ( | ( | |||||||||||||||||
| Interest income | |||||||||||||||||||||||
| Interest expense | ( | ||||||||||||||||||||||
| Other income, net | ( | ||||||||||||||||||||||
| Loss before income taxes | $ | ( | |||||||||||||||||||||
| Codman Specialty Surgical | Tissue Technologies | Corporate and Other | Total | ||||||||||||||||||||
| 2024 Total revenue, net | $ | $ | $ | $ | |||||||||||||||||||
| Cost of goods sold | |||||||||||||||||||||||
| Research and development | |||||||||||||||||||||||
| Selling, general and administrative | |||||||||||||||||||||||
| Intangible asset amortization | |||||||||||||||||||||||
| 2024 Total cost and expenses | |||||||||||||||||||||||
| 2024 Operating income (loss) | $ | $ | $ | ( | |||||||||||||||||||
| Interest income | |||||||||||||||||||||||
| Interest expense | ( | ||||||||||||||||||||||
| Other income, net | |||||||||||||||||||||||
| Loss before income taxes | $ | ( | |||||||||||||||||||||
| Codman Specialty Surgical | Tissue Technologies | Corporate and Other | Total | ||||||||||||||||||||
| 2023 Total revenue, net | $ | $ | $ | $ | |||||||||||||||||||
| Cost of goods sold | |||||||||||||||||||||||
| Research and development | |||||||||||||||||||||||
| Selling, general and administrative | |||||||||||||||||||||||
| Intangible asset amortization | |||||||||||||||||||||||
| 2023 Total cost and expenses | |||||||||||||||||||||||
| 2023 Operating income (loss) | $ | $ | $ | ( | |||||||||||||||||||
| Interest income | |||||||||||||||||||||||
| Interest expense | ( | ||||||||||||||||||||||
| Other income, net | |||||||||||||||||||||||
| Income before income taxes | $ | ||||||||||||||||||||||
F-52
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
The Company does not allocate any assets to the reportable segments. No asset information is reported to the CODM and disclosed in the financial information for each segment. The Company attributes revenues to geographic areas based on the location of the customer. Total revenue by major geographic area consisted of the following:
| Dollars in thousands | United States | Europe | Asia Pacific | Rest of the World | Consolidated | ||||||||||||||||||||||||
| Total revenue, net: | |||||||||||||||||||||||||||||
| 2025 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| 2024 | |||||||||||||||||||||||||||||
| 2023 | |||||||||||||||||||||||||||||
| Total long-lived assets: | |||||||||||||||||||||||||||||
| 2025 | |||||||||||||||||||||||||||||
| 2024 | |||||||||||||||||||||||||||||
| 2023 | |||||||||||||||||||||||||||||
17. FAIR VALUE MEASUREMENTS
FASB Topic 820, Fair Value Measurement (“ASC 820”) defines fair value as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. When determining fair value, the Company considers the principal or most advantageous market in which it would transact and considers assumptions that market participants would use when pricing the asset or liability.
Valuation techniques used to measure fair value should maximize the use of observable inputs and minimize the use of unobservable inputs. ASC 820 establishes a three-level hierarchy of the inputs (i.e., assumptions that market participants would use in pricing an asset or liability) used to measure fair value, giving the highest priority to quoted prices in active markets and the lowest priority to unobservable inputs in measuring fair value. The categorization within the valuation hierarchy is based on the lowest level of input that is significant to the entire fair value measurement. The three levels of the valuation hierarchy are defined as follows:
Level 1: Inputs to the valuation methodology are quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs to the valuation methodology are other observable inputs, including quoted market prices for similar assets or liabilities in active markets; quoted prices for identical or similar assets or liabilities in markets that are not active; inputs other than quoted prices that are observable for the asset or liability; and inputs that are derived principally from or corroborated by observable market data by correlation or other means.
Level 3: Inputs to the valuation methodology are unobservable inputs that are supported by little or no market activity and are based on management’s best estimate of inputs market participants would use in pricing the asset or liability at the measurement date, including assumptions about risk.
Assets and Liabilities Measured at Fair Value on a Recurring Basis
The Company has investments in time deposits that are classified within Level 1 of the fair value hierarchy because they are valued using quoted market prices, as well as certain debt obligations that are classified within Level 1 of the fair value hierarchy because they are valued using quoted market prices. The investments in time deposits are classified as cash and cash equivalents and short-term investments on the consolidated balance sheets which is determined based on maturities at the time of purchase and re-evaluated at each balance sheet date.
The Company also has investments in derivative instruments, which are comprised of interest rate swaps, cross currency swaps, net investment hedges, and forward foreign currency contracts that are classified within Level 2 of the fair value hierarchy because they are valued using analyses obtained from independent third-party valuation specialists based on market observable inputs. The fair values of these derivative contracts represent the estimated amounts the Company would receive or pay to terminate the contracts. Refer to Note 6. Derivative Instruments for further discussion and information on these derivative contracts.
In addition, the Company has contingent consideration liabilities that are classified within Level 3 of the fair value hierarchy because they are measured at fair value using significant unobservable inputs, including management’s forecast of future revenues for the acquired businesses as well as management’s estimates of the likelihood of achieving the other specified criteria. Refer to Note 15. Commitments and Contingencies for additional information on these contingent consideration liabilities.
F-53
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS (continued)
Assets and liabilities measured and recorded at fair value on a recurring basis as of December 31, 2025, 2024, and 2023 consisted of the following:
| Years Ended | ||||||||||||||
| Dollars in thousands | Fair Value Measurement | 2025 | 2024 | 2023 | ||||||||||
| Assets: | ||||||||||||||
| Cash and marketable securities | Level 1 | $ | $ | $ | ||||||||||
| Short-term investments | Level 1 | |||||||||||||
| Interest rate swaps | Level 2 | |||||||||||||
| Pension plan assets | Level 2 | |||||||||||||
| Foreign currency forward contracts | Level 2 | |||||||||||||
| Foreign currency forward contracts (not designated as hedges) | Level 2 | |||||||||||||
| Total Assets: | $ | $ | $ | |||||||||||
| Liabilities: | ||||||||||||||
| Cross currency swaps | Level 2 | $ | $ | $ | ||||||||||
| Net investment hedges | Level 2 | |||||||||||||
| Foreign currency forward contracts | Level 2 | |||||||||||||
| Contingent consideration | Level 3 | |||||||||||||
| Pension projected benefit obligation | ||||||||||||||
| Total Liabilities: | $ | $ | $ | |||||||||||
There were no transfers into or out of Level 3 during the years ended December 31, 2025, December 31, 2024, and December 31, 2023.
Assets and Liabilities Measured at Fair Value on a Non-Recurring Basis
The Company remeasures the fair value of certain assets and liabilities, including property, plant and equipment, operating lease - right of use assets, and goodwill and other intangible assets, upon the occurrence of certain events. The amounts recognized were recorded to remeasure the carrying amount of assets to the assets’ fair values, which were generally estimated, based upon a market participant’s perspective, using Level 3 measurements, including values estimated using the income approach.
Other than the fair value estimates disclosed in Note 4. Acquisitions and Divestitures, Note 7. Goodwill and Other Intangibles, and Note 11. Lease and Related Party Leases, there were no non-recurring fair value measurements during the years ended December 31, 2025, December 31, 2024, and December 31, 2023.
F-54
IART-2025.12.31-Exhibit 4.4
Description of the Company’s Common Stock Registered
Under Section 12 of the Exchange Act
The following is a description of the common stock of Integra LifeSciences Holdings Corporation (the “Company”). The description does not purport to be complete and is subject to and qualified in its entirety by reference to the Company’s amended and restated certificate of incorporation, or the certificate of incorporation, and its third amended and restated by-laws, or the bylaws) each of which are filed as exhibits to this Annual Report on Form 10-K, and to the provisions of the Delaware General Corporation Law (“DGCL”).
General Matters
Authorized Shares
The Company’s authorized capital stock consists of 255,000,000 shares of stock, of which 240,000,000 shares are designated as common stock, par value $0.01 per share, and 15,000,000 shares are designated as preferred stock, no par value.
Dividends
We have not paid any cash dividends on our common stock since our formation. Subject to preferences that may apply to shares of preferred stock outstanding at the time, the holders of outstanding shares of common stock are entitled to receive dividends out of assets legally available therefor at such times and in such amounts as the board of directors may from time to time determine. However, our senior credit facility limits the amount of dividends that we may pay. Any future determinations to pay cash dividends on our common stock will be at the discretion of our board of directors and will depend upon our financial condition, results of operations, cash flows and other factors that our board of directors deems relevant.
Voting Rights
Each stockholder is entitled to one vote in person or by proxy for each share of the capital stock having voting power held by such stockholder. Stockholders do not have cumulative voting rights. The Company’s board of directors is not classified and each director is elected annually. The voting standard for the election of directors is a majority of votes cast in uncontested elections. In contested elections where the number of nominees exceeds the number of directors to be elected, the vote standard is a plurality of the votes cast. Holders of a majority of the outstanding shares of common stock entitled to vote in any election of directors may elect all of the directors standing for election.
Preemptive or Similar Rights
Our common stock is not entitled to preemptive rights and is not subject to conversion or redemption.
Right to Receive Liquidation Distributions
Upon the occurrence of a liquidation, dissolution or winding-up, the holders of shares of common stock would be entitled to share ratably in the distribution of all of our assets remaining available for distribution after satisfaction of all its liabilities and the payment of the liquidation preference of any outstanding preferred stock.
Stock Exchange
Our common stock is traded on the Nasdaq Global Select Market under the symbol “IART”.
Preferred Stock
The Company’s Board of Directors has the authority to issue up to 15,000,000 shares of Preferred Stock from time to time in one or more series and with such rights and preferences as determined by the Board with respect to each series. The issuance of preferred stock could have the effect of decreasing the market price of our common stock and could adversely affect the voting and other rights of holders of common stock.
Statutory Business Combination Provision
As a Delaware corporation, we are subject to Section 203 of the General Corporation Law of the State of Delaware, or DGCL. In general, Section 203 of the DGCL prevents an “interested stockholder,” which is defined generally as a person owning 15% or more of a Delaware corporation’s outstanding voting stock or any affiliate or associate of that person, from engaging in a broad range of “business combinations” with the corporation for three years following the date that person became an interested stockholder unless:
•before that person became an interested stockholder, the board of directors of the corporation approved the transaction in which that person became an interested stockholder or approved the business combination;
•on completion of the transaction that resulted in that person’s becoming an interested stockholder, that person owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, other than stock held by (1) directors who are also officers of the corporation or (2) any employee stock plan that does not provide employees with the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or
•following the transaction in which that person became an interested stockholder, both the board of directors of the corporation and the holders of at least two-thirds of the outstanding voting stock of the corporation not owned by that person approve the business combination.
Under Section 203 of the DGCL, the restrictions described above also do not apply to specific business combinations proposed by an interested stockholder following the announcement or notification of designated extraordinary transactions involving the corporation and a person who had not been an interested stockholder during the previous three years or who became an interested stockholder with the approval of a majority of the corporation’s directors, if a majority of the directors who were directors prior to any person’s becoming an interested stockholder during the previous three years, or were recommended for election or elected to succeed those directors by a majority of those directors, approve or do not oppose that extraordinary transaction.
Anti-Takeover Effects of our Certificate of Incorporation and our Bylaws
Some of the provisions of our certificate of incorporation and bylaws discussed below may have the effect, either alone or in combination with the provisions of our certificate of incorporation discussed above and Section 203 of the DGCL, of making more difficult or discouraging a tender offer, proxy contest, merger or other takeover attempt that our board of directors opposes but that a stockholder might consider to be in its best interest.
Special Meetings of Stockholders. Our bylaws provide that a special meeting of our stockholders may only be called by (i) the chairman of our board of directors, (ii) the president or (iii) our board of directors.
Stockholder Action by Written Consent. Our stockholders may act by written consent without a meeting, subject to the requirements in our bylaws for setting a record date for the written consent. Any stockholder seeking to have the stockholders authorize or take corporate action must request that our Board of Directors fix a record date. Such notice must include the same information required for a stockholder proposal and be submitted to our Board of Directors as described in our bylaws.
Vacancies on the Board of Directors. Our certificate of incorporation provides that the number of directors will be fixed exclusively by, and may be increased or decreased exclusively by, our board of directors from time to time, but will not be less than three nor more than thirteen. Our bylaws provide that vacancies on the board of directors arising through death, resignation, retirement or removal shall be filled only by a majority of the directors then in office whether or not the remaining directors constitute a quorum. These provisions will prevent our stockholders from removing incumbent directors without cause and filling the resulting vacancies with their own nominees.
Our certificate of incorporation provides that the number of directors will be fixed exclusively by, and may be increased or decreased exclusively by, our board of directors from time to time, but will not be less than three nor more than thirteen. Our certificate of incorporation provides that directors may be removed only by the Delaware Chancery Court under Section 225(c) of the DGCL or for cause (as such term is defined in our certificate of incorporation) as determined by a vote of at least 80% of the voting power of our outstanding voting stock. A vacancy on our board of directors may be filled by a vote of a majority of the directors in office, and a director appointed to fill a vacancy serves for the remainder of the term of the class of directors in which the vacancy occurred.
Advance Notice Requirements for Stockholder Proposals and Director Nominations. Our bylaws contain provisions requiring that advance notice be delivered to us of any business to be brought by a stockholder before an annual meeting of stockholders and providing for certain procedures to be followed by stockholders in nominating persons for election to our board of directors. Generally, the advance notice provisions provide that the stockholder must give written notice to our Secretary not less than 90 days nor more than 120 days prior to the anniversary date of the immediately preceding annual meeting, except that in the event that the annual meeting is called for a date that is more than 30 days before or more than 60 days after such anniversary date, notice by the stockholder to be timely must be so delivered not later than the 90th day prior to the date of such annual meeting (or, if later, then the 10th day following the day on which public disclosure of the date of such annual meeting was first made). The notice must set forth specific information regarding that stockholder and that business or director nominee, as described in our bylaws.
Amendment of Certain Provisions of the Certificate of Incorporation and Bylaws. Under the DGCL, the stockholders of a corporation have the right to adopt, amend or repeal the bylaws and, with the approval of the board of directors, the certificate of incorporation of a corporation. In addition, if the certificate of incorporation so provides, the bylaws may be adopted, amended or repealed by the board of directors. Our Certificate provides that the bylaws may be amended or repealed by our board of directors. Our certificate of incorporation and bylaws also confer on our board of directors the power to adopt, amend or repeal our amended and restated bylaws with the affirmative vote of a majority of the directors then in office.
Forum Selection. Our bylaws provide, unless we consent in writing to the selection of an alternative forum, that the sole and exclusive forum for (a) any derivative action or proceeding brought on our behalf, (b) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our stockholders, (c) any action asserting a claim against us or any of our directors, officers or other employees arising pursuant to any provision of the DGCL, our certificate of incorporation or our bylaws (in each case, as they may be amended from time to time) or (d) any action asserting a claim against us or any of our directors, officers or other employees governed by the internal affairs doctrine, will be a state court located within the State of Delaware (or, if no state court located within the State of Delaware has jurisdiction, the federal district court for the District of Delaware). Any person that purchases or otherwise acquires an interest in our stock will be deemed to have notice of and agree to comply with the foregoing provisions.
Our bylaws provide that a state court of the State of Delaware (or, if no state court located within the State of Delaware has jurisdiction, the federal district court for the District of Delaware) shall be the sole and exclusive forum for: (i) any derivative action or proceeding brought on behalf of Integra; (ii) any action asserting a claim of breach of a fiduciary duty owed by any director or officer of Integra to Integra or the stockholders; (iii) any action asserting a claim against Integra arising pursuant to any provision of the DGCL, our certificate of incorporation or our bylaws (as each may be amended, from time to time); or (iv) any other action asserting a claim against Integra or any director or officer of Integra that is governed by or subject to the internal affairs doctrine for choice of law purposes. However, the forum selection provision does not apply to any claims, actions or proceedings arising under the Securities Act of 1933, as amended, which we refer to as the “Securities Act,” or the Exchange Act. Unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States of America shall be the sole and exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act of 1933, as amended, the Exchange Act, or the respective rules and regulations promulgated thereunder. Any person or entity purchasing or otherwise acquiring any interest in shares of our stock will be deemed to have notice of and consented to the exclusive forum provisions in our bylaws.
Preferred Stock. As discussed above under “General Matters—Preferred Stock,” our certificate of incorporation authorizes our board of directors, without the approval of our stockholders, to provide for the issuance of all or any shares of our preferred stock in one or more series and to determine the designation, powers, preferences and relative, participating, optional or other special rights, and the qualifications, limitations or restrictions applicable to any of those rights, including dividend rights, voting rights, conversion or exchange rights, terms of redemption and liquidation preferences, of each series. The issuance of shares of our preferred stock, or the issuance of rights to purchase shares of preferred stock, could be used to discourage an unsolicited acquisition proposal. In addition, under some circumstances, the issuance of preferred stock could adversely affect the voting power of our common stockholders.
IART-2025.12.31-EX-10.3(f)
RESTRICTED STOCK UNIT AWARD AGREEMENT
THIS RESTRICTED STOCK UNIT AWARD AGREEMENT (the “Award Agreement”), dated as of [ ] (the “Award Date”), is made by and between Integra LifeSciences Holdings Corporation, a Delaware corporation (the “Company”), and [ ], hereinafter referred to as the “Participant,” a Key Employee or Associate (as defined in the Plan).
WHEREAS, the Company has established and maintains the Integra LifeSciences Holdings Corporation Fifth Amended and Restated 2003 Equity Incentive Plan, as amended from time to time (the “Plan”);
WHEREAS the Company has determined that it would be to the advantage and best interest of the Company and its shareholders to grant an award of Restricted Stock Units (“RSUs”) provided for herein to the Participant as an incentive for increased efforts during the Participant’s employment with or services for the Company or its Related Corporations or Affiliates;
WHEREAS, Restricted Stock Units can be granted, and the shares of common stock of the Company (the “Shares”) subject to the RSUs can be issued, under Section 7.8 of the Plan; and
NOW, THEREFORE, in consideration of the various covenants herein contained, and intending to be legally bound hereby, the parties hereto agree as follows:
ARTICLE I.
DEFINITIONS
DEFINITIONS
Capitalized terms not otherwise defined below shall have the meaning set forth in the Plan. The masculine pronoun shall include the feminine and neuter, and the singular the plural, where the context so indicates.
Section 1.1“Award Date” shall have the meaning set forth in the recitals.
Section 1.2“Cause” shall mean, with respect to any Participant, “Cause” as defined in such Participant’s employment agreement or severance agreement with the Company if such an agreement exists and contains a definition of Cause or, if no such agreement exists or such agreement does not contain a definition of Cause, then Cause shall mean (i) the Participant’s neglect of duties or responsibilities that he or she is required to perform for the Company or any willful failure by the Participant to obey a lawful direction of the Board or the Company; (ii) the Participant’s engaging in any act of dishonesty, fraud, embezzlement, misrepresentation or other act of moral turpitude; (iii) the Participant’s knowing violation of any federal or state law or regulation applicable to the Company’s business; (iv) the Participant’s material breach of any confidentiality, non-compete agreement or invention assignment agreement or any other material agreement between the Participant and the Company; (v) the Participant’s conviction of, or plea of nolo contendere to, any felony or crime of moral turpitude which conviction or plea is materially and demonstrably injurious to the Company or any of its subsidiaries; (vi) failure by the Participant to comply with the Company’s material written policies or rules; or (vii) the Participant’s act or omission in the course of his or her employment which constitutes gross negligence or willful misconduct.
Section 1.3“Covenants Agreement” shall mean the Confidentiality and Invention Disclosure and Non-Compete Agreement by and between the Company and the Participant, executed in connection with the Award Agreement in Appendix A.
Section 1.4“Plan” shall have the meaning set forth in the recitals.
Section 1.5“Restricted Stock Units” or “RSUs” shall mean a conditional right to receive Shares pursuant to the terms of the Plan and this Award Agreement upon vesting and settlement, subject to the Participant’s continued employment through the vesting date set forth in the Notice of Grant of Award, unless otherwise set forth in this Award Agreement.
Section 1.6“Rule 16b-3” shall mean that certain Rule 16b-3 under the U.S. Exchange Act, as such Rule may be amended from time to time.
Section 1.7“Secretary” shall mean the Secretary of the Company.
Section 1.8“Service Recipient” shall mean the Company, Related Corporation or Affiliate with which the Participant has an employment or service relationship.
Section 1.9“Termination Date” shall mean the date the Participant is no longer actively providing services to the Company, a Related Corporation or Affiliate (regardless of the reason for such termination and whether or not later found to be invalid or in breach of applicable laws in the jurisdiction where the Participant is employed or rendering services or the terms of the Participant’s employment or service agreement, if any) and, unless otherwise expressly provided for in this Award Agreement or determined by the Company, will not be extended by any notice period (e.g., the Participant’s period of employment or service will not include any contractual notice period ). The Committee shall have the exclusive discretion to determine when the Participant’s service is terminated for purposes of this Award of RSUs (including when the Participant is no longer considered to be providing service while on a leave of absence).
ARTICLE II.
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
Section 2.1Grant of RSUs. The Company hereby grants [ ] RSUs to the Participant as of the Award Date, in accordance with and subject to the terms, conditions and restrictions of this Award Agreement and the Plan. RSUs shall be credited to an account maintained for the Participant on the books of the Company, as of the Award Date.
Section 2.2Vesting of RSUs. Subject to the terms and conditions of this Award Agreement and the Plan, [ ]% of the RSUs awarded hereunder shall vest on each of the [ ] of the Award Date and [ ]% shall vest on the [ ] anniversary of the Award Date (each such date a “Vesting Date”) provided the Participant remains a Key Employee or Associate from the Award Date until the applicable Vesting Date.
Section 2.3Termination of Service (other than by (i) Death or Disability or (ii) a Qualifying Termination on or Following a Change in Control). Upon the Participant’s termination of service prior to a Vesting Date, other than by (i) death or Disability or (ii) a Qualifying Termination on or within twelve months following the date of a Change in Control and, except as otherwise determined by the Compensation Committee of the Company (the “Committee”), any RSUs granted to the Participant under this Award Agreement which have not yet vested on the Termination Date shall terminate without payment and shall be of no further force or effect from and after the Termination Date.
Section 2.4Death or Disability. If a Participant dies or suffers a Disability prior to the Vesting Date, all RSUs held by the Participant immediately prior to death or Disability, which have not yet vested at the time of death or Disability, shall become fully vested and all forfeiture restrictions thereon shall lapse upon such death or Disability.
Section 2.5Change in Control. In the event that a Change in Control occurs and the Participant incurs a Qualifying Termination (which for purposes of this Award Agreement must qualify as a “separation from service” within the meaning of Section 409A) on or within twelve (12) months following the date of such Change in Control, the RSUs shall become fully vested and all forfeiture restrictions shall lapse upon such Qualifying Termination.
Section 2.6Acceleration of Vesting. Notwithstanding the provisions of Sections 2.2, 2.3, 2.4, and 2.5, the Committee may, in its sole discretion, at any time prior to or following the events contemplated in such Sections, permit the vesting of any or all RSUs held by the Participant and the issuance of Shares in respect of such RSUs in the manner and on the terms authorized by the Committee, provided that the Committee will not, in any case, authorize the vesting of an RSU or the issuance of a Share pursuant to this Section beyond the Vesting Date. If so accelerated, such RSUs will be considered as having vested as of the date specified by the Committee. The payment of Shares upon vesting pursuant to this Section 2.6 shall in all cases be paid at a time or in a manner that is exempt from, or complies with, Section 409A of the Code and the regulations promulgated thereunder (“Section 409A”). The prior sentence may be superseded in a future agreement or amendment to this Award Agreement only by direct and specific reference to such sentence. Notwithstanding anything in the Plan or this Award Agreement or any other agreement (whether entered into before, on or after the Award Date), if the vesting of the balance, or some lesser portion of the balance, of the RSUs is accelerated in connection with the Participant’s termination of service (provided that such termination is a “separation from service” within the meaning of Section 409A, as determined by the Company), other than due to the Participant’s death, and if (x) the Participant is a “specified employee” within the meaning of Section 409A at the time of such termination of service and (y) the payment of such accelerated RSUs will result in the imposition of additional tax under Section 409A if paid to the Participant on or within the six (6) month period following the Participant’s termination of service, then the payment of such accelerated RSUs will not be made until the date six (6) months and one (1) day following the date of Participant’s termination of service, unless the Participant dies following his or her termination of service, in which case, the RSUs will be paid in Shares to the Participant’s estate as soon as practicable following his or her death.
Section 2.7Settlement. On or as soon as administratively practicable (and any event within 30 days) following each Vesting Date, the Company shall cause to be issued to the Participant Shares with respect to the RSUs that become vested on such Vesting Date or sooner pursuant to Section 2.4, 2.5 or 2.6.
Section 2.8Fractions. No fractional Share will be issued pursuant to an award granted hereunder. The number of Shares issuable to the Participant upon payment of any award granted under this Award Agreement will be rounded down to the nearest whole number of Share. No payment or other adjustment will be made with respect to the fractional Share so disregarded.
Section 2.9Section 409A. It is the intent of this Award Agreement and the Plan that this Award Agreement and the Plan and all payments and benefits to U.S. taxpayers thereunder be exempt from, or comply with, the requirements of Section 409A so that none of the RSUs provided under this Award Agreement or Shares issuable thereunder will be subject to the additional tax imposed under Section 409A, and any ambiguities herein will be interpreted in accordance with such intention. Each payment payable under this Award Agreement is intended to constitute a separate payment for purposes of Section 409A. However, in no event will the
Company reimburse the Participant, or be otherwise responsible for, any taxes or costs that may be imposed on the Participant as a result of Section 409A. To the extent that any provision of the Award Agreement or the Plan would cause a conflict with the requirements of Section 409A of the Code or would cause the administration of the Award Agreement to fail to satisfy the requirements of Section 409A, such provision shall be deemed null and void to the extent permitted by applicable law.
Section 2.10Adjustments to Restricted Stock Units. In the event of any subdivision, consolidation, stock dividend, capital reorganization, reclassification, exchange, or other change with respect to the Shares, or a consolidation, amalgamation, merger, spin-off, sale, lease or exchange of all or substantially all of the property of the Company or other distribution of the Company's assets to stockholders (other than the payment of ordinary cash dividends), the account of the Participant and the RSUs held by the Participant shall be adjusted in such manner, if any, as the Committee may in its discretion deem appropriate to preserve, proportionally, the interests of the Participant under the Plan.
Section 2.11Restrictive Covenants. As a condition precedent to the grant of the RSUs, the Participant hereby agrees to be subject to the confidentiality, inventions assignment, non-competition and other restrictive covenants set forth in the Covenants Agreement.
Section 2.12Forfeiture/Clawback. Notwithstanding anything contained in the Plan or the Award Agreement to the contrary, the RSUs shall be subject to the provisions of any clawback, repayment, recapture or recoupment policy implemented by the Company, including any such policy(ies) adopted to comply with applicable law (including without limitation the Dodd-Frank Wall Street Reform and Consumer Protection Act and Section 10D of the Securities Exchange Act of 1934, as amended), governmental regulation or securities exchange listing standards and any rules or regulations promulgated thereunder, to the extent set forth in such policy and/or in any notice or agreement relating to the RSUs under the Plan. The Company may reduce, cancel, or withhold against the RSUs or any other outstanding unvested or vested cash or equity based compensation owed or due to the Grantee, in each case, to the fullest extent permitted by applicable law in order to enforce the terms of any such clawback policy(ies).
ARTICLE III.
ADDITIONAL TERMS AND CONDITIONS
ADDITIONAL TERMS AND CONDITIONS
Section 3.1Status of Plan. The terms of the Plan are incorporated by reference into, and made part of, this Award Agreement. In the event of a conflict between the Plan and this Award Agreement, the terms of the Plan shall govern.
Section 3.2Committee’s Authority. The Committee shall have the power to interpret the Plan and this Award Agreement and to adopt such rules for the administration, interpretation and application of the Plan as are consistent therewith and to interpret or revoke any such rules (including, but not limited to, the determination of whether or not any RSUs have vested). All actions taken and all interpretations and determinations made by the Committee in good faith shall be final and binding upon the Participant, the Company and all other interested persons. The Committee shall not be personally liable for any action, determination or interpretation made in good faith with respect to the Plan or this Award Agreement. The Committee shall, in its absolute discretion, determine when such conditions have been fulfilled.
Section 3.3Nature of Grant. By accepting the grant of the RSUs, the Participant acknowledges, understands and agrees that:
(i)the Plan is established voluntarily by the Company, is discretionary in nature and may be modified, amended, suspended or terminated by the Company at any time to the extent permitted in the Plan;
(ii)the grant of the RSUs is exceptional voluntary and occasional and does not create any contractual or other right to receive future grants of RSUs or benefits in lieu of RSUs, even if RSUs have been awarded in the past;
(iii)all decisions with respect to future grants of RSUs, if any, will be at the sole discretion of the Company;
(iv)the Participant is voluntarily participating in the Plan;
(v)in consideration of an in exchange for the issuance of the RSUs pursuant to this Award Agreement and other good and valuable consideration as set forth herein, the receipt and sufficiency of which are hereby acknowledged by the Participant, the Participant shall be bound by the Covenants Agreement;
(vi)except to the extent explicitly and minimally required under applicable legislation, no Affiliate or Related Corporation (including, but not limited to, the Service Recipient) has any obligation to make any payment of any kind to the Participant under this Award Agreement;
(vii)the grant of the RSUs and any Shares subject to the RSUs, and the income from and value of same, are not intended to replace any pension rights or compensation;
(viii)unless otherwise agreed with the Company in writing, the RSUs and the Shares subject to the RSUs, and the income from and value of same, are not granted as consideration for, or in connection with, any service the Participant may provide as a director of a Related Corporation or an Affiliate;
(ix)except to the extent explicitly and minimally required under applicable legislation, the award of the RSUs and the Shares subject to the RSUs, and the income from and value of same are not part of normal or expected compensation or salary for any purposes, including, but not limited to, calculating any severance, resignation, termination, end of service payments, bonuses, retirement or welfare benefits or similar payments and in no event should be considered as compensation for, or relating in any way to, past services for the Company or any Related Corporation or Affiliate;
(x)the future value of the Shares underlying the RSUs is unknown, indeterminable and cannot be predicted with certainty;
(xi)except to the extent explicitly and minimally required under applicable legislation, no claim or entitlement to compensation or damages shall arise from forfeiture of the RSUs resulting from the Participant’s termination of employment, or other service relationship (for any reason whatsoever and whether or not later found to be invalid or in breach of employment laws in the jurisdiction where the Participant is employed or the terms of the Participant’s employment agreement, if any); and
(xii)the Company is not providing any tax, legal or financial advice, nor is the Company making any recommendations regarding the Participant’s participation in the Plan or sale of the Shares acquired upon vesting and settlement of the RSUs. The Participant should consult with his or her own personal tax, legal and financial advisors regarding his or her participation in the Plan before taking any action related to the Plan.
Section 3.4Responsibility for Taxes. The Participant acknowledges that, regardless of any action taken by the Company or, if different, the Service Recipient, the ultimate liability for all income tax, social insurance, payroll tax, fringe benefits tax, payment on account, and other tax-related items related to the Participant’s participation in the Plan and legally applicable to the Participant or deemed applicable to the Participant (“Tax-Related Items”) is and remains Participant’s responsibility and may exceed the amount, if any, actually withheld by the Company or the Service Recipient. The Participant further acknowledges that the Company and/or the Service Recipient (i) make no representations or undertakings regarding the treatment of any Tax-Related Items in connection with any aspect of the RSUs or the underlying Shares, including, but not limited to, the grant, vesting or settlement of the RSUs, the subsequent sale of Shares acquired pursuant to such settlement and the receipt of any dividends or other distributions paid on the Shares, and (ii) do not commit to and are under no obligation to structure the terms of the grant or any aspect of the RSUs to reduce or eliminate the Participant’s liability for Tax-Related Items or achieve any particular tax result. Further, if the Participant is subject to Tax-Related Items in more than one jurisdiction, the Participant acknowledges that the Company and/or the Service Recipient (or former service recipient, as applicable) may be required to withhold or account for Tax-Related Items in more than one jurisdiction.
(i)Tax Withholding. Prior to any relevant taxable or tax withholding event, as applicable, the Participant agrees to make arrangements satisfactory to the Company and the Service Recipient to satisfy any applicable withholding obligations the Company or the Service Recipient may have for Tax-Related Items. In this regard, the Participant authorizes the Company and the Service Recipient, as applicable, and their respective agents, at their discretion, to satisfy any applicable withholding obligation for Tax-Related Items by one or a combination of the following:
(a)withholding from wages or other cash compensation payable to the Participant by the Company or any Service Recipient;
(b)requiring the Participant to tender a cash payment or make a payment in a form acceptable to the Company or the Service Recipient;
(c)withholding from proceeds of the sale of Shares to be issued upon vesting of the RSUs either through a voluntary sale or through a mandatory sale arranged by the Company (on the Participant’s behalf pursuant to this authorization without further consent);
(d)withholding in Shares to be issued upon vesting of the RSUs (in which case the Participant will be deemed to have been issued the full number of Shares subject to the vested portion of the RSUs, notwithstanding that a number of the Shares are held back solely for the purpose of paying the Tax-Related Items); and
(e)any other method acceptable to the Company and to the extent required under the Plan and applicable laws, approved by the Committee.
The Company and/or the Service Recipient may withhold or account for Tax-Related Items by considering statutory withholding rates or other applicable withholding rates, including minimum or maximum rates applicable in the Participant’s jurisdiction(s). In the event of over-withholding or if the maximum applicable rate for the Participant’s jurisdiction is used in connection with the withholding methods described in (c) or (d) above, the Participant may receive a refund of any over-withheld amount in cash (with no entitlement to the equivalent amount in Shares), or if not refunded, the Participant may be able to seek a refund from the applicable tax authorities. In the event of under-withholding, the Participant may be required to pay additional Tax-Related Items directly to the applicable tax authorities or to the Company and/or the Service Recipient. If any applicable withholding obligation for Tax-Related Items is
satisfied by withholding in Shares, for tax purposes, the Participant will be deemed to have been issued the full number of Shares subject to the vested RSUs, notwithstanding that a number of the Shares is held back solely for the purpose of paying the Tax-Related Items. The Participant agrees to pay to the Company or the Service Recipient any amount of Tax-Related Items that the Company or the Service Recipient may be required to withhold or account for as a result of the Participant’s participation in the Plan that cannot be satisfied by the means previously described. The Company may refuse to deliver the Shares or the proceeds of the sale of Shares, if the Participant fails to comply with the Participant’s obligations for Tax-Related Items.
(ii)Tax Consequences. The Participant has reviewed with his or her own tax advisors the applicable tax consequences of this investment and the transactions contemplated by this Award Agreement. With respect to such matters, the Participant relies solely on such advisors and not on any statements or representations of the Company or any of its agents, written or oral.
Section 3.5Restricted Stock Units Non-Transferable. Except to the limited extent provided in Section 2, this Award and the rights and privileges conferred hereby will not be transferred, assigned, pledged or hypothecated in any way (whether by operation of law or otherwise) and will not be subject to sale under execution, attachment or similar process. Upon any attempt to transfer, assign, pledge, hypothecate or otherwise dispose of this Award, or any right or privilege conferred hereby, or upon any attempted sale under any execution, attachment or similar process, this Award and the rights and privileges conferred hereby immediately will become null and void.
Section 3.6Unfunded and Unsecured Plan. The Participant acknowledges that the Plan is unfunded and the Company’s obligations under the Plan and this Award Agreement are unsecured and that, to the extent the Participant or his or her estate holds any rights by virtue of a grant of RSUs, such rights shall be no greater than the rights of an unsecured creditor of the Company.
Section 3.7No Stockholder Rights. Under no circumstances shall RSUs be considered Shares or other securities of the Company, nor shall they entitle the Participant to exercise voting rights, to receive dividends or other distributions or credit therefor, or to exercise or receive any other rights attaching to the ownership of Shares or other securities of the Company, nor shall the Participant be considered the owner of Shares by virtue of the award of RSUs.
Section 3.8No Guarantee of Continued Employment. THE PARTICIPANT ACKNOWLEDGES AND AGREES THAT THE VESTING OF THE RESTRICTED STOCK UNITS PURSUANT TO THE VESTING SCHEDULE HEREOF IS EARNED ONLY BY CONTINUING AS AN EMPLOYEE OR SERVICE PROVIDER AT THE WILL OF THE COMPANY (OR, IF DIFFERENT, THE SERVICE RECIPIENT) AND NOT THROUGH THE ACT OF BEING HIRED, BEING GRANTED THIS AWARD OF RESTRICTED STOCK UNITS OR, AS APPLICABLE, ACQUIRING SHARES HEREUNDER. THE PARTICIPANT FURTHER ACKNOWLEDGES AND AGREES THAT THIS AWARD AGREEMENT, THE TRANSACTIONS CONTEMPLATED HEREUNDER AND THE VESTING SCHEDULE SET FORTH IN THE NOTICE OF GRANT OF AWARD DO NOT CONSTITUTE AN EXPRESS OR IMPLIED PROMISE OF CONTINUED ENGAGEMENT AS AN EMPLOYEE OR SERVICE PROVIDER FOR THE VESTING PERIOD, FOR ANY PERIOD, OR AT ALL, AND SHALL NOT INTERFERE IN ANY WAY WITH THE PARTICIPANT’S RIGHT OR THE RIGHT OF THE COMPANY (OR SUBSIDIARY EMPLOYING THE PARTICIPANT) TO TERMINATE THE PARTICIPANT’S EMPLOYMENT OR SERVICE RELATIONSHIP AT ANY TIME, WITH OR WITHOUT CAUSE.
Section 3.9Notices. Any notice to be given under the terms of this Award Agreement to the Company shall be addressed to the Company in care of its Secretary, and any notice to be given to the Participant shall be addressed to him at the address given beneath his signature hereto. By a notice given pursuant to this Section 3.9, either party may hereafter designate a different address for notices to be given to it or him. Any notice which is required to be given to the Participant shall, if the Participant is then deceased, be given to the Participant’s personal representative if such representative has previously informed the Company of his status and address by written notice under this Section 3.9. Any notice shall have been deemed duly given when enclosed in a properly sealed envelope or wrapper addressed as aforesaid, deposited (with postage prepaid) in a post office or branch post office regularly maintained by the U.S. Postal Service or comparable foreign postal service.
Section 3.10Compliance with Laws. Notwithstanding any other provision of the Plan or this Award Agreement, unless there is an available exemption from any registration, qualification or other legal requirement applicable to the Shares, the Company shall not be required to permit the issuance and/or deliver any Shares prior to the completion of any registration or qualification of the Shares under any U.S. or non-U.S. local, state or federal securities or exchange control law or under rulings or regulations of the U.S. Securities and Exchange Commission (“SEC”) or of any other governmental regulatory body, or prior to obtaining any approval or other clearance from any U.S. or non-U.S. local, state or federal governmental agency, which registration, qualification or approval the Company shall, in its absolute discretion, deem necessary or advisable. The Participant understands that the Company is under no obligation to register or qualify the Shares with the SEC or any state or non-U.S. securities commission or to seek approval or clearance from any governmental authority for the issuance or sale of the Shares subject to the RSUs. Further, the Participant agrees that the Company shall have unilateral authority to amend this Award Agreement without the Participant’s consent to the extent necessary to comply with securities or other laws applicable to issuance of the Shares subject to the RSUs.
Section 3.11Modifications to the Award Agreement. Modifications to this Award Agreement or the Plan can be made only in an express written contract executed by a duly authorized officer of the Company. Notwithstanding anything to the contrary in the Plan or this Award Agreement, the Company reserves the right to revise this Award Agreement as it deems necessary or advisable, in its sole discretion and without the consent of the Participant, to comply with Section 409A of the Code or to otherwise avoid imposition of any additional tax or income recognition under Section 409A of the Code prior to the actual issuance of Shares pursuant to this award of RSUs.
Section 3.12Amendment, Suspension or Termination of the Plan. By accepting this Award Agreement or RSUs, the Participant expressly warrants that he or she has received an award of RSUs under the Plan, and has received, read and understood a description of the Plan. The Participant understands that the Plan is discretionary in nature and may be amended, suspended or terminated by the Company at any time.
Section 3.13Governing Law and Venue. The laws of the State of Delaware shall govern the interpretation, validity, administration, enforcement and performance of the terms of this Award Agreement regardless of the law that might be applied under principles of conflicts of laws. For purposes of litigating any dispute that arises under this grant or this Award Agreement, the parties hereby submit to and consent to the jurisdiction of the State of New Jersey and agree that such litigation shall be conducted in the state and/or federal courts located in New Jersey, where this grant is made and/or to be performed.
Section 3.14Assignment. Rights and obligations of the Company under this Award Agreement may be assigned by the Company to a successor in the business of the Company, any
company resulting from any amalgamation, reorganization, combination, merger or arrangement of the Company, or any company acquiring all or substantially all of the assets or business of the Company.
Section 3.15Electronic Delivery and Acceptance. The Participant hereby consents to receive the Notice of Grant of Award and Award Agreement and any other documents related to this award or future awards by electronic delivery and to accept this or future awards through an on-line or electronic system established and maintained by the Company or another third-party designated by the Company. The Participant acknowledges that he/she has read, understands and agrees to the terms of the Notice of Grant of Award and Award Agreement. Further, if the Participant does not decline the Award by written notice to the Company no later than 60 days following the date of grant or such other date that may be communicated by the Company, the Company will automatically accept the Award, subject to all terms and conditions set forth in the Notice of Grant of Award and Award Agreement and the Plan, on the Participant’s behalf. If the Participant properly declines the Award, the Award will be cancelled and the Participant will not be entitled to any benefits from the Award nor any compensation or benefits in lieu of the cancelled Award.
Section 3.16Waivers. No waiver by either party of any breach of, or of compliance with, any condition or provision of this Award Agreement by the other party shall be considered a waiver of any other condition or provision or of the same condition or provision at another time.
Section 3.17Entire Agreement. The Notice of Grant of Award, this Award Agreement, the Covenants Agreement, and the Plan constitute the entire contract between the parties hereto with regard to the subject matter hereof. They supersede any other agreements, representations or understandings (whether oral or written and whether express or implied) which relate to the subject matter hereof. The Participant expressly warrants that he or she is not accepting this Award Agreement in reliance on any promises, representations, or inducements other than those contained herein.
Section 3.18Severability. The provisions of this Award Agreement are severable and if any one or more provisions are determined to be illegal or otherwise unenforceable, in whole or in part, the remaining provisions shall nevertheless be binding and enforceable.
Section 3.19Language. The Participant acknowledges that the Participant is sufficiently proficient in English or has consulted with an advisor who is sufficiently proficient in English so as to allow the Participant to understand the terms and conditions of this Award Agreement. If the Participant has received this Award Agreement, or any other document related to the Plan translated into a language other than English and if the meaning of the translated version is different than the English version, the English version will control.
Section 3.20Insider Trading and Market-abuse Laws. The Participant acknowledges that, depending on the Participant’s or his or her broker’s country of residence or where the Shares are listed, the Participant may be subject to insider-trading restrictions and/or market-abuse laws, which may affect the Participant’s ability to accept, acquire, sell or otherwise dispose of Shares, rights to Shares (e.g., the RSUs), or rights linked to the value of Shares (e.g., phantom awards, futures) during such times as the Participant is considered to have “inside information” regarding the Company (as defined by law or regulations in the Participant’s country). Local insider trading laws and regulations may prohibit the cancellation or amendment of orders the Participant placed before the Participant possessed inside information. Furthermore, the Participant could be prohibited from (i) disclosing the inside information to any third party (other than on a “need to know” basis) and (ii) “tipping” third parties, including
fellow employees, or causing them otherwise to buy or sell securities. Any restrictions under these laws or regulations are separate from and in addition to any restrictions that may be imposed under the Company’s insider-trading policy. The Participant understands that it is the Participant’s responsibility to comply with any applicable restrictions as well as any Company insider trading policy, and the Participant should consult his or her personal legal advisor on this matter.
Section 3.21Imposition of Other Requirements. The Company reserves the right to impose other requirements on the Participant’s participation in the Plan, on this award of RSUs and on any Shares received from the RSUs, to the extent the Company determines it is necessary or advisable for legal or administrative reasons, and to require the Participant to sign any additional agreements or undertakings that may be necessary to accomplish the foregoing.
[SIGNATURE PAGE FOLLOWS]
IN WITNESS HEREOF, this Award Agreement has been executed and delivered by the parties hereto.
THE PARTICIPANT Electronic signature to be provided and recorded via online grant acceptance process on www.etrade.com | INTEGRA LIFESCIENCES HOLDINGS CORPORATION By Name: Title: | ||||
APPENDIX A
[This appendix has been omitted pursuant to Item 601(a)(5) of Regulation S-K under the Securities Act of 1933, as amended. The registrant agrees to furnish supplementally a copy of the omitted annex to the Securities and Exchange Commission upon request.]
IART-2025.12.31-EX-10.3(g)
PERFORMANCE STOCK AGREEMENT
THIS PERFORMANCE STOCK AGREEMENT (the “Award Agreement”), dated as of [ ] (the “Award Date”), is made by and between Integra LifeSciences Holdings Corporation, a Delaware corporation (the “Company”), and [ ], an employee of the Company (or one or more of its Related Corporations or Affiliates), hereinafter referred to as the “Participant.”
WHEREAS, the Company has determined to grant to the Participant an award of Performance Stock (as defined below), on the terms set forth herein, under the Integra LifeSciences Holdings Corporation Fifth Amended and Restated 2003 Equity Incentive Plan, as amended (the “Plan”), the terms of which are hereby incorporated by reference and made part of this Award Agreement.
NOW, THEREFORE, in consideration of the various covenants herein contained, and intending to be legally bound hereby, the parties hereto agree as follows:
ARTICLE I.DEFINITIONS
Capitalized terms not otherwise defined below shall have the meaning set forth in the Plan. The masculine pronoun shall include the feminine and neuter, and the singular the plural, where the context so indicates.
Section 1.1Adjusted EBITDA. “Adjusted EBITDA” shall mean GAAP net income excluding: (i) depreciation and amortization; (ii) other income (expense); (iii) interest income and expense; (iv) income tax expense (benefit); (v) impairment charges; and (vi) those operating expenses also excluded from adjusted net income.
Section 1.2Annual Adjusted EBITDA Margin. “Annual Adjusted EBITDA Margin” shall mean, with respect to an applicable fiscal year, the percentage obtained by dividing Adjusted EBITDA for such applicable fiscal year by the Company’s GAAP Revenue for the same period and expressing the result as a percentage. All calculations shall be made in accordance with the Company’s accounting principles, policies, and practices in effect as of the beginning of the Plan Year, unless otherwise determined by the Committee. Determination of the Annual Adjusted EBITDA Margin for each applicable Performance Period shall be subject to the sole discretion of the Company.
Section 1.3Annual Organic Revenue. “Annual Organic Revenue” shall mean the Company’s gross revenue with respect to an applicable fiscal year excluding the effects of currency exchange rates, acquired revenues, product discontinuances and divestitures.
Section 1.4Catch-Up Performance Goal. “Catch-Up Performance Goal” shall mean the specific goal determined by the Committee, as specified in Exhibit A.
Section 1.5Catch-Up Shares. “Catch-Up Shares” shall have the meaning as specified in Exhibit A.
Section 1.6Change in Control. “Change in Control” shall have the meaning set forth in the Plan.
A-1
Section 1.7Chief Human Resources Officer. “Chief Human Resources Officer” shall mean the Chief Human Resources Officer of the Company.
Section 1.8Covenants Agreement. “Covenants Agreement” shall mean the Confidentiality and Invention Disclosure and Non-Compete Agreement by and between the Company and the Participant, as set forth in Appendix A hereto, which shall be deemed executed in connection with and as appropriate consideration for benefits provided in this Award Agreement.
Section 1.9Good Reason. “Good Reason” shall have the meaning set forth in the Plan.
Section 1.10Performance Goals. “Performance Goals” shall mean the specific goal or goals determined by the Committee, as specified in Exhibit A, including (if applicable) the Catch-Up Performance Goal.
Section 1.11Performance Period. “Performance Period” shall mean the period or periods of time that the Performance Goals must be met, as specified in Exhibit A.
Section 1.12Performance-Vest. “Performance-Vest” shall mean that, with respect to a share of Performance Stock, the applicable Performance Goals have been achieved.
Section 1.13Performance Vesting Percentage. “Performance Vesting Percentage” shall mean the percentage determined in accordance with Exhibit A attached hereto, which is a function of whether and to what extent the Performance Goals are achieved during the Performance Period.
Section 1.14Qualifying Termination. “Qualifying Termination” shall mean a Termination of Service by the Company without Cause or by the Participant for Good Reason.
Section 1.15Rule 16b-3. “Rule 16b-3” shall mean that certain Rule 16b-3 under the Exchange Act, as such Rule may be amended from time to time.
Section 1.16Termination of Service. “Termination of Service” shall mean the time when the Participant ceases to provide services to the Company and its Related Corporations and Affiliates as an employee or Associate for any reason with or without Cause, including, but not by way of limitation, a termination by resignation, discharge, death, or Disability. A Termination of Service shall not include a termination where the Participant is simultaneously reemployed by, or remains employed by, or continues to provide services to, the Company and/or one or more of its Related Corporations and Affiliates or a successor entity thereto.
Section 1.17Vest or Vested. “Vest” or “Vested” shall mean that, with respect to a share of Performance Stock, both (i) such share of Performance Stock has Performance-Vested and (ii) the continued service condition has been satisfied.
ARTICLE IIAWARD OF PERFORMANCE STOCK
Section 2.1Award of Shares of Performance Stock. Effective as of the Award Date, the Company grants to the Participant an award of [ ] target shares of Performance Stock (the “Target Performance Shares”). Each share of Performance Stock represents the Participant’s right to receive one Share under this Award Agreement if the Performance Goals are met during the Performance Period and the vesting conditions set forth herein are satisfied.
2
Section 2.2Forfeiture. Shares of Performance Stock shall be subject to forfeiture as provided in Section 3.2 below.
Section 2.3Dividend Equivalents. The Participant shall be entitled to receive, with respect to each outstanding Vested but unissued share of Performance Stock, dividend equivalent amounts equal to the regular quarterly cash dividend paid or made with respect to the Shares underlying such Vested but unissued shares of Performance Stock (to the extent regular quarterly cash dividends are paid). Such dividend equivalent amounts shall be aggregated and paid to the Participant within thirty (30) days following the date on which the Shares underlying the Vested shares of Performance Stock are issued to the Participant, but in no event later than December 31 of the year in which the Shares underlying the Vested shares of Performance Stock are issued to the Participant. Notwithstanding the foregoing, if a “Change in Control” occurs prior to the date on which such dividend equivalent amounts are paid, such dividend equivalent amounts shall be paid to the Participant on the date of the Change in Control; provided, however, that such payment shall only occur if the Change in Control meets the requirements of Section 409A(a)(2)(A)(v) of the Internal Revenue Code of 1986, as amended (the “Code”) and its corresponding regulations. For the avoidance of doubt, such dividend equivalent amounts shall only be paid to the extent that the shares of Performance Stock are Vested as of the applicable dividend payment date, and the Participant shall not be entitled to receive any dividend equivalent amounts with respect to shares of Performance Stock that have not Vested as of such dividend payment date. The dividend equivalents and any amounts that may become payable in respect thereof shall be treated separately from the shares of Performance Stock and the rights arising in connection therewith for purposes of the designation of time and form of payments required by Code Section 409A.
Section 2.4Voting Rights. The Participant shall not have any voting rights in respect of the shares of Performance Stock and any Shares underlying the shares of Performance Stock unless and until such Shares shall have been issued by the Company and the Participant becomes the holder of record of such Shares (as evidenced by the appropriate entry on the books of the Company or of a duly authorized transfer agent of the Company).
ARTICLE IIIRESTRICTIONS
Section 3.1Vesting.
(a)Subject to paragraph (b) below and Sections 3.2 and 3.5 below, shares of Performance Stock shall Vest in cumulative installments as follows:
(i)With respect to fiscal year [ ], a number of shares of Performance Stock equal to the product of (x) thirty-three percent (33%) of the Target Performance Shares, multiplied by (y) the applicable Performance Vesting Percentage determined in accordance with Exhibit A attached hereto, shall Vest on the first anniversary of the Award Date;
(ii)With respect to fiscal year [ ], a number of shares of Performance Stock equal to the product of (x) thirty-three percent (33%) of the Target Performance Shares, multiplied by (y) the applicable Performance Vesting Percentage determined in accordance with Exhibit A attached hereto, shall Vest on the second anniversary of the Award Date; and
(iii)With respect to fiscal year [ ], a number of shares of Performance Stock equal to the product of (x) thirty-four percent (34%) of the Target Performance
3
Shares, multiplied by (y) the applicable Performance Vesting Percentage determined in accordance with Exhibit A attached hereto, shall Vest on the third anniversary of the Award Date.
(b) Subject to Sections 3.2 and 3.5 below, in the event that the Company achieves the Catch-Up Performance Goal with respect to the Performance Period, then any Catch-Up Shares shall Vest on the third anniversary of the Award Date.
Section 3.2Effect of Termination of Service; Forfeiture.
(a) In the event the Participant incurs, prior to or on the last day of the Performance Period, a Termination of Service by reason of the Participant’s Disability or death, any shares of Performance Stock which have not Vested in accordance with Section 3.1 above on or prior to such Termination of Service shall remain outstanding and eligible to Vest in accordance with Section 3.1 above and Section 3.5 below based on the Company’s achievement of the Performance Goals during the Performance Period.
(b) Immediately upon the Participant’s Termination of Service that is not either (i) a Qualifying Termination within twelve (12) months following the date of a Change in Control (and prior to or on the last day of the Performance Period) or (ii) a Termination of Service by reason of the Participant’s Disability or death, the Participant shall automatically and without further action forfeit all shares of Performance Stock (and all dividend equivalent rights with respect to such shares of Performance Stock) which have not Vested in accordance with Section 3.1 above or Section 3.5 below on or prior to such Termination of Service, and the Participant shall have no further right to or interest in or with respect to such shares of Performance Stock (or such dividend equivalents).
(c) Any shares of Performance Stock that do not Performance-Vest in connection with a Change in Control pursuant to Sections 3.5(a) and 3.5(b) below (and all dividend equivalent rights with respect to such shares of Performance Stock) shall thereupon automatically be forfeited as of such Change in Control, and the Participant shall have no further right to or interest in or with respect to such shares of Performance Stock (or such dividend equivalents).
(d) Any shares of Performance Stock that fail to vest as of the third anniversary of the Award Date (and all dividend equivalent rights with respect to such Performance Stock) shall automatically and without further action be cancelled and forfeited, and the Participant shall have no further right to or interest in or with respect to such unvested shares of Performance Stock (or such dividend equivalents).
Section 3.3Issuance of Shares.
(a) Subject to a determination of the Committee as to whether and to what extent the applicable Performance Goals have been met, Shares represented by shares of Performance Stock which Vest pursuant to Section 3.1 above or Section 3.5 below shall be issued to the Participant or his or her legal representative on or within five (5) business days following the
4
date on which such shares of Performance Stock Vest pursuant to Section 3.1 above or Section 3.5 below (but in no event later than December 31 of the applicable year in which such shares of Performance Stock Vest).
(b) All Shares issued hereunder shall be issued in certificated form or shall be recorded with the Company’s transfer agent. All such Shares shall be issued free from any restrictions; provided, however, that such Shares shall be subject to any restrictions and conditions as may be required pursuant to Section 4.6 below and those that the Company imposes on its employees in general with respect to selling its Shares. Notwithstanding the foregoing, the Company shall not be required to issue or record such Shares in the name of the Participant or his or her legal representative unless the Participant or his or her legal representative shall have satisfied the full amount of all federal, state and local withholding or other employment taxes applicable to the taxable income of the Participant resulting from the vesting of the shares of Performance Stock and issuance of the Shares as provided in this Award Agreement (including, without limitation, in the manner set forth in Section 4.3 below).
(c) As a condition precedent to the grant of the Target Performance Shares, the Participant hereby agrees to be subject to the confidentiality, inventions assignment, non-competition and other restrictive covenants set forth in the Covenants Agreement.
Section 3.4Clawback. Notwithstanding anything contained in the Plan or the Award Agreement to the contrary, the shares of Performance Stock, and any related payments, shall be subject to the provisions of any clawback, repayment or recapture policy implemented by the Company, including any such policy adopted to comply with applicable law (including without limitation the Dodd-Frank Wall Street Reform and Consumer Protection Act) or securities exchange listing standards and any rules or regulations promulgated thereunder, to the extent set forth in such policy and/or in any notice or agreement relating to the shares of Performance Stock under the Plan.
Section 3.5Change in Control. In the event that a Change in Control occurs during the Performance Period:
(a)A number of shares of Performance Stock shall Performance-Vest equal to a number determined at the greater of (i) the achievement of the “Target Level” Performance Vesting Percentage with respect to the fiscal year in which the Change in Control occurs, as specified in Exhibit A attached hereto and (ii) the Company’s actual achievement of the Performance Goal for such year through the Change in Control. Subject to Sections 3.5(d) and (e) below, such Performance-Vested shares of Performance Stock shall remain outstanding and eligible to Vest on the anniversary of the Award Date immediately following the Change in Control, subject to the Participant’s continuous service.
(b)In addition, and subject to Sections 3.5(d) and (e) below, a number of shares of Performance Stock shall Performance-Vest equal to the number of shares of Performance Stock that could vest with respect to each fiscal year of the Performance Period following the fiscal year in which the Change in Control occurs (if any) based on the achievement of the “Target Level” Performance Vesting Percentage with respect to each such year, as specified in Exhibit A, and shall remain outstanding and eligible to Vest on the date(s) outlined in Section 3.1(a)(ii) and/or (iii) (excluding any Catch-Up Shares which are forfeited in the event of a Change in Control), subject to the Participant’s continued service.
5
(c)In addition, if the Change in Control occurs following the completion of a fiscal year in the Performance Period but prior to the date on which shares of Performance Stock with respect to such year become Vested pursuant to Section 3.1(a) above, then such shares of Performance Stock shall Vest as of immediately prior to the Change in Control in a number determined in accordance with Section 3.1(a) above.
(d)If the Participant incurred a Termination of Service by reason of the Participant’s Disability or death, in either case, prior to the Change in Control date, then any shares of Performance Stock that Performance-Vest in accordance with Sections 3.5(a) and (b) above shall Vest as of immediately prior to the Change in Control.
(e) Notwithstanding Sections 3.5(a) and 3.5(b) above, if the Participant incurs (1) a Qualifying Termination on or within twelve (12) months following the date of a Change in Control and prior to or on the last day of the Performance Period, or (2) a Termination of Service by reason of the Participant’s Disability or death on or following a Change in Control and prior to or on the last day of the Performance Period, then in either case any Performance-Vested shares of Performance Stock that are then-outstanding and have not yet Vested shall Vest in full upon such Termination of Service.
ARTICLE IV MISCELLANEOUS
Section 4.1No Additional Rights. Nothing in this Award Agreement or in the Plan shall confer upon any person any right to a position as an Associate or continued employment by the Company or any of its Related Corporations or Affiliates or affect in any way the right of any of the foregoing to terminate the services of an individual at any time.
Section 4.2Anti-Assignment. The Participant shall have no right to sell, assign, transfer, pledge, or otherwise encumber or dispose of the Participant’s award of shares of Performance Stock.
Section 4.3Tax Withholding. In satisfaction of all applicable requirements with respect to amounts required by federal, state or local tax law to be withheld with respect to the vesting, distribution or payment of the shares of Performance Stock, the Company shall withhold Shares otherwise issuable upon such distribution or payment of the shares of Performance Stock having a Fair Market Value equal to the sums required to be withheld. Subject to the following sentence, the number of Shares which shall be so withheld in order to satisfy the Participant’s federal, state and local withholding tax liabilities with respect to the vesting of the shares of Performance Stock or issuance of Shares in payment of the shares of Performance Stock shall be limited to the number of Shares which have a Fair Market Value on the date of issuance equal to the aggregate amount of such liabilities based on the minimum statutory withholding rates for federal, state and local tax purposes that are applicable to, and required in connection with, all or a portion of such supplemental taxable income. In the event that the number of Shares having a Fair Market Value equal to the sums required to be withheld is not a whole number of Shares, the number of Shares so withheld shall be rounded up to the nearest whole share.
Section 4.4Notices. Any notice to be given under the terms of this Award Agreement to the Company shall be addressed to the Company in care of its Chief Human Resources Officer, and any notice to be given to the Participant shall be addressed to the Participant at his or her address of record maintained by the Human Resources Department. By a notice given pursuant to this Section 4.4, either party may hereafter designate a different address for notices to be given to it or him. Any notice which is required to be given to the Participant shall, if the Participant is then deceased, be given to the Participant’s personal representative if such
6
representative has previously informed the Company of his or her status and address by written notice under this Section 4.4. Any notice shall have been deemed duly given when enclosed in a properly sealed envelope or wrapper addressed as aforesaid, deposited (with postage prepaid) in a post office or branch post office regularly maintained by the United States Postal Service.
Section 4.5Titles. Titles are provided herein for convenience only and are not to serve as a basis for interpretation or construction of this Award Agreement.
Section 4.6Conformity to Securities Laws. This Award Agreement is intended to conform to the extent necessary with all provisions of the Securities Act and the Exchange Act and any and all regulations and rules promulgated by the Securities and Exchange Commission thereunder, including, without limitation, Rule 16b-3. Notwithstanding anything herein to the contrary, this Award Agreement shall be administered, and the shares of Performance Stock shall be issued, only in such a manner as to conform to such laws, rules and regulations. To the extent permitted by applicable law, this Award Agreement and the shares of Performance Stock issued hereunder shall be deemed amended to the extent necessary to conform to such laws, rules and regulations.
Section 4.7Amendment. This Award Agreement may be amended only by a writing executed by the parties hereto which specifically states that it is amending this Award Agreement.
Section 4.8Governing Law. The laws of the State of Delaware shall govern the interpretation, validity, administration, enforcement and performance of the terms of this Award Agreement regardless of the law that might be applied under principles of conflicts of laws.
Section 4.9Section 409A. This Award Agreement shall be interpreted in accordance with the requirements of Section 409A of the Code. Notwithstanding any provision in this Award Agreement to the contrary, if a payment is deemed to be deferred compensation subject to the requirements of Section 409A of the Code, such payment may only be made under this Award Agreement upon an event and in a manner permitted by Section 409A of the Code. If a payment is not made by the designated payment date under this Award Agreement, the payment shall be made by December 31 of the calendar year in which the designated date occurs. In no event may the Participant, directly or indirectly, designate the calendar year of payment. A termination of service shall not be deemed to have occurred for purposes of any provision of this Award Agreement providing for the payment of any amounts or benefits upon or following a termination of service that are considered “nonqualified deferred compensation” under Section 409A of the Code unless such termination is also a “separation from service” within the meaning of Section 409A of the Code and, for purposes of any such provision of this award Agreement, references to a “termination,” “Termination of Service” or like terms shall mean “separation from service.” Notwithstanding anything to the contrary in this Award Agreement, no amounts payable to the Participant under this Award Agreement shall be paid to the Participant prior to the expiration of the 6-month period following the Participant’s “separation from service” if the Company determines that paying such amounts at the time or times indicated in this Award Agreement would be a prohibited distribution under Section 409A(a)(2)(b)(i) of the Code. If the payment of any such amounts is delayed as a result of the previous sentence, then on the first day following the end of such 6-month period, the Company shall pay the Participant a lump-sum amount equal to the cumulative amount that would have otherwise been payable to the Participant during such 6-month period.
Section 4.10Electronic Delivery and Acceptance. Participant hereby consents to receive the Notice of Grant of Award and Award Agreement and any other documents related to this award or future awards by electronic delivery and to accept this or future awards through an on-line or electronic system established and maintained by the Company or another third party
7
designated by the Company. Participant acknowledges that he/she has read, understand and agrees to the terms of the Notice of Grant of Award, Award Agreement and Plan. By clicking the “ACCEPT” button on E*TRADE Morgan Stanley at Work’s on-line grant agreement response page, it will act as Participant’s electronic signature to these documents and will result in a contract between Integra LifeSciences Holdings Corporation and the Participant with respect to the award. In the event of any conflict or inconsistency between the terms and conditions of this Award Agreement or any other contracts or documents related to this award, on the one hand, and any terms or conditions set forth in the Plan, the terms and conditions set forth in the Plan shall prevail.
[Signature page follows]
8
IN WITNESS WHEREOF, the parties hereto have executed this Performance Stock Agreement as of the date first above written.
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
THE PARTICIPANT Electronic signature to be provided and recorded via online grant acceptance process on www.etrade.com | INTEGRA LIFESCIENCES HOLDINGS CORPORATION By Name: Title: | ||||
EXHIBIT A
PERFORMANCE GOALS AND PERFORMANCE PERIOD
Capitalized terms shall have the meaning set forth in Performance Stock Agreement.
The “Performance Period” shall be the three-year period beginning [ ] and ending [ ].
The “Catch-Up Performance Goal” shall mean that the Company achieves, as of the end of the Performance Period (but not due to a Change in Control), an average 3-year Annual Organic Revenue growth rate of at least [ ]% or an average 3-year Annual Adjusted EBITDA Margin of at least [ ]%.
With respect to each fiscal year in the Performance Period, the “Performance Goals” are that the Company achieves a Threshold Level or higher level of growth in Annual Organic Revenue over the immediately preceding fiscal year and achieves a Threshold Level or higher level of Annual Adjusted EBITDA Margin in the preceding fiscal year, as set forth in the tables below. A number of shares of Performance Stock will Performance-Vest in accordance with Section 3.1 of the Performance Stock Agreement based on the percentage growth in Annual Organic Revenue over the immediately preceding fiscal year and the Annual Adjusted EBITDA Margin in the preceding fiscal year. Each Performance Goal shall be weighted equally in the Performance Period, meaning 50% shall be attributed to Annual Organic Revenue over the immediately preceding fiscal year and 50% shall be attributed to Annual Adjusted EBITDA Margin in the preceding fiscal year. Notwithstanding the foregoing, for each Performance Period, the Company retains sole discretion to determine whether the applicable Performance Goals, and relatedly, the Participant’s Performance-Vest, have been achieved.
Growth in Annual Organic Revenue over the Prior Fiscal Year %:
Performance Year | Below Threshold | “Threshold Level” | “Target Level” | “Maximum” | ||||||||||
[ ] | [ ] | [ ] | [ ] | [ ] | ||||||||||
[ ] | [ ] | [ ] | [ ] | [ ] | ||||||||||
[ ] | [ ] | [ ] | [ ] | [ ] | ||||||||||
Performance Vesting Percentage | [ ] | [ ] | [ ] | [ ] | ||||||||||
Annual Adjusted EBITDA Margin in the preceding fiscal year (%):
Performance Year | Below Threshold | “Threshold Level” | “Target Level” | “Maximum” | ||||||||||
[ ] | [ ] | [ ] | [ ]% | [ ] | ||||||||||
[ ] | [ ] | [ ] | [ ]% | [ ] | ||||||||||
A-1
[ ] | [ ] | [ ] | [ ]% | [ ] | ||||||||||
Performance Vesting Percentage | [ ] | [ ] | [ ] | [ ] | ||||||||||
In the event that the growth in Annual Organic Revenue over the immediately preceding fiscal year falls between the “Threshold Level” and the “Target Level,” then the Performance Vesting Percentage shall be determined by means of linear interpolation between the “Threshold Level” and the “Target Level.” In the event that the growth in Annual Organic Revenue over the immediately preceding fiscal year falls between the “Target Level” and the “Maximum Level,” then the Performance Vesting Percentage shall be determined by means of linear interpolation between the “Target Level” and the “Maximum Level.”
In the event that the Annual Adjusted EBITDA Margin over the immediately preceding fiscal year falls between the “Threshold Level” and the “Target Level,” then the Performance Vesting Percentage shall be determined by means of linear interpolation between the “Threshold Level” and the “Target Level.” Annual Adjusted EBITDA Margin over the immediately preceding fiscal year falls between the “Target Level” and the “Maximum Level,” then the Performance Vesting Percentage shall be determined by means of linear interpolation between the “Target Level” and the “Maximum Level.”
Notwithstanding the forgoing, in the event that (i) a Change in Control does not occur during the Performance Period, (ii) the Performance Goal with respect to a given fiscal year in the Performance Period is not achieved at the applicable Target Level or higher, and (iii) the Catch-Up Performance Goal is achieved, then a number of shares of Performance Stock equal to the difference between (x) the number of shares of Performance Stock which would have Vested in the event that the Performance Goal had been achieved at the Target Level with respect to such fiscal year and (y) the number of shares of Performance Stock which actually became Vested based on the applicable Performance Vesting Percentage for such fiscal year, shall become Vested in accordance with Section 3.1(b) of the Performance Stock Agreement (such number of shares, the “Catch-Up Shares”). In no event shall the Catch-Up Shares exceed the Target number of shares granted.
A-2
APPENDIX A
[This appendix has been omitted pursuant to Item 601(a)(5) of Regulation S-K under the Securities Act of 1933, as amended. The registrant agrees to furnish supplementally a copy of the omitted annex to the Securities and Exchange Commission upon request.]
IART-2025.12.31-EX-10.3(h)
RESTRICTED STOCK UNIT AWARD AGREEMENT FOR DIRECTORS
THIS RESTRICTED STOCK UNIT AWARD AGREEMENT (the “Award Agreement”), dated as of [ ] (the “Award Date”), is made by and between Integra LifeSciences Holdings Corporation, a Delaware corporation (the “Company”), and [ ], a non-employee director of the Company, hereinafter referred to as the “Participant”:
WHEREAS, the Company maintains the Integra LifeSciences Holdings Corporation Fifth Amended and Restated 2003 Equity Incentive Plan, as amended (the “Plan”), and wishes to carry out the Plan, the terms of which are hereby incorporated by reference and made part of this Award Agreement; and
NOW, THEREFORE, in consideration of the various covenants herein contained, and intending to be legally bound hereby, the parties hereto agree as follows:
ARTICLE I.
DEFINITIONS
DEFINITIONS
Capitalized terms not otherwise defined below shall have the meaning set forth in the Plan. The masculine pronoun shall include the feminine and neuter, and the singular the plural, where the context so indicates.
Section 1.1“Award Date” shall have the meaning set forth in the recitals.
Section 1.2“Plan” shall have the meaning set forth in the recitals.
Section 1.3“Restricted Stock Units” or “RSUs” shall mean a conditional right to receive Shares pursuant to the terms of the Plan and this Award Agreement upon vesting and settlement, subject to the Participant’s continued service through the vesting date set forth in the Notice of Grant of Award, unless otherwise set forth in this Award Agreement.
Section 1.4“Secretary” shall mean the Secretary of the Company.
Section 1.5“Service Recipient” shall mean the Company, Related Corporation or Affiliate with which the Participant has an employment or service relationship.
Section 1.6“Termination Date” shall mean the date the Participant is no longer actively providing services to the Company, a Related Corporation or Affiliate (regardless of the reason for such termination and whether or not later found to be invalid or in breach of applicable laws in the jurisdiction where the Participant is rendering services or the terms of the Participant’s service agreement, if any) but excluding a termination where the Participant is simultaneously employed by, or continues to provide services to, the Company and/or one or more of its Related Corporations and Affiliates or a successor entity thereto. The Committee shall have the exclusive discretion to determine when the Participant’s service is terminated for purposes of this Award of RSUs (including when the Participant is no longer considered to be providing service while on a leave of absence).
ARTICLE II.
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
Section 2.1Grant of RSUs. The Company hereby grants [ ] RSUs to the Participant as of the Award Date, in accordance with and subject to the terms, conditions and restrictions of this Award Agreement and the Plan. RSUs shall be credited to an account maintained for the Participant on the books of the Company, as of the Award Date.
Section 2.2Vesting of RSUs. Subject to the terms and conditions of this Award Agreement and the Plan, the RSUs awarded hereunder shall vest in full on the first anniversary of the Award Date (the “Vesting Date”) provided the Participant remains an Associate or Key Employee as of the Vesting Date.
Section 2.3Termination of Service (other than on or Following a Change in Control). Upon the Participant’s termination of service prior to the Vesting Date, other than on or within twelve months following the date of a Change in Control and, except as otherwise determined by the Compensation Committee of the Company (the “Committee”), any RSUs granted to the Participant under this Award Agreement which have not yet vested on the Termination Date shall terminate without payment and shall be of no further force or effect from and after the Termination Date.
Section 2.4Death. Notwithstanding anything contained in this Award Agreement to the contrary, if a Participant dies prior to the Vesting Date, all RSUs held by the Participant immediately prior to death, which have not yet vested at the time of death, shall become fully vested and all forfeiture restrictions thereon shall lapse upon such death.
Section 2.5Change in Control. In the event that a Change in Control occurs and the Participant incurs a termination of service by the Company (which for purposes of this Award Agreement must qualify as a “separation from service” within the meaning of Section 409A) on or within twelve (12) months following the date of such Change in Control, the RSUs shall become fully vested and all forfeiture restrictions shall lapse upon such termination of service.
Section 2.6Acceleration of Vesting. Notwithstanding the provisions of Sections 2.2, 2.3, 2.4, and 2.5, the Committee may, in its sole discretion, at any time prior to or following the events contemplated in such Sections, permit the vesting of any or all RSUs held by the Participant and the issuance of Shares in respect of such RSUs in the manner and on the terms authorized by the Committee, provided that the Committee will not, in any case, authorize the vesting of an RSU or the issuance of a Share pursuant to this Section beyond the Vesting Date. If so accelerated, such RSUs will be considered as having vested as of the date specified by the Committee. The payment of Shares upon vesting pursuant to this Section 2.6 shall in all cases be paid at a time or in a manner that is exempt from, or complies with, Section 409A of the Code and the regulations promulgated thereunder (“Section 409A”). The prior sentence may be superseded in a future agreement or amendment to this Award Agreement only by direct and specific reference to such sentence. Notwithstanding anything in the Plan or this Award Agreement or any other agreement (whether entered into before, on or after the Award Date), if the vesting of the balance, or some lesser portion of the balance, of the RSUs is accelerated in connection with the Participant’s termination of service (provided that such termination is a “separation from service” within the meaning of Section 409A, as determined by the Company), other than due to the Participant’s death, and if (x) the Participant is a “specified employee” within the meaning of Section 409A at the time of such termination of service and (y) the payment of such accelerated RSUs will result in the imposition of additional tax under Section 409A if paid to the Participant on or within the six (6) month period following the Participant’s termination of service, then the payment of such accelerated RSUs will not be made until the date six (6) months and one (1) day following the date of Participant’s termination of service, unless the Participant dies following his or her termination of service, in which case, the RSUs will be paid in Shares to the Participant’s estate as soon as practicable following his or her death.
Section 2.7Settlement. On or as soon as administratively practicable (and in any event within 30 days) following the Vesting Date, the Company shall cause to be issued to the Participant Shares with respect to the RSUs that become vested on the Vesting Date or sooner pursuant to Section 2.4, 2.5 or 2.6.
Section 2.8Fractions. No fractional Share will be issued pursuant to an award granted hereunder. The number of Shares issuable to the Participant upon payment of any award granted under this Award Agreement will be rounded down to the nearest whole number of Share. No payment or other adjustment will be made with respect to the fractional Share so disregarded.
Section 2.9Section 409A. It is the intent of this Award Agreement and the Plan that this Award Agreement and the Plan and all payments and benefits to U.S. taxpayers thereunder be exempt from, or comply with, the requirements of Section 409A so that none of the RSUs provided under this Award Agreement or Shares issuable thereunder will be subject to the additional tax imposed under Section 409A, and any ambiguities herein will be interpreted in accordance with such intention. Each payment payable under this Award Agreement is intended to constitute a separate payment for purposes of Section 409A. However, in no event will the Company reimburse the Participant, or be otherwise responsible for, any taxes or costs that may be imposed on the Participant as a result of Section 409A. To the extent that any provision of the Award Agreement or the Plan would cause a conflict with the requirements of Section 409A of the Code or would cause the administration of the Award Agreement to fail to satisfy the requirements of Section 409A, such provision shall be deemed null and void to the extent permitted by applicable law. To the extent that the RSUs constitute “deferred compensation” under Section 409A, a termination of Service occurs only upon an event that would be a “separation from service” within the meaning of Section 409A. If, at the time of the Participant’s separation from service, (i) the Participant is a “specified employee” within the meaning of Section 409A, and (ii) the Company makes a good faith determination that an amount payable on account of the Participant’s separation from service constitutes deferred compensation (within the meaning of Section 409A), the payment of which is required to be delayed pursuant to the six (6)-month delay rule set forth in Section 409A to avoid taxes or penalties under Section 409A (the “Delay Period”), then the Company shall not pay such amount on the otherwise scheduled payment date but shall instead pay it in a lump sum on the first business day after the Delay Period (or upon the Participant’s death, if earlier), without interest. Each installment of RSUs that vest under this Award Agreement (if there is more than one installment) shall be considered one of a series of separate payments for purposes of Section 409A.
Section 2.10Adjustments to Restricted Stock Units. In the event of any subdivision, consolidation, stock dividend, capital reorganization, reclassification, exchange, or other change with respect to the Shares, or a consolidation, amalgamation, merger, spin-off, sale, lease or exchange of all or substantially all of the property of the Company or other distribution of the Company's assets to stockholders (other than the payment of ordinary cash dividends), the account of the Participant and the RSUs held by the Participant shall be adjusted in such manner, if any, as the Committee may in its discretion deem appropriate to preserve, proportionally, the interests of the Participant under the Plan.
Section 2.11Forfeiture/Clawback. Notwithstanding anything contained in the Plan or the Award Agreement to the contrary, the RSUs shall be subject to the provisions of any clawback, repayment, recapture or recoupment policy implemented by the Company, including any such policy(ies) adopted to comply with applicable law (including without limitation the Dodd-Frank Wall Street Reform and Consumer Protection Act and Section 10D of the Securities Exchange Act of 1934, as amended), governmental regulation or securities exchange listing standards and any rules or regulations promulgated thereunder, to the extent set forth in such
policy and/or in any notice or agreement relating to the RSUs under the Plan. The Company may reduce, cancel, or withhold against the RSUs or any other outstanding unvested or vested cash or equity based compensation owed or due to the Grantee, in each case, to the fullest extent permitted by applicable law in order to enforce the terms of any such clawback policy(ies).
ARTICLE III.
ADDITIONAL TERMS AND CONDITIONS
ADDITIONAL TERMS AND CONDITIONS
Section 3.1Status of Plan. The terms of the Plan are incorporated by reference into, and made part of, this Award Agreement. In the event of a conflict between the Plan and this Award Agreement, the terms of the Plan shall govern.
Section 3.2Committee’s Authority. The Committee shall have the power to interpret the Plan and this Award Agreement and to adopt such rules for the administration, interpretation and application of the Plan as are consistent therewith and to interpret or revoke any such rules (including, but not limited to, the determination of whether or not any RSUs have vested). All actions taken and all interpretations and determinations made by the Committee in good faith shall be final and binding upon the Participant, the Company and all other interested persons. The Committee shall not be personally liable for any action, determination or interpretation made in good faith with respect to the Plan or this Award Agreement. The Committee shall, in its absolute discretion, determine when such conditions have been fulfilled.
Section 3.3Nature of Grant. By accepting the grant of the RSUs, the Participant acknowledges, understands and agrees that:
(i)the Plan is established voluntarily by the Company, is discretionary in nature and may be modified, amended, suspended or terminated by the Company at any time to the extent permitted in the Plan;
(ii)the grant of the RSUs is exceptional voluntary and occasional and does not create any contractual or other right to receive future grants of RSUs or benefits in lieu of RSUs, even if RSUs have been awarded in the past;
(iii)all decisions with respect to future grants of RSUs, if any, will be at the sole discretion of the Company;
(iv)the Participant is voluntarily participating in the Plan;
(v)except to the extent explicitly and minimally required under applicable legislation, no Affiliate or Related Corporation (including, but not limited to, the Service Recipient) has any obligation to make any payment of any kind to the Participant under this Award Agreement;
(vi)the grant of the RSUs and any Shares subject to the RSUs, and the income from and value of same, are not intended to replace any pension rights or compensation;
(vii)unless otherwise agreed with the Company in writing, the RSUs and the Shares subject to the RSUs, and the income from and value of same, are not granted as consideration for, or in connection with, any service the Participant may provide as a director of a Related Corporation or an Affiliate;
(viii)except to the extent explicitly and minimally required under applicable legislation, the award of the RSUs and the Shares subject to the RSUs, and the income from and value of same are not part of normal or expected compensation or salary for any purposes,
including, but not limited to, calculating any severance, resignation, termination, end of service payments, bonuses, retirement or welfare benefits or similar payments and in no event should be considered as compensation for, or relating in any way to, past services for the Company or any Related Corporation or Affiliate;
(ix)the future value of the Shares underlying the RSUs is unknown, indeterminable and cannot be predicted with certainty;
(x)except to the extent explicitly and minimally required under applicable legislation, no claim or entitlement to compensation or damages shall arise from forfeiture of the RSUs resulting from the Participant’s termination of employment, or other service relationship (for any reason whatsoever and whether or not later found to be invalid or in breach of employment laws in the jurisdiction where the Participant is employed or the terms of the Participant’s employment agreement, if any); and
(xi)the Company is not providing any tax, legal or financial advice, nor is the Company making any recommendations regarding the Participant’s participation in the Plan or sale of the Shares acquired upon vesting and settlement of the RSUs. The Participant should consult with his or her own personal tax, legal and financial advisors regarding his or her participation in the Plan before taking any action related to the Plan.
Section 3.4Responsibility for Taxes. The Participant acknowledges that, regardless of any action taken by the Company or, if different, the Service Recipient, the ultimate liability for all income tax, social insurance, payroll tax, fringe benefits tax, payment on account, and other tax-related items related to the Participant’s participation in the Plan and legally applicable to the Participant or deemed applicable to the Participant (“Tax-Related Items”) is and remains Participant’s responsibility. The Participant further acknowledges that the Company and/or the Service Recipient (i) make no representations or undertakings regarding the treatment of any Tax-Related Items in connection with any aspect of the RSUs or the underlying Shares, including, but not limited to, the grant, vesting or settlement of the RSUs, the subsequent sale of Shares acquired pursuant to such settlement and the receipt of any dividends or other distributions paid on the Shares, and (ii) do not commit to and are under no obligation to structure the terms of the grant or any aspect of the RSUs to reduce or eliminate the Participant’s liability for Tax-Related Items or achieve any particular tax result. Further, if the Participant is subject to Tax-Related Items in more than one jurisdiction, the Participant acknowledges that the Company and/or the Service Recipient (or former service recipient, as applicable) may be required to withhold or account for Tax-Related Items in more than one jurisdiction.
(i)Tax Withholding. The Company is not responsible for withholding with regard to the Tax-Related Items. However, the Company reserves the right to withhold any Tax-Related Items to the extent circumstances change and it is required to do so. Prior to any relevant taxable or tax withholding event, as applicable, the Participant agrees to make arrangements satisfactory to the Company and the Service Recipient to satisfy any applicable withholding obligations the Company or the Service Recipient may have for Tax-Related Items. In this regard, the Participant authorizes the Company and the Service Recipient, as applicable, and their respective agents, at their discretion, to satisfy any applicable withholding obligation for Tax-Related Items by one or a combination of the following:
(a)withholding from wages or other cash compensation payable to the Participant by the Company or any Service Recipient;
(b)requiring the Participant to tender a cash payment or make a payment in a form acceptable to the Company or the Service Recipient;
(c)withholding from proceeds of the sale of Shares to be issued upon vesting of the RSUs either through a voluntary sale or through a mandatory sale arranged by the Company (on the Participant’s behalf pursuant to this authorization without further consent);
(d)withholding in Shares to be issued upon vesting of the RSUs (in which case the Participant will be deemed to have been issued the full number of Shares subject to the vested portion of the RSUs, notwithstanding that a number of the Shares are held back solely for the purpose of paying the Tax-Related Items); and
(e)any other method acceptable to the Company and to the extent required under the Plan and applicable laws, approved by the Committee.
The Company and/or the Service Recipient may withhold or account for Tax-Related Items by considering statutory withholding rates or other applicable withholding rates, including minimum or maximum rates applicable in the Participant’s jurisdiction(s). In the event of over-withholding or if the maximum applicable rate for the Participant’s jurisdiction is used in connection with the withholding methods described in (c) or (d) above, the Participant may receive a refund of any over-withheld amount in cash (with no entitlement to the equivalent amount in Shares), or if not refunded, the Participant may be able to seek a refund from the applicable tax authorities. In the event of under-withholding, the Participant may be required to pay additional Tax-Related Items directly to the applicable tax authorities or to the Company and/or the Service Recipient. If any applicable withholding obligation for Tax-Related Items is satisfied by withholding in Shares, for tax purposes, the Participant will be deemed to have been issued the full number of Shares subject to the vested RSUs, notwithstanding that a number of the Shares is held back solely for the purpose of paying the Tax-Related Items. The Participant agrees to pay to the Company or the Service Recipient any amount of Tax-Related Items that the Company or the Service Recipient may be required to withhold or account for as a result of the Participant’s participation in the Plan that cannot be satisfied by the means previously described. The Company may refuse to deliver the Shares or the proceeds of the sale of Shares, if the Participant fails to comply with the Participant’s obligations for Tax-Related Items.
(ii)Tax Consequences. The Participant has reviewed with his or her own tax advisors the applicable tax consequences of this investment and the transactions contemplated by this Award Agreement. With respect to such matters, the Participant relies solely on such advisors and not on any statements or representations of the Company or any of its agents, written or oral.
Section 3.5Restricted Stock Units Non-Transferable. Except to the limited extent provided in Section 2, this Award and the rights and privileges conferred hereby will not be transferred, assigned, pledged or hypothecated in any way (whether by operation of law or otherwise) and will not be subject to sale under execution, attachment or similar process. Upon any attempt to transfer, assign, pledge, hypothecate or otherwise dispose of this Award, or any right or privilege conferred hereby, or upon any attempted sale under any execution, attachment or similar process, this Award and the rights and privileges conferred hereby immediately will become null and void.
Section 3.6Unfunded and Unsecured Plan. The Participant acknowledges that the Plan is unfunded and the Company’s obligations under the Plan and this Award Agreement are unsecured and that, to the extent the Participant or his or her estate holds any rights by virtue of a grant of RSUs, such rights shall be no greater than the rights of an unsecured creditor of the Company.
Section 3.7No Stockholder Rights. Under no circumstances shall RSUs be considered Shares or other securities of the Company, nor shall they entitle the Participant to
exercise voting rights, to receive dividends or other distributions or credit therefor, or to exercise or receive any other rights attaching to the ownership of Shares or other securities of the Company, nor shall the Participant be considered the owner of Shares by virtue of the award of RSUs.
Section 3.8No Guarantee of Continued Service. THE PARTICIPANT ACKNOWLEDGES AND AGREES THAT THE VESTING OF THE RESTRICTED STOCK UNITS PURSUANT TO THE VESTING SCHEDULE HEREOF IS EARNED ONLY BY CONTINUING AS A SERVICE PROVIDER AT THE WILL OF THE COMPANY (OR, IF DIFFERENT, THE SERVICE RECIPIENT) AND NOT THROUGH THE ACT OF BEING GRANTED THIS AWARD OF RESTRICTED STOCK UNITS OR, AS APPLICABLE, ACQUIRING SHARES HEREUNDER. THE PARTICIPANT FURTHER ACKNOWLEDGES AND AGREES THAT THIS AWARD AGREEMENT, THE TRANSACTIONS CONTEMPLATED HEREUNDER AND THE VESTING SCHEDULE SET FORTH IN THE NOTICE OF GRANT OF AWARD DO NOT CONSTITUTE AN EXPRESS OR IMPLIED PROMISE OF CONTINUED ENGAGEMENT AS A SERVICE PROVIDER FOR THE VESTING PERIOD, FOR ANY PERIOD, OR AT ALL, AND SHALL NOT INTERFERE IN ANY WAY WITH THE PARTICIPANT’S RIGHT OR THE RIGHT OF THE COMPANY (OR SUBSIDIARY IF APPLICABLE) TO TERMINATE THE PARTICIPANT’S SERVICE RELATIONSHIP AT ANY TIME, WITH OR WITHOUT CAUSE.
Section 3.9Notices. Any notice to be given under the terms of this Award Agreement to the Company shall be addressed to the Company in care of its Secretary, and any notice to be given to the Participant shall be addressed to him at the address given beneath his signature hereto. By a notice given pursuant to this Section 3.9, either party may hereafter designate a different address for notices to be given to it or him. Any notice which is required to be given to the Participant shall, if the Participant is then deceased, be given to the Participant’s personal representative if such representative has previously informed the Company of his status and address by written notice under this Section 3.9. Any notice shall have been deemed duly given when enclosed in a properly sealed envelope or wrapper addressed as aforesaid, deposited (with postage prepaid) in a post office or branch post office regularly maintained by the U.S. Postal Service or comparable foreign postal service.
Section 3.10Compliance with Laws. Notwithstanding any other provision of the Plan or this Award Agreement, unless there is an available exemption from any registration, qualification or other legal requirement applicable to the Shares, the Company shall not be required to permit the issuance and/or deliver any Shares prior to the completion of any registration or qualification of the Shares under any U.S. or non-U.S. local, state or federal securities or exchange control law or under rulings or regulations of the Securities and Exchange Commission (“SEC”) or of any other governmental regulatory body, or prior to obtaining any approval or other clearance from any U.S. or non-U.S. local, state or federal governmental agency, which registration, qualification or approval the Company shall, in its absolute discretion, deem necessary or advisable. The Participant understands that the Company is under no obligation to register or qualify the Shares with the SEC or any state or non-U.S. securities commission or to seek approval or clearance from any governmental authority for the issuance or sale of the Shares subject to the RSUs. Further, the Participant agrees that the Company shall have unilateral authority to amend this Award Agreement without the Participant’s consent to the extent necessary to comply with securities or other laws applicable to issuance of the Shares subject to the RSUs.
Section 3.11Modifications to the Award Agreement. Modifications to this Award Agreement or the Plan can be made only in an express written contract executed by a duly authorized officer of the Company. Notwithstanding anything to the contrary in the Plan or this
Award Agreement, the Company reserves the right to revise this Award Agreement as it deems necessary or advisable, in its sole discretion and without the consent of the Participant, to comply with Section 409A of the Code or to otherwise avoid imposition of any additional tax or income recognition under Section 409A of the Code prior to the actual issuance of Shares pursuant to this award of RSUs.
Section 3.12Amendment, Suspension or Termination of the Plan. By accepting this Award Agreement or RSUs, the Participant expressly warrants that he or she has received an award of RSUs under the Plan, and has received, read and understood a description of the Plan. The Participant understands that the Plan is discretionary in nature and may be amended, suspended or terminated by the Company at any time.
Section 3.13Governing Law and Venue. The laws of the State of Delaware shall govern the interpretation, validity, administration, enforcement and performance of the terms of this Award Agreement regardless of the law that might be applied under principles of conflicts of laws. For purposes of litigating any dispute that arises under this grant or this Award Agreement, the parties hereby submit to and consent to the jurisdiction of the State of New Jersey and agree that such litigation shall be conducted in the state and/or federal courts located in New Jersey, where this grant is made and/or to be performed.
Section 3.14Assignment. Rights and obligations of the Company under this Award Agreement may be assigned by the Company to a successor in the business of the Company, any company resulting from any amalgamation, reorganization, combination, merger or arrangement of the Company, or any company acquiring all or substantially all of the assets or business of the Company.
Section 3.15Electronic Delivery and Acceptance. The Participant hereby consents to receive the Notice of Grant of Award and Award Agreement and any other documents related to this award or future awards by electronic delivery and to accept this or future awards through an on-line or electronic system established and maintained by the Company or another third-party designated by the Company. The Participant acknowledges that he/she has read, understands and agrees to the terms of the Notice of Grant of Award and Award Agreement. Further, if the Participant does not decline the Award by written notice to the Company no later than 60 days following the date of grant or such other date that may be communicated by the Company, the Company will automatically accept the Award, subject to all terms and conditions set forth in the Notice of Grant of Award and Award Agreement and the Plan, on the Participant’s behalf. If the Participant properly declines the Award, the Award will be cancelled and the Participant will not be entitled to any benefits from the Award nor any compensation or benefits in lieu of the cancelled Award.
Section 3.16Waivers. No waiver by either party of any breach of, or of compliance with, any condition or provision of this Award Agreement by the other party shall be considered a waiver of any other condition or provision or of the same condition or provision at another time.
Section 3.17Entire Agreement. The Notice of Grant of Award, this Award Agreement and the Plan constitute the entire contract between the parties hereto with regard to the subject matter hereof. They supersede any other agreements, representations or understandings (whether oral or written and whether express or implied) which relate to the subject matter hereof. The Participant expressly warrants that he or she is not accepting this Award Agreement in reliance on any promises, representations, or inducements other than those contained herein.
Section 3.18Severability. The provisions of this Award Agreement are severable and if any one or more provisions are determined to be illegal or otherwise unenforceable, in whole or in part, the remaining provisions shall nevertheless be binding and enforceable.
Section 3.19Language. The Participant acknowledges that the Participant is sufficiently proficient in English or has consulted with an advisor who is sufficiently proficient in English so as to allow the Participant to understand the terms and conditions of this Award Agreement. If the Participant has received this Award Agreement, or any other document related to the Plan translated into a language other than English and if the meaning of the translated version is different than the English version, the English version will control.
Section 3.20Insider Trading and Market-abuse Laws. The Participant acknowledges that, depending on the Participant’s or his or her broker’s country of residence or where the Shares are listed, the Participant may be subject to insider-trading restrictions and/or market-abuse laws, which may affect the Participant’s ability to accept, acquire, sell or otherwise dispose of Shares, rights to Shares (e.g., the RSUs), or rights linked to the value of Shares (e.g., phantom awards, futures) during such times as the Participant is considered to have “inside information” regarding the Company (as defined by law or regulations in the Participant’s country). Local insider trading laws and regulations may prohibit the cancellation or amendment of orders the Participant placed before the Participant possessed inside information. Furthermore, the Participant could be prohibited from (i) disclosing the inside information to any third party (other than on a “need to know” basis) and (ii) “tipping” third parties, including employees, or causing them otherwise to buy or sell securities. Any restrictions under these laws or regulations are separate from and in addition to any restrictions that may be imposed under the Company’s insider-trading policy. The Participant understands that it is the Participant’s responsibility to comply with any applicable restrictions as well as any Company insider trading policy, and the Participant should consult his or her personal legal advisor on this matter.
Section 3.21Imposition of Other Requirements. The Company reserves the right to impose other requirements on the Participant’s participation in the Plan, on this award of RSUs and on any Shares received from the RSUs, to the extent the Company determines it is necessary or advisable for legal or administrative reasons, and to require the Participant to sign any additional agreements or undertakings that may be necessary to accomplish the foregoing.
[SIGNATURE PAGE FOLLOWS]
IN WITNESS HEREOF, this Award Agreement has been executed and delivered by the parties hereto.
INTEGRA LIFESCIENCES
THE PARTICIPANT HOLDINGS CORPORATION
By
[ ] Name:
Title:
IART-2025.12.31-EX-10.3(p)
RESTRICTED STOCK UNIT AWARD AGREEMENT FOR PARTICIPANTS OUTSIDE THE UNITED STATES
THIS RESTRICTED STOCK UNIT AWARD AGREEMENT including any exhibit, appendix or addendum hereto (the “Award Agreement”), dated as of [ ] (the “Award Date”), is made by and between Integra LifeSciences Holdings Corporation, a Delaware corporation (the “Company”), and [ ], hereinafter referred to as the “Participant,” a Key Employee or Associate (as defined in the Plan).
WHEREAS, the Company has established and maintains the Integra LifeSciences Holdings Corporation Fifth Amended and Restated 2003 Equity Incentive Plan, as amended from time to time (the “Plan”);
WHEREAS the Company has determined that it would be to the advantage and best interest of the Company and its shareholders to grant an award of Restricted Stock Units (“RSUs”) provided for herein to the Participant as an incentive for increased efforts during the Participant’s employment with or services for the Company or its Related Corporations or Affiliates;
WHEREAS, Restricted Stock Units can be granted, and the shares of common stock of the Company (the “Shares”) subject to the RSUs can be issued, under Section 7.8 of the Plan; and
NOW, THEREFORE, in consideration of the various covenants herein contained, and intending to be legally bound hereby, the parties hereto agree as follows:
ARTICLE I.
DEFINITIONS
DEFINITIONS
Capitalized terms not otherwise defined below shall have the meaning set forth in the Plan. The masculine pronoun shall include the feminine and neuter, and the singular the plural, where the context so indicates.
Section 1.1“Award Date” shall have the meaning set forth in the recitals.
Section 1.2“Cause” shall mean, with respect to any Participant, “Cause” as defined in such Participant’s employment agreement or severance agreement with the Company if such an agreement exists and contains a definition of Cause or, if no such agreement exists or such agreement does not contain a definition of Cause, then Cause shall mean (i) the Participant’s neglect of duties or responsibilities that he or she is required to perform for the Company or any willful failure by the Participant to obey a lawful direction of the Board or the Company; (ii) the Participant’s engaging in any act of dishonesty, fraud, embezzlement, misrepresentation or other act of moral turpitude; (iii) the Participant’s knowing violation of any federal or state law or regulation applicable to the Company’s business; (iv) the Participant’s material breach of any confidentiality, non-compete agreement or invention assignment agreement or any other material agreement between the Participant and the Company; (v) the Participant’s conviction of, or plea of nolo contendere to, any felony or crime of moral turpitude which conviction or plea is materially and demonstrably injurious to the Company or any of its subsidiaries; (vi) failure by the Participant to comply with the Company’s material written
policies or rules; or (vii) the Participant’s act or omission in the course of his or her employment which constitutes gross negligence or willful misconduct.
Section 1.3“Plan” shall have the meaning set forth in the recitals.
Section 1.4“Restricted Stock Units” or “RSUs” shall mean a conditional right to receive Shares pursuant to the terms of the Plan and this Award Agreement upon vesting and settlement, subject to the Participant’s continued employment through each vesting date set forth in the Notice of Grant of Award, unless otherwise set forth in this Award Agreement.
Section 1.5“Rule 16b-3” shall mean that certain Rule 16b-3 under the U.S. Exchange Act, as such Rule may be amended from time to time.
Section 1.6“Secretary” shall mean the Secretary of the Company.
Section 1.7“Service Recipient” shall mean the Company, Related Corporation or Affiliate with which the Participant has an employment or service relationship.
Section 1.8“Termination Date” shall mean the date the Participant is no longer actively providing services to the Company, a Related Corporation or Affiliate (regardless of the reason for such termination and whether or not later found to be invalid or in breach of applicable laws in the jurisdiction where the Participant is employed or rendering services or the terms of the Participant’s employment or service agreement, if any) and, unless otherwise expressly provided for in this Award Agreement or determined by the Company, will not be extended by any notice period (e.g., the Participant’s period of employment or service will not include any contractual notice period or any period of “garden leave” or similar period mandated under applicable laws in the jurisdiction where the Participant is rendering services or the terms of the Participant’s employment or service agreement, if any). The Committee shall have the exclusive discretion to determine when the Participant’s service is terminated for purposes of this Award of RSUs (including when the Participant is no longer considered to be providing service while on a leave of absence).
ARTICLE II.
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
GRANT OF RESTRICTED STOCK UNITS AND ISSUANCE OF SHARES
Section 2.1Grant of RSUs. The Company hereby grants [ ] RSUs to the Participant as of the Award Date, in accordance with and subject to the terms, conditions and restrictions of this Award Agreement and the Plan. RSUs shall be credited to an account maintained for the Participant on the books of the Company, as of the Award Date.
Section 2.2Vesting of RSUs. Subject to the terms and conditions of this Award Agreement and the Plan, [ ]% of the RSUs awarded hereunder shall vest on [ ] and [ ]% will vest on [ ] (each a “Vesting Date”) provided the Participant remains a Key Employee or Associate from the Award Date until the Vesting Date.
Section 2.3Termination of Service (other than by (i) Death or Disability or (ii) a Qualifying Termination on or Following a Change in Control). Upon the Participant’s termination of service prior to the Vesting Date, other than by (i) death or Disability or (ii) a Qualifying Termination on or within twelve months following the date of a Change in Control and, except as otherwise determined by the Compensation Committee of the Company (the “Committee”), any RSUs granted to the Participant under this Award Agreement which have not yet vested on the Termination Date shall terminate without payment and shall be of no further force or effect from and after the Termination Date.
Section 2.4Death or Disability. If a Participant dies or suffers a Disability prior to the Vesting Date, all RSUs held by the Participant immediately prior to death or Disability, which have not yet vested at the time of death or Disability, shall become fully vested and all forfeiture restrictions thereon shall lapse upon such death or Disability.
Section 2.5Change in Control. In the event that a Change in Control occurs and the Participant incurs a Qualifying Termination on or within twelve (12) months following the date of such Change in Control, the RSUs shall become fully vested and all forfeiture restrictions shall lapse upon such Qualifying Termination.
Section 2.6Acceleration of Vesting. Notwithstanding the provisions of Sections 2.2, 2.3, 2.4, and 2.5, the Committee may, in its sole discretion, at any time prior to or following the events contemplated in such Sections, permit the vesting of any or all RSUs held by the Participant and the issuance of Shares in respect of such RSUs in the manner and on the terms authorized by the Committee, provided that the Committee will not, in any case, authorize the vesting of an RSU or the issuance of a Share pursuant to this Section beyond the Vesting Date. If so accelerated, such RSUs will be considered as having vested as of the date specified by the Committee. If the Participant is a U.S. taxpayer, the payment of Shares upon vesting pursuant to this Section 2.6 shall in all cases be paid at a time or in a manner that is exempt from, or complies with, Section 409A. The prior sentence may be superseded in a future agreement or amendment to this Award Agreement only by direct and specific reference to such sentence. Notwithstanding anything in the Plan or this Award Agreement or any other agreement (whether entered into before, on or after the Award Date), if the vesting of the balance, or some lesser portion of the balance, of the RSUs is accelerated in connection with the Participant’s termination of service (provided that such termination is a “separation from service” within the meaning of Section 409A, as determined by the Company), other than due to the Participant’s death, and if (x) the Participant is a U.S. taxpayer and a “specified employee” within the meaning of Section 409A at the time of such termination of service and (y) the payment of such accelerated RSUs will result in the imposition of additional tax under Section 409A if paid to the Participant on or within the six (6) month period following the Participant’s termination of service, then the payment of such accelerated RSUs will not be made until the date six (6) months and one (1) day following the date of Participant’s termination of service, unless the Participant dies following his or her termination of service, in which case, the RSUs will be paid in Shares to the Participant’s estate as soon as practicable following his or her death.
Section 2.7Settlement. On or as soon as administratively practicable (and any event within 30 days) following each Vesting Date, the Company shall cause to be issued to the Participant Shares with respect to the RSUs that become vested on such Vesting Date.
Section 2.8Fractions. No fractional Share will be issued pursuant to an award granted hereunder. The number of Shares issuable to the Participant upon payment of any award granted under this Award Agreement will be rounded down to the nearest whole number of Share. No payment or other adjustment will be made with respect to the fractional Share so disregarded.
Section 2.9Section 409A. It is the intent of this Award Agreement that it and all payments and benefits to U.S. taxpayers hereunder be exempt from, or comply with, the requirements of Section 409A so that none of the RSUs provided under this Award Agreement or Shares issuable thereunder will be subject to the additional tax imposed under Section 409A, and any ambiguities herein will be interpreted to be so exempt or so comply. Each payment payable under this Award Agreement is intended to constitute a separate payment for purposes of Treasury Regulation Section 1.409A-2(b)(2). However, in no event will the Company reimburse the Participant, or be otherwise responsible for, any taxes or costs that may be imposed on the Participant as a result of Section 409A.
Section 2.10Adjustments to Restricted Stock Units. In the event of any subdivision, consolidation, stock dividend, capital reorganization, reclassification, exchange, or other change with respect to the Shares, or a consolidation, amalgamation, merger, spin-off, sale, lease or exchange of all or substantially all of the property of the Company or other distribution of the Company's assets to stockholders (other than the payment of ordinary cash dividends), the account of the Participant and the RSUs held by the Participant shall be adjusted in such manner, if any, as the Committee may in its discretion deem appropriate to preserve, proportionally, the interests of the Participant under the Plan.
Section 2.11Forfeiture/Clawback. Notwithstanding anything contained in the Plan or the Award Agreement to the contrary, the RSUs shall be subject to the provisions of any clawback, repayment or recapture policy implemented by the Company, including any such policy adopted to comply with applicable law (including without limitation the Dodd-Frank Wall Street Reform and Consumer Protection Act) or securities exchange listing standards and any rules or regulations promulgated thereunder, to the extent set forth in such policy and/or in any notice or agreement relating to the RSUs under the Plan.
ARTICLE III.
ADDITIONAL TERMS AND CONDITIONS
ADDITIONAL TERMS AND CONDITIONS
Section 3.1Status of Plan. The terms of the Plan are incorporated by reference into, and made part of, this Award Agreement. In the event of a conflict between the Plan and this Award Agreement, the terms of the Plan shall govern.
Section 3.2Committee’s Authority. The Committee shall have the power to interpret the Plan and this Award Agreement and to adopt such rules for the administration, interpretation and application of the Plan as are consistent therewith and to interpret or revoke any such rules (including, but not limited to, the determination of whether or not any RSUs have vested). All actions taken and all interpretations and determinations made by the Committee in good faith shall be final and binding upon the Participant, the Company and all other interested persons. The Committee shall not be personally liable for any action, determination or interpretation made in good faith with respect to the Plan or this Award Agreement. The Committee shall, in its absolute discretion, determine when such conditions have been fulfilled.
Section 3.3Nature of Grant. By accepting the grant of the RSUs, the Participant acknowledges, understands and agrees that:
(i)the Plan is established voluntarily by the Company, is discretionary in nature and may be modified, amended, suspended or terminated by the Company at any time to the extent permitted in the Plan;
(ii)the grant of the RSUs is exceptional voluntary and occasional and does not create any contractual or other right to receive future grants of RSUs or benefits in lieu of RSUs, even if RSUs have been awarded in the past;
(iii)all decisions with respect to future grants of RSUs, if any, will be at the sole discretion of the Company;
(iv)the Participant is voluntarily participating in the Plan;
(v)the grant of the RSUs and any Shares subject to the RSUs, and the income from and value of same, are not intended to replace any pension rights or compensation;
(vi)unless otherwise agreed with the Company in writing, the RSUs and the Shares subject to the RSUs, and the income from and value of same, are not granted as consideration for, or in connection with, any service the Participant may provide as a director of a Related Corporation or an Affiliate;
(vii)the award of the RSUs and the Shares subject to the RSUs, and the income from and value of same are not part of normal or expected compensation or salary for any purposes, including, but not limited to, calculating any severance, resignation, termination, redundancy, end of service payments, bonuses, holiday pay, long-service awards, pension or retirement or welfare benefits or similar payments and in no event should be considered as compensation for, or relating in any way to, past services for the Company or any Related Corporation or Affiliate;
(viii)the future value of the Shares underlying the RSUs is unknown, indeterminable and cannot be predicted with certainty;
(ix)no claim or entitlement to compensation or damages shall arise from forfeiture of the RSUs resulting from the Participant’s termination of employment, or other service relationship (for any reason whatsoever and whether or not later found to be invalid or in breach of employment laws in the jurisdiction where the Participant is employed or the terms of the Participant’s employment agreement, if any);
(x)neither the Company, the Service Recipient nor any other Related Corporation or Affiliate shall be liable for any exchange rate fluctuation between the Participant’s local currency and the U.S. Dollar that may affect the value of the RSUs or of any amounts due to the Participant pursuant to the vesting and settlement of the RSUs or the subsequent sale of any Shares acquired upon settlement; and
(xi)the Company is not providing any tax, legal or financial advice, nor is the Company making any recommendations regarding the Participant’s participation in the Plan or sale of the Shares acquired upon vesting and settlement of the RSUs. The Participant should consult with his or her own personal tax, legal and financial advisors regarding his or her participation in the Plan before taking any action related to the Plan.
Section 3.4Responsibility for Taxes. The Participant acknowledges that, regardless of any action taken by the Company or, if different, the Service Recipient, the ultimate liability for all income tax, social insurance, payroll tax, fringe benefits tax, payment on account, and other tax-related items related to the Participant’s participation in the Plan and legally applicable to the Participant or deemed applicable to the Participant (“Tax-Related Items”) is and remains Participant’s responsibility and may exceed the amount, if any, actually withheld by the Company or the Service Recipient. The Participant further acknowledges that the Company and/or the Service Recipient (i) make no representations or undertakings regarding the treatment of any Tax-Related Items in connection with any aspect of the RSUs or the underlying Shares, including, but not limited to, the grant, vesting or settlement of the RSUs, the subsequent sale of Shares acquired pursuant to such settlement and the receipt of any dividends or other distributions paid on the Shares, and (ii) do not commit to and are under no obligation to structure the terms of the grant or any aspect of the RSUs to reduce or eliminate the Participant’s liability for Tax-Related Items or achieve any particular tax result. Further, if the Participant is subject to Tax-Related Items in more than one jurisdiction, the Participant acknowledges that the Company and/or the Service Recipient (or former service recipient, as applicable) may be required to withhold or account for Tax-Related Items in more than one jurisdiction.
(i)Tax Withholding. Prior to any relevant taxable or tax withholding event, as applicable, the Participant agrees to make arrangements satisfactory to the Company and the
Service Recipient to satisfy any applicable withholding obligations the Company or the Service Recipient may have for Tax-Related Items. In this regard, the Participant authorizes the Company and the Service Recipient, as applicable, and their respective agents, at their discretion, to satisfy any applicable withholding obligation for Tax-Related Items by one or a combination of the following:
(a)withholding from wages or other cash compensation payable to the Participant by the Company or any Service Recipient;
(b)requiring the Participant to tender a cash payment or make a payment in a form acceptable to the Company or the Service Recipient;
(c)withholding from proceeds of the sale of Shares to be issued upon vesting of the RSUs either through a voluntary sale or through a mandatory sale arranged by the Company (on the Participant’s behalf pursuant to this authorization without further consent);
(d)withholding in Shares to be issued upon vesting of the RSUs (in which case the Participant will be deemed to have been issued the full number of Shares subject to the vested portion of the RSUs, notwithstanding that a number of the Shares are held back solely for the purpose of paying the Tax-Related Items); and
(e)any other method acceptable to the Company and to the extent required under the Plan and applicable laws, approved by the Committee.
The Company and/or the Service Recipient may withhold or account for Tax-Related Items by considering statutory withholding rates or other applicable withholding rates, including minimum or maximum rates applicable in the Participant’s jurisdiction(s). In the event of over-withholding or if the maximum applicable rate for the Participant’s jurisdiction is used in connection with the withholding methods described in (c) or (d) above, the Participant may receive a refund of any over-withheld amount in cash (with no entitlement to the equivalent amount in Shares), or if not refunded, the Participant may be able to seek a refund from the applicable tax authorities. In the event of under-withholding, the Participant may be required to pay additional Tax-Related Items directly to the applicable tax authorities or to the Company and/or the Service Recipient. If any applicable withholding obligation for Tax-Related Items is satisfied by withholding in Shares, for tax purposes, the Participant will be deemed to have been issued the full number of Shares subject to the vested RSUs, notwithstanding that a number of the Shares is held back solely for the purpose of paying the Tax-Related Items. The Participant agrees to pay to the Company or the Service Recipient any amount of Tax-Related Items that the Company or the Service Recipient may be required to withhold or account for as a result of the Participant’s participation in the Plan that cannot be satisfied by the means previously described. The Company may refuse to deliver the Shares or the proceeds of the sale of Shares, if the Participant fails to comply with the Participant’s obligations for Tax-Related Items.
(ii)Tax Consequences. The Participant has reviewed with his or her own tax advisors the applicable tax consequences of this investment and the transactions contemplated by this Award Agreement. With respect to such matters, the Participant relies solely on such advisors and not on any statements or representations of the Company or any of its agents, written or oral.
Section 3.5Restricted Stock Units Non-Transferable. Except to the limited extent provided in Section 2, this Award and the rights and privileges conferred hereby will not be transferred, assigned, pledged or hypothecated in any way (whether by operation of law or otherwise) and will not be subject to sale under execution, attachment or similar process. Upon any attempt to transfer, assign, pledge, hypothecate or otherwise dispose of this Award, or any
right or privilege conferred hereby, or upon any attempted sale under any execution, attachment or similar process, this Award and the rights and privileges conferred hereby immediately will become null and void.
Section 3.6Unfunded and Unsecured Plan. The Participant acknowledges that the Plan is unfunded and the Company’s obligations under the Plan and this Award Agreement are unsecured and that, to the extent the Participant or his or her estate holds any rights by virtue of a grant of RSUs, such rights shall be no greater than the rights of an unsecured creditor of the Company.
Section 3.7No Stockholder Rights. Under no circumstances shall RSUs be considered Shares or other securities of the Company, nor shall they entitle the Participant to exercise voting rights, to receive dividends or other distributions or credit therefor, or to exercise or receive any other rights attaching to the ownership of Shares or other securities of the Company, nor shall the Participant be considered the owner of Shares by virtue of the award of RSUs.
Section 3.8No Guarantee of Continued Employment. THE PARTICIPANT ACKNOWLEDGES AND AGREES THAT THE VESTING OF THE RESTRICTED STOCK UNITS PURSUANT TO THE VESTING SCHEDULE HEREOF IS EARNED ONLY BY CONTINUING AS AN EMPLOYEE OR SERVICE PROVIDER AT THE WILL OF THE COMPANY (OR, IF DIFFERENT, THE SERVICE RECIPIENT) AND NOT THROUGH THE ACT OF BEING HIRED, BEING GRANTED THIS AWARD OF RESTRICTED STOCK UNITS OR, AS APPLICABLE, ACQUIRING SHARES HEREUNDER. THE PARTICIPANT FURTHER ACKNOWLEDGES AND AGREES THAT THIS AGREEMENT, THE TRANSACTIONS CONTEMPLATED HEREUNDER AND THE VESTING SCHEDULE SET FORTH IN THE NOTICE OF GRANT OF AWARD DO NOT CONSTITUTE AN EXPRESS OR IMPLIED PROMISE OF CONTINUED ENGAGEMENT AS AN EMPLOYEE OR SERVICE PROVIDER FOR THE VESTING PERIOD, FOR ANY PERIOD, OR AT ALL, AND SHALL NOT INTERFERE IN ANY WAY WITH THE PARTICIPANT’S RIGHT OR THE RIGHT OF THE COMPANY (OR SUBSIDIARY EMPLOYING THE PARTICIPANT) TO TERMINATE THE PARTICIPANT’S EMPLOYMENT OR SERVICE RELATIONSHIP AT ANY TIME, WITH OR WITHOUT CAUSE.
Section 3.9Notices. Any notice to be given under the terms of this Award Agreement to the Company shall be addressed to the Company in care of its Secretary, and any notice to be given to the Participant shall be addressed to him at the address given beneath his signature hereto. By a notice given pursuant to this Section 3.9, either party may hereafter designate a different address for notices to be given to it or him. Any notice which is required to be given to the Participant shall, if the Participant is then deceased, be given to the Participant’s personal representative if such representative has previously informed the Company of his status and address by written notice under this Section 3.9. Any notice shall have been deemed duly given when enclosed in a properly sealed envelope or wrapper addressed as aforesaid, deposited (with postage prepaid) in a post office or branch post office regularly maintained by the U.S. Postal Service or comparable foreign postal service.
Section 3.10Compliance with Laws. Notwithstanding any other provision of the Plan or this Award Agreement, unless there is an available exemption from any registration, qualification or other legal requirement applicable to the Shares, the Company shall not be required to permit the issuance and/or deliver any Shares prior to the completion of any registration or qualification of the Shares under any U.S. or non-U.S. local, state or federal securities or exchange control law or under rulings or regulations of the U.S. Securities and Exchange Commission (“SEC”) or of any other governmental regulatory body, or prior to
obtaining any approval or other clearance from any U.S. or non-U.S. local, state or federal governmental agency, which registration, qualification or approval the Company shall, in its absolute discretion, deem necessary or advisable. The Participant understands that the Company is under no obligation to register or qualify the Shares with the SEC or any state or non-U.S. securities commission or to seek approval or clearance from any governmental authority for the issuance or sale of the Shares subject to the RSUs. Further, the Participant agrees that the Company shall have unilateral authority to amend this Award Agreement without the Participant’s consent to the extent necessary to comply with securities or other laws applicable to issuance of the Shares subject to the RSUs.
Section 3.11Modifications to the Award Agreement. Modifications to this Award Agreement or the Plan can be made only in an express written contract executed by a duly authorized officer of the Company. Notwithstanding anything to the contrary in the Plan or this Award Agreement, the Company reserves the right to revise this Award Agreement as it deems necessary or advisable, in its sole discretion and without the consent of the Participant, to comply with Section 409A of the Code or to otherwise avoid imposition of any additional tax or income recognition under Section 409A of the Code prior to the actual issuance of Shares pursuant to this award of RSUs.
Section 3.12Amendment, Suspension or Termination of the Plan. By accepting this Award Agreement or RSUs, the Participant expressly warrants that he or she has received an award of RSUs under the Plan, and has received, read and understood a description of the Plan. The Participant understands that the Plan is discretionary in nature and may be amended, suspended or terminated by the Company at any time.
Section 3.13Governing Law and Venue. The laws of the State of Delaware shall govern the interpretation, validity, administration, enforcement and performance of the terms of this Award Agreement regardless of the law that might be applied under principles of conflicts of laws. For purposes of litigating any dispute that arises under this grant or this Award Agreement, the parties hereby submit to and consent to the jurisdiction of the State of New Jersey and agree that such litigation shall be conducted in the state and/or federal courts located in New Jersey, where this grant is made and/or to be performed.
Section 3.14Assignment. Rights and obligations of the Company under this Award Agreement may be assigned by the Company to a successor in the business of the Company, any company resulting from any amalgamation, reorganization, combination, merger or arrangement of the Company, or any company acquiring all or substantially all of the assets or business of the Company.
Section 3.15Data Privacy.
(a)Pursuant to applicable data protection laws, the Participant is hereby notified of the collection, use and transfer, in electronic or other form, of the Data (defined below) by and among, as applicable, the Participant's employer, the Company and any Related Corporation or Affiliate for the exclusive purpose of granting, administering and managing the Plan.
(b)The Participant understands that the Company and any Related Corporation or Affiliate may hold (but only process or transfer to the extent required or permitted by local law) the following personal information about the Participant, including, but not limited to, the Participant's name, home address, email address and telephone number, date of birth, social insurance number, passport number or other identification number, salary, nationality, job title, any Shares or directorships held in the Company, details of all RSUs or any other entitlement to Shares awarded, canceled, purchased, exercised, vested, unvested or
outstanding in the Participant's favor, for the exclusive purpose of implementing, administering and managing the Plan (“Data”).
(c)Where required by applicable data protection laws, the legal basis for the processing of the Data is that it is necessary for the performance of a contract to grant, administer and manage the Plan. In addition to the below-identified recipients and, where required under applicable law, the Data also may be disclosed to certain securities or other regulatory authorities where the Company's securities are listed or traded or regulatory filings are made. The legal basis, where required, for such disclosure is compliance with applicable law. The Participant's refusal to provide Data would make it impossible for the Participant's employer and Company to perform its contractual obligations and may affect the Participant's ability to be granted awards under the Plan.
(d)The Participant understands that Data will be transferred to E*TRADE from Morgan Stanley Securities LLC and its affiliated companies (“E*TRADE from Morgan Stanley”), Equiniti and its affiliated companies (“Equiniti”), or such other stock plan service provider used currently or as may be selected by the Company in the future, which is assisting the Company with the implementation, administration and management of the Plan. Where required under applicable law, Data also may be disclosed to certain securities or other regulatory authorities where the Company's shares are listed or traded or regulatory filings are made. The Participant understands that the recipients of the Data may be located in the United States or elsewhere, and that the recipient’s country (e.g., the United States) may have different data privacy laws and protections than the Participant's country, which may not provide the same statutory level of protection. The Participant understands that if the Participant resides outside the United States, under certain applicable laws, the Participant may request a list with the names and addresses of any potential recipients of the Data by contacting privacy@integralife.com.
(e)The Participant understands that Data will be held only as long as is necessary to implement, administer and manage the Participant's participation in the Plan, including any requisite transfer of such Data as may be required to a broker or other third party with whom the Participant may elect to deposit any Shares acquired at vesting of the RSUs.
(f)The Participant understands that, if the Participant resides outside the United States, the Participant may, under certain applicable laws, view Data, request additional information about the storage and processing of Data, require any necessary amendments to Data or refuse or withdraw the consents where applicable, in any case without cost, by contacting privacy@integralife.com.
(g)If the Participant's consent is required to process the Data, the Participant understands that the Participant is providing such consent on a purely voluntary basis. If the Participant does not consent, or if the Participant later seeks to revoke his or her consent, the Participant's employment status or service with the Company will not be affected; the only consequence of refusing or withdrawing consent is that the Company would not be able to grant RSUs to the Participant or administer or maintain such RSUs. Therefore, the Participant understands that refusing or withdrawing his or her consent may affect the Participant's ability to participate in the Plan. For more information on the consequences of the Participant's refusal to consent or withdrawal of consent, the Participant understands that the Participant may contact privacy@integralife.com.
Section 3.16Electronic Delivery and Acceptance. The Participant hereby consents to receive the Notice of Grant of Award and Award Agreement and any other documents related to this award or future awards by electronic delivery and to accept this or future awards through an on-line or electronic system established and maintained by the
Company or another third-party designated by the Company. The Participant acknowledges that he/she has read, understands and agrees to the terms of the Notice of Grant of Award and Award Agreement. Further, if the Participant does not decline the Award by written notice to the Company no later than 60 days following the date of grant or such other date that may be communicated by the Company, the Company will automatically accept the Award, subject to all terms and conditions set forth in the Notice of Grant of Award and Award Agreement and the Plan, on the Participant’s behalf. If the Participant properly declines the Award, the Award will be cancelled and the Participant will not be entitled to any benefits from the Award nor any compensation or benefits in lieu of the cancelled Award.
Section 3.17Waivers. No waiver by either party of any breach of, or of compliance with, any condition or provision of this Award Agreement by the other party shall be considered a waiver of any other condition or provision or of the same condition or provision at another time.
Section 3.18Appendix. Notwithstanding any provisions in this Award Agreement, this Award of RSUs shall be subject to any special terms and conditions for the Participant’s country set forth in Appendix A attached hereto. Moreover, if the Participant relocates to one of the countries included in Appendix A, the special terms and conditions for such country will apply to the Participant, to the extent the Company determines that the application of such terms and conditions is necessary or advisable for legal or administrative reasons. Appendix A constitutes part of this Award Agreement.
Section 3.19Entire Agreement. The Notice of Grant of Award, this Award Agreement and the Plan constitute the entire contract between the parties hereto with regard to the subject matter hereof. They supersede any other agreements, representations or understandings (whether oral or written and whether express or implied) which relate to the subject matter hereof. The Participant expressly warrants that he or she is not accepting this Award Agreement in reliance on any promises, representations, or inducements other than those contained herein.
Section 3.20Severability. The provisions of this Award Agreement are severable and if any one or more provisions are determined to be illegal or otherwise unenforceable, in whole or in part, the remaining provisions shall nevertheless be binding and enforceable.
Section 3.21Language. The Participant acknowledges that the Participant is sufficiently proficient in English or has consulted with an advisor who is sufficiently proficient in English so as to allow the Participant to understand the terms and conditions of this Award Agreement. If the Participant has received this Award Agreement, or any other document related to the Plan translated into a language other than English and if the meaning of the translated version is different than the English version, the English version will control.
Section 3.22Foreign Asset/Account Reporting Requirements, Exchange Controls and Tax Requirements. The Participant acknowledges that his or her country may have certain foreign asset and/or account reporting requirements and exchange controls which may affect his or her ability to acquire or hold Shares under the Plan or cash received from participating in the Plan (including from any dividends or sale proceeds arising from the sale of Shares) in a brokerage or bank account outside his or her country. The Participant understands that he or she may be required to report such accounts, assets or transactions to the tax or other authorities in his or her country. The Participant also may be required to repatriate sale proceeds or other funds received as a result of the Participant’s participation in the Plan to his or her country through a designated bank or broker and/or within a certain time after receipt. In addition, the Participant may be subject to tax payment and/or reporting obligations in connection with any
income realized under the Plan and/or from the sale of Shares. The Participant acknowledges that it is his or her responsibility to be compliant with all such requirements, and that the Participant should consult his or her personal legal and tax advisors, as applicable, to ensure the Participant’s compliance.
Section 3.23Insider Trading and Market-abuse Laws. The Participant acknowledges that, depending on the Participant’s or his or her broker’s country of residence or where the Shares are listed, the Participant may be subject to insider-trading restrictions and/or market-abuse laws, which may affect the Participant’s ability to accept, acquire, sell or otherwise dispose of Shares, rights to Shares (e.g., the RSUs), or rights linked to the value of Shares (e.g., phantom awards, futures) during such times as the Participant is considered to have “inside information” regarding the Company (as defined by law or regulations in the Participant’s country). Local insider trading laws and regulations may prohibit the cancellation or amendment of orders the Participant placed before the Participant possessed inside information. Furthermore, the Participant could be prohibited from (i) disclosing the inside information to any third party (other than on a “need to know” basis) and (ii) “tipping” third parties, including fellow employees, or causing them otherwise to buy or sell securities. Any restrictions under these laws or regulations are separate from and in addition to any restrictions that may be imposed under the Company’s insider-trading policy. The Participant understands that it is the Participant’s responsibility to comply with any applicable restrictions as well as any Company insider trading policy, and the Participant should consult his or her personal legal advisor on this matter.
Section 3.24Imposition of Other Requirements. The Company reserves the right to impose other requirements on the Participant’s participation in the Plan, on this award of RSUs and on any Shares received from the RSUs, to the extent the Company determines it is necessary or advisable for legal or administrative reasons, and to require the Participant to sign any additional agreements or undertakings that may be necessary to accomplish the foregoing.
[SIGNATURE PAGE FOLLOWS]
IN WITNESS HEREOF, this Award Agreement has been executed and delivered by the parties hereto.
THE PARTICIPANT Electronic signature to be provided and recorded via online grant acceptance process on www.etrade.com | INTEGRA LIFESCIENCES HOLDINGS CORPORATION By Name: Title: | ||||
APPENDIX A
[This appendix has been omitted pursuant to Item 601(a)(5) of Regulation S-K under the Securities Act of 1933, as amended. The registrant agrees to furnish supplementally a copy of the omitted annex to the Securities and Exchange Commission upon request.]
IART-2025.12.31-EX-10.12
INTEGRA LIFESCIENCES
EXECUTIVE SEPARATION PAY PLAN
(Plan Document and Summary Plan Description)
Effective February 24, 2026
PLAN NAME
Integra LifeSciences Executive Separation Pay Plan (referred to herein as the “Plan”).
PURPOSE
The purpose of the Plan is to provide certain payments to Eligible Employees who experience a Qualifying Termination (generally, an involuntary termination of employment as a result of job elimination, or similar termination unrelated to performance), subject to the execution of a General Release and satisfaction of the other terms and conditions of the Plan.
PLAN EFFECTIVE DATE AND SUMMARY PLAN DESCRIPTION
This information constitutes both the Plan Document and the Summary Plan Description required by the Employee Retirement Income Security Act of 1974, as amended (“ERISA”). The Plan is intended to constitute an “employee welfare benefit plan” within the meaning of Section 3(1) of ERISA.
Integra LifeSciences Holdings Corporation, a Delaware corporation (the “Company”) has adopted this Plan for its Eligible Employees, effective February 18, 2026. The Plan is effective with respect to notifications of terminations of employment occurring on and after February 18, 2026, and it supersedes and replaces any other programs and practices for separation pay for Eligible Employees.
PLAN ADMINISTRATION
The Company’s Vice President of Total Rewards (the “Plan Administrator”) shall be the “administrator” and the “named fiduciary” described in Sections 3(16) and 402(a)(2), respectively, of ERISA for the Plan and shall have the complete discretionary authority to:
(1)Determine eligibility for benefits and amount of benefits in accordance with the provisions of the Plan and make any factual assessments relating to the Plan;
(2)Construe, interpret, resolve any ambiguity, or supply any omission in the terms of the Plan;
(3)Control and manage the operation of the Plan, including by establishing policies, procedures, forms, or regulations governing the Plan; and
(4)Exercise his or her discretion as provided for by specific provisions of the Plan.
Any use of discretion, as permitted under this Plan, by the Plan Administrator, or his or her delegate, shall be made upon the application of non-discriminatory criteria uniformly applied and in accordance with standards of conduct set forth in Part 4 of Subtitle B of Title I of ERISA. The Plan Administrator’s decision, or that of his or her delegate, shall be final and conclusive as to all facts and all interpretations of the terms of the Plan, its operations, and the benefits intended to be provided and shall not be reversed unless found by a court of competent jurisdiction to be arbitrary and capricious.
The Plan Administrator has appointed the Director of Executive Compensation* and the Lead Benefits and Equity Plan Coordinator* each as the Plan Manager responsible for the initial determinations of benefit eligibility and for the day-to-day administration of the Plan. Contact information for the Plan Administrator and the Plan Manager may be found in the Supplement to Summary Plan Description at the end of this document.
2
Integra LifeSciences Executive Separation Pay Plan
* The above titles may be updated from time to time. In the event of a change in title, restructuring, or similar event, these references will be automatically updated (without the necessity of a formal Plan amendment) to reflect the then-current, reasonably equivalent position.
DEFINITIONS
Certain terms, when used with an initial capital letter, have specific meanings as used in the Plan. Many of those terms are defined in this Section, while others may be found in the body of this Plan document, so placed for easier reference.
Code
The Internal Revenue Code of 1986, as amended, and any regulations issued under any particular Section of the Code.
Eligible Employee
An “Eligible Employee” is an Employee on the Executive Leadership Team of the Company, as determined in the sole discretion of the Company.
An Eligible Employee also must be a non-temporary, benefitted full-time or benefitted part-time Employee of an Company, employed in the United States and on the United States payroll who is: (a) actively at work; (b) eligible to participate in the generally available Company-sponsored employee benefit plans (i.e., employed in a “benefitted” position); and (c) regularly scheduled to work at least 20 hours per week. Whether an Employee is an Eligible Employee under this Plan is determined in the sole discretion of the Plan Administrator or Plan Manager.
Notwithstanding the above, no individual in any of the following categories shall be an Eligible Employee:
•Classified as seasonal, occasional, limited duration, or a temporary employee;
•Covered by an employment, separation or other written agreement, contract or plan (either covering the person individually or covering a group of employees which includes the individual, such as for members of the Company’s Executive Committee) that provides compensation and/or benefits for periods following their termination of employment (a “Separate Agreement”), except where the Separate Agreement expressly provides for continued eligibility under this Plan; or
•Has not been actively at work for 26 or more weeks on account of illness or injury.
Employee
Any person employed by the Company on its United States payroll. The term does not include: (a) an individual classified by the Company as an independent contractor (even if subsequently reclassified by a court, agency, or the Company itself); (b) any leased employee and/or person being paid through an employee leasing company or other third-party agency; or (c) an individual classified as a freelance worker or consultant.
General Release
A letter agreement containing a general release of claims and/or restrictive covenants, in the form specified by and acceptable to the Company. The General Release, among other things, waives, releases,
3
Integra LifeSciences Executive Separation Pay Plan
and discharges the Company from all waivable claims and liabilities relating to the Eligible Employee’s employment with the Company and/or termination of the Eligible Employee’s employment. Such General Release may impose certain non-competition, non-solicitation, confidentiality and other commercially appropriate restrictions that the Company deems appropriate as a condition of the Employee receiving the Separation Pay (or may reaffirm pre-existing restrictive covenants, such as those provided in the Restrictive Covenant Agreement), and may include provisions regarding forfeiture and/or repayment of benefits in the event of reemployment or upon breach of the General Release or breach of other applicable restrictive covenants or other agreement with the Company that remains effective following the Qualifying Termination.
Month of Base Pay
With respect to a full-time Eligible Employee or a salaried, part-time Eligible Employee, a “Month of Base Pay” is such Employee’s monthly base salary at the time of the Eligible Employee’s Notification. The amount of a Month of Base Pay shall be determined by the Company’s payroll records and in no event will include special adjustments or increments such as bonuses, overtime pay, shift differential, commissions, incentive or deferred compensation or other additional compensation or allowances. Such amount will, however, include amounts deducted from base pay on a pre-tax basis for taxes, retirement contributions, welfare benefit premiums, and the like.
Notification
A Notification under the Plan occurs when an Eligible Employee receives a written notice from the Plan Manager advising such individual of a Qualifying Termination under the terms of the Plan.
Participant
An Eligible Employee who has received a Notification and incurs a Qualifying Termination under the Plan. Participants are entitled to the Separation Pay and other benefits under the Plan, subject to the execution of a General Release and satisfaction of other terms and conditions of the Plan.
Qualifying Termination
Generally occurs upon an involuntary termination of employment with the Company at a time when the individual is an Eligible Employee, as a result of: (a) a job elimination; or (b) other business reason not related to individual performance and not otherwise excluded below.
Under no circumstances will any of the following be considered a Qualifying Termination:
•A voluntary resignation or retirement (including electing to participate in a voluntary early retirement or voluntary separation program);
•A change in work schedule or employee status (e.g., a change from full-time to part-time, benefitted to non-benefitted, change in work location, or change in remote to in-person) or the establishment, change, or termination of a particular working arrangement (such as a flexible or remote work arrangement), whether voluntary or involuntary, without regard to the commuting distance;
•There is no actual termination of employment with the Company (such as, but not limited to, a revocation or deferral of the Notification or a placement in an alternative position);
•The Company notifies the Plan Manager that the Employee’s termination occurred as a result of the individual’s:
4
Integra LifeSciences Executive Separation Pay Plan
oUnsatisfactory job performance, as determined by the Company in its sole and absolute discretion, which shall include by way of example and not limitation, the individual’s poor performance, attendance and/or punctuality, inappropriate conduct, violation of or disregard for the policies and procedures of the Company (including, but not limited to, the Company’s Global Code of Conduct and the Restrictive Covenant Agreement (as defined herein)), or any other improper disclosure of confidential information or trade secrets), insubordination, fraud, or conduct that would constitute “Cause” under any employment agreement or Company policy;
oDeath, disability, voluntarily resigning from (or providing notice of intention to resign from) or abandoning any position with the Company;
oDeclining the Company’s offer of employment with the Company or any subsidiary or affiliate (or a joint venture of any such entity);
oAccepting (either after affirmatively applying for the same or being offered the position, without regard to whether the position was identified through a job posting program available to existing employees of the Company or through the individual’s own efforts) and subsequently declining, a position with the Company;
oBeing offered (whether or not such offer is accepted) employment with a third party as a result of the sale, outsourcing or other divestiture of one or more of the Company’s businesses to such third party (regardless of whether the transaction is accomplished through the sale of stock or assets, a merger, a joint venture, or an outsourcing contract), where the arrangement, by its terms as in effect on the day prior to the effective date (the “Closing”) of such transaction, requires the third party to offer the individual employment (or continued employment) regardless of whether the offered employment is comparable to the position the individual held immediately before the Closing and regardless of whether the agreement provides for any minimum guaranteed period of employment or minimum terms or conditions of employment following the Closing;
oLayoff, reduction in force or similar condition which the Company, in its sole and absolute discretion, determines is indefinite or temporary;
oBecoming unemployed as a result of any strike, slowdown, work stoppage, picketing, or concerted action at a location or locations of the Company;
oSeparation from the workforce as a result of any war or hostile action; governmental regulations or controls over the amount or kind of goods or services the Company may use or sell; sabotage or insurrection; or any act of God such as fire, flood, earthquakes, tornadoes, terrorist action, and the like; or
oReceiving or eligibility to receive any benefit from the Company of equal or greater value as Plan benefits, where such benefits are intended to provide benefits in the nature of separation or severance benefits (including but not limited to receiving benefits under the Separate Agreement).
5
Integra LifeSciences Executive Separation Pay Plan
Restrictive Covenant Agreement
The Confidentiality and Invention Disclosure and Non-Compete Agreement executed by the Employee, as applicable, as amended or updated by the Company from time to time.
Separation Pay
The cash payment to which a Participant may be entitled under the Plan, which is generally expressed as a number of Months of Base Pay plus, if applicable, a health care stipend, a prorated bonus payment, and career transition services, as further described in the Plan. Any compensation earned during a Notification period or pay in lieu of notice is not considered Separation Pay.
ELIGIBILITY FOR SEPARATION PAY BENEFITS
Subject to the execution by the employee of a General Release (described below) and the other terms and conditions of the Plan, an Eligible Employee is eligible to receive Separation Pay under the Plan if the Company terminates his or her employment in a Qualifying Termination.
Notification of Eligibility and Separation Date
Eligible Employees will be notified in writing of: (i) the date of the occurrence of the Qualifying Termination, which will constitute his or her official “Separation Date”; (ii) the amount of Separation Pay and any other benefits to which he or she is entitled under the Plan; (iii) the form of General Release that such Eligible Employee must sign and not revoke to receive any Separation Pay; and (iv) any other terms and conditions as may be applicable or deemed relevant by the Plan Manager or his or her delegate. Such written notice constitutes the “Notification” and will generally be given at least two (2) weeks in advance of the Separation Date, unless business needs, as determined by the Plan Manager in his or her sole discretion after consultation with the Eligible Employee’s manager, require a longer or shorter notice period.
Oral communications or oral or written estimates of possible benefits do not constitute a formal Notification under the Plan. Eligible Employees who have received a Notification are not eligible to receive merit increases with an effective date after the Separation Date.
Amount of Separation Pay Benefits
A Participant who timely executes, and does not revoke, the required General Release will receive the following “Separation Pay”:
A. An amount equal to twelve (12) Months of Base Pay. For purposes of the Plan, this period shall be deemed the “Separation Period”.
B. For a Participant who is enrolled in Company-sponsored medical, dental, and/or vision coverage (“Health Coverage”) as of the Notification Date, an amount equal to the Company’s contribution towards such Health Coverage as in effect for similarly situated active employees (as applicable, based on the Participant’s enrollment status and covered dependents, if any) in effect on the date of Notification, all as determined by the Plan Manager in his or her discretion, covering a period through the earlier of (x) the end of the Separation Period, or (y) the date the Eligible Employee becomes eligible for group medical care coverage through other employment (the “COBRA Benefit”). For clarity, regardless of whether a payment is due under this Section B, all active Health Coverage will end as of the end of the month in which the Separation Date occurs.
6
Integra LifeSciences Executive Separation Pay Plan
Participants are solely responsible for electing coverage under the Consolidated Omnibus Budget Reconciliation Act (COBRA) if continued Health Coverage under the Company’s plan is desired.
C.For a Participant who participates in any cash incentive or other bonus program sponsored by the Company providing for annual bonus payments, and provided the Participant has remained continuously employed by the Company for a minimum of nine (9) months during the calendar year of the Employee’s Qualifying Termination, the Participant will receive an amount equal to the actual bonus payment which would have been paid to the Employee had he or she remained in continuous employment through the end of the calendar year of the bonus period, subject to the funding of the Company, individual performance of the Employee and any additional eligibility criteria for such bonus payment, pro-rated based on the number of calendar days the Employee was employed by the Company during the calendar year of the Qualifying Termination. Payment of the pro-rated annual bonus, if any, under this Section C will be subject to the Company’s sole discretion.
Outplacement Services
Participants entitled to Separation Pay shall additionally be offered career transition assistance during the Separation Period, pursuant to the policies and procedures, and through the Company’s global outplacement vendor or other outplacement agent as may be selected by the Plan Manager in his or her sole discretion (the “Company Outplacement Services”). If the Employee chooses to utilize career transition assistance from a preferred outplacement provider of the Employee’s choice, the Company will reimburse the amount of such services, up to a maximum amount which is typically contractually paid to the Company Outplacement Services.
Regardless of the duration, type or scope (if any) of such career transition assistance, in no event will the Company or the Plan be responsible for any other costs or expenses associated with the Employee’s search for new employment.
Release Requirement
In order to receive Separation Pay and any other benefits under the Plan, the Eligible Employee must sign and not revoke a General Release or General Releases within a period specified by the Plan Manager and ending not more than sixty (60) days following the Separation Date.
Method of Payment
Participants eligible to receive Separation Pay will be paid in substantially equal installments during the Separation Period, in accordance with the Company’s normal payroll cycle and beginning as soon as administratively practicable after the Plan Administrator (or his or her designee) receives the General Release executed by the Participant and the expiration of any applicable period during which an Employee may elect to revoke the General Release.
The Separation Pay is paid through payroll and subject to withholding required by law or otherwise authorized by the Employee and will be reduced by any monies owed (e.g., personal account expenses and tuition reimbursement) by the Employee to the Company, to the extent permitted by law.
Offer of Reemployment
Notwithstanding the above, in the event a Participant is offered a different position for full-time employment at the Company prior to the end of the Separation Period (including during the period following the Notification and prior to the start of any Separation Pay), regardless of whether the Participant accepts such new offer of employment, all Separation Pay and benefits shall cease as of the
7
Integra LifeSciences Executive Separation Pay Plan
date one (1) week following such offer of reemployment; provided, that if such offer of reemployment by the Company offers a base salary which is less than eighty-five percent (85%) of the Participant’s base salary at the time of such Participant’s prior Notification by the Company and the Participant rejects such offer, the Participant will remain eligible to receive the Separation Pay.
Reduction of Benefits
Notwithstanding anything in the Plan to the contrary, a Participant’s Separation Pay is not meant to duplicate pay and benefits which may be due by the Company to the Participant in connection with an eligible termination under the federal Worker Adjustment Retraining and Notification Act and any state or local equivalent (collectively the "WARN Act"). If the Plan Administrator determines that a Participant is entitled to notice, payment, or benefits under the WARN Act, the Plan Administrator in its sole and absolute discretion may, to the extent permitted under applicable law, reduce the Participant's Separation Pay under the Plan by any WARN Act required pay and benefits (the “WARN Benefit”) after receiving WARN Act notice.
In the event the amount of any WARN Benefit due to a Participant exceeds the amount of the Separation Pay to which such Participant is entitled under the Plan, the Company may, in its sole discretion, provide an additional amount of up to four (4) Weeks of Base Pay to the Participant and/or retain the Participant’s eligibility for the COBRA Benefit, each subject to the terms and conditions in the Plan and conditioned upon the Participant’s execution and non-revocation of the General Release.
Offsets and Clawback
All Separation Pay is subject to and conditioned upon the effectiveness of a Participant’s General Release. If any Participant violates the terms of his or her General Release, the Company shall be entitled, and the Plan Manager has full discretion and authority to require, repayment of the full (gross) amount of any Separation Pay previously paid (determined prior to any withholdings or deductions for taxes or otherwise).
Miscellaneous
Correction of Errors
The Plan Administrator strives to operate the Plan as accurately as possible. Occasionally, however, errors in determining eligibility and/or calculation of Separation Pay or other entitlements may occur. In such situations, the Plan Administrator expressly reserves the right to correct any and all errors. This includes, by way of example but not limitation, the right to demand repayment of any excess Separation Pay erroneously made to a separated employee due to erroneous assessment of eligibility or title, or miscalculations of the Month of Base Pay, or health plan subsidies. Any past failure or declination to correct any error in the operation of the Plan will not prevent or otherwise restrict the correction of a future error.
No Additional Rights
Neither the establishment of the Plan, nor any modification thereof, nor the payment of any Plan benefits will be construed as giving any employee or other individual any legal or equitable right against the Company or any officer, director or employee thereof; and in no event shall the terms and conditions of any individual’s employment or other service be modified in any way by the existence of this Plan. All employees remain subject to termination or corrective action to the same extent as if this Plan had not been put into effect.
8
Integra LifeSciences Executive Separation Pay Plan
Records
The determinations by the Company, Plan Administrator, or Plan Manager with respect to Month of Base Pay, absences, employment history, reasons for termination, title, and other relevant matters shall be conclusive and binding for all purposes under the Plan.
Construction
The terms of this Plan will be construed, wherever possible, to be in conformity with the provisions of ERISA, the Code, and other applicable laws. To the extent not in conflict with the prior sentence or other express provisions of the Plan, the construction and administration of the Plan will be in accordance with the laws of the State of Delaware (without regard to its conflict of laws provisions). All actions and proceedings relating to or arising, directly or indirectly, from this Plan shall be litigated exclusively in the State of Delaware (and shall be subject to the provisions in the Claims Procedures section requiring exhaustion of claims and appeals to the maximum extent permitted under ERISA). By virtue of participation in this Plan, all persons waive trial by jury in any judicial proceeding involving any matter related to or connected with this Plan.
All words used in the singular will be construed to include the plural, where required for the context, and all words used in the plural may likewise be construed to include the singular. Any words used the feminine or masculine gender will be construed to refer to any other gender.
Effect of Conflicting Provisions
This Plan document, together with any subsequent amendments adopted by the Company or the Plan Administrator, sets forth all the terms and conditions governing the provision of separation benefits to Eligible Employees. In the event that any written or oral description of Plan benefits (including, without limitation, any information in a Notification or communication about the Plan) conflicts with the terms of this Plan document, the terms of this Plan document will control.
Severability
Should any provision of this Plan be deemed or held to be unlawful or invalid for any reason, such provision shall (i) first be construed and interpreted to effectuate the intent of such provision to the maximum extent as may be deemed not unlawful or invalid by such court or arbiter assessing the issue, and (ii) if such reformation or restricted interpretation is not feasible, shall otherwise be disregarded such that the invalid or unlawful provision will not adversely affect the other provisions of the Plan.
Incompetency
In the event that the Plan Manager finds that a Participant is unable to care for his or her own affairs because of illness, accident or other condition, then benefits payable hereunder, unless claim has been made therefore by a duly appointed guardian, committee, or other legal representative, may be paid in such manner as the Plan Manager shall determine for the benefit of the Participant, and the application thereof shall be a complete discharge of all liability for any benefits to which such Participant was or would have been otherwise entitled under this Plan.
Nontransferability
9
Integra LifeSciences Executive Separation Pay Plan
In no event shall the Company make any payment under this Plan to any assignee or creditor of a Participant, except as otherwise required by law. Prior to the time of a payment hereunder, an Employee shall have no rights by way of anticipation or otherwise to assign or otherwise dispose of any interest under this Plan, nor shall rights be assigned or transferred by operation of law.
Amendment and Termination
It is the Company’s intent that the Plan will continue indefinitely. However, the Company reserves the right to amend, modify, suspend or terminate the Plan, by action of the Plan Administrator, in whole or in part, prospectively or retroactively, generally or in individual circumstances, and with or without prior notice. Any such action would be documented in writing and maintained with the records of the Plan. Plan amendment, modification, suspension or termination may be made for any reason, and at any time. No Eligible Employee obtains any vested right to future benefits under the Plan. If the Plan is terminated, amended or modified, individuals’ rights to participate in, or receive benefits under, the Plan may be changed; provided, however that the Separation Pay payable or due to a Participant who has received a Notification prior to such termination, amendment or modification, may not be reduced by virtue of such termination, amendment or modification. Eligible Employees will be notified of any material modification to the Plan.
Funding
The Plan is an unfunded plan and Separation Pay and other benefits under the Plan are provided from the general assets of the Company. There is no trust or insurance policy funding Plan benefits. In the event of the insolvency of the Company, each Participant has a right to benefits no greater than the rights of the Company’s unsecured general creditors.
409A Compliance
The Plan is intended to be exempt from the requirements of Section 409A of the Code to the maximum extent feasible as a “short-term deferral” and/or “separation pay plan” as used in Section 409A of the Code, and otherwise to comply with Section 409A, and it will be construed and interpreted accordingly. In the event that any provision that is necessary for the Plan to comply with Section 409A is determined by the Plan Administrator, in its sole discretion, to have been omitted, such omitted provision shall be deemed included herein and is hereby incorporated as part of the Plan. Notwithstanding the foregoing, under no circumstances will the Company, the Plan Administrator, the Plan Manager, or any employee, officer, director or representative of any of the foregoing be responsible for any taxes or tax liabilities of an Employee or Participant, including any taxes under Section 409A of the Code. If, at the time of a Participant’s separation from service, (i) the Participant is a “specified employee” within the meaning of Section 409A, and (ii) the Company makes a good faith determination that an amount payable on account of the Participant’s separation from service constitutes deferred compensation (within the meaning of Section 409A), the payment of which is required to be delayed pursuant to the six (6)-month delay rule set forth in Section 409A to avoid taxes or penalties under Section 409A, then the Company shall not pay such amount on the otherwise scheduled payment date but shall instead pay it in a lump sum on the first business day after such delay period (or upon the Participant’s death, if earlier), without interest.
CLAIMS AND APPEALS; OTHER REGULATORY MATTERS
Claim and Appeal Procedures
A. Eligible Employees who incur a Qualifying Termination will receive written Notification of their eligibility for benefits under this Plan. Employees who are not so notified and believe that they are entitled to benefits (or disagree with the amount of benefits provided in the Notification or
10
Integra LifeSciences Executive Separation Pay Plan
otherwise have a disagreement with the administration of this Plan by the Plan Administrator), must present their disagreement or claim for benefits in writing to the Plan Manager.
B. In the event a request of any Employee or other individual, which may include an authorized representative, (hereinafter referred to as the “Claimant”) for a benefit under the Plan (“Claim”) is partially or completely denied, the Plan Manager, or his or her designee, shall give written notice of such denial to the Claimant within ninety (90) days after receipt of the Claim (or if special circumstances require an extension of time for processing the Claim, within one hundred and eighty (180) days after receipt of the Claim, provided that written notice of and the reasons for the delay are given to the Claimant within the original ninety (90)-day period). Such notice shall set forth: (1) the specific reason or reasons for the denial (with reference to pertinent Plan provisions upon which the denial is based); (2) an explanation of additional material or information, if any, necessary for the Claimant to perfect the Claim and a statement of why the material or information is necessary; (3) an explanation of the Plan’s claims review and appeal procedure and the time limits applicable to the Plan’s review procedure; and (4) a statement of the Claimant’s right to bring a civil action under Section 502(a) of ERISA following an adverse benefit determination on appeal.
C. An appeal of a denied Claim may be made by writing to the Plan Administrator within sixty (60) days after receiving the written Claim denial from the Plan Manager, or his or her designee. Such written request for review shall set forth the basis (whether legal or factual) upon which the review is requested, including, but not limited to, pertinent Plan provisions, employment records, prior decisions of the Plan Administrator and courts of competent jurisdiction, and/or statements of such facts or circumstances in the possession of the person requesting the review to which the Plan Administrator may not have had access.
The Claimant or his authorized representative shall also have the right to submit documents, records, and other information relating to the Claim regardless of whether it was submitted or considered in connection with the initial Claim, and shall be provided, upon request and free of charge, reasonable access to and copies of all documents, records, and other information relevant to the Claim.
D. The Plan Administrator will conduct a review of the Claim and render a written decision on appeal to the Claimant within sixty (60) days after receipt of the written appeal (or, if special circumstances require an extension of time, within one hundred and twenty (120) days, provided that written notice of and the reasons for the delay are given to the Claimant within the original sixty (60)-day period).
The Plan Administrator’s review shall take into account all comments, documents, records, and other information submitted by the Claimant or his authorized representative, without regard to whether such information was submitted or considered in the initial Claim determination. The decision by the Plan Administrator on review will be binding on all parties, will be written in a manner calculated to be understood by the Claimant, and will contain specific reasons for the decision and specific references to the pertinent Plan provisions on which the decision is based. The written notice of the decision denying a Claim on appeal shall also contain (1) a statement that the Claimant is entitled to receive, upon request and free of charge, reasonable access to copies of all documents, records, and other information relevant to the Claimant’s Claim for benefits, and (2) a statement of the Claimant’s right to bring an action under Section 502(a) of ERISA.
11
Integra LifeSciences Executive Separation Pay Plan
For purposes of this claim and appeal procedure, information is considered “relevant” to a Claimant’s Claim if such document, record, or other information (1) was relied upon in making the benefit determination; (2) was submitted, considered, or generated in the course of making the benefit determination, without regard to whether such document, record, or other information was relied upon in making the determination; or (3) demonstrates compliance with the Plan’s review procedures and, if appropriate, that the Plan provisions have been applied consistently with respect to similarly situated claimants.
E. In deciding claims and appeals, the Plan Administrator shall have complete and exclusive discretionary authority and responsibility to construe and interpret the Plan, to decide all questions of fact and eligibility, and to determine the amount, time, and manner of payment of any benefits under the Plan.
F. Failure to appeal any denial of a request for benefits in the manner and within the deadlines specified above, constitutes a waiver of the right to request a review of the Plan Administrator’s decision and is a bar to making the same request for benefits at some future date, whether through this claims procedure, in court, or otherwise. Additionally, no Claimant may bring suit for benefits hereunder after the earlier of (a) the date that is one (1) year after the denial of his or her appeal, or (b) two years after the Qualifying Termination (or alleged Qualifying Termination) on which the claim is based.
Rights and Obligations of the Company
The Company expects that circumstances will continue in order to maintain the Plan and its benefits. Since is it impossible to predict events and conditions in the future, the Company cannot promise or guarantee that the Plan or any of its benefits will remain in effect.
Therefore, although this Plan Document and Summary Plan Description describes the features of the Plan in effect as of February 18, 2026, the Company reserves the right to change or terminate the Plan or any of its benefits at any time.
Nothing in the Plan Document and Summary Plan Description should be construed as a promise of continuation of the Plan or any benefit from the Plan with respect to any future period. The Plan Administrator reserves the right to make the final determination and resolution of any dispute that may arise concerning interpretation of the Plan document.
Rights Under ERISA
The following statement is drafted by and required by federal law.
As a participant in the Plan, you are entitled to certain rights and protections under the Employee Retirement Income Security Act of 1974 (ERISA). ERISA provides that all Plan participants shall be entitled to:
Examine, without charge, at the Plan Administrator’s office and at other specified locations, such as work sites, all Plan documents, and copies of all documents filed by the Plan with the U.S. Department of Labor.
Obtain copies of all Plan documents and other Plan information upon written request to the Plan Administrator. The Plan Administrator may make a reasonable charge for the copies.
12
Integra LifeSciences Executive Separation Pay Plan
In addition to creating rights for Plan participants, ERISA imposes duties upon the people who are responsible for the operation of the employee benefit plan. The people who operate your Plan, called “fiduciaries” of the Plan, have a duty to do so prudently and in the interest of you and other Plan participants and beneficiaries. No one, including the Company or any other person, may fire you or otherwise discriminate against you in any way to prevent you from obtaining a benefit or exercising your rights under ERISA.
If your claim for a benefit is denied or ignored, in whole or in part, you have a right to know why this was done, to obtain copies of documents relating to the decision without charge, and to appeal any denial, all within certain time schedules.
Under ERISA, there are steps you can take to enforce the above rights. For instance, if you request a copy of Plan documents from the Plan and do not receive them within 30 days, you may file suit in a Federal court. In such a case, the court may require the Plan Administrator to provide the materials and pay you up to $110 a day until you receive the materials, unless the materials were not sent because of reasons beyond the control of the Plan Administrator. If you have a request for benefits which is denied or ignored, in whole or in part, you may file suit in a state or Federal court. If it should happen that Plan fiduciaries misuse the Plan’s money, or if you are discriminated against for asserting your rights, you may seek assistance from the U.S. Department of Labor, or you may file suit in a Federal court. The court will decide who should pay court costs and legal fees. If you are successful, the court may order the person you have sued to pay these costs and fees. If you lose, the court may order you to pay these costs and fees, for example, if it finds your claim is frivolous.
If you have any questions about your Plan, you should contact the Plan Administrator. If you have any questions about this statement or about your rights under ERISA, or if you need assistance in obtaining documents from the Plan Administrator, you should contact the nearest office of the Employee Benefits Security Administration, U.S. Department of Labor, listed in your telephone directory or the Division of Technical Assistance and Inquiries, Employee Benefits Security Administration, U.S. Department of Labor, 200 Constitution Avenue, N.W., Washington, D.C. 20210. You may also obtain certain publications about your rights and responsibilities under ERISA by calling the publications hotline of the Employee Benefits Security Administration. You may further contact the Plan Manager.
13
Integra LifeSciences Executive Separation Pay Plan
IART-2025.12.31-EX-10.13
Integra LifeSciences Holdings Corporation
Directors Deferred Compensation Plan
Effective Date
April 1, 2026
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Article I
Establishment and Purpose 1
Article II
Definitions 1
Article III
Eligibility and Participation 3
Article IV
Deferrals 3
Article V
Payments from Accounts 6
Article VI
Valuation of Account Balances; Investments 8
Article VII
Administration 9
Article VIII
Amendment and Termination 10
Article IX
Informal Funding 11
Article X
Claims 11
Article XI
General Provisions 16
Page 1 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Article I
Establishment and Purpose
Integra LifeSciences Holdings Corporation (the “Company”) has adopted this Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan, applicable to Compensation deferred under Compensation Deferral Agreements submitted on and after the Effective Date.
The purpose of the Plan is to attract and retain non-employee members of the Board of Directors of the Company by providing them with an opportunity to defer receipt of a portion of their director fees and equity awards. The Plan is intended to meet the requirements of applicable law, including Code Section 409A, and shall be operated and interpreted consistent with that intent.
The Plan constitutes an unsecured promise by the Company to pay benefits in the future. Participants in the Plan shall have the status of general unsecured creditors of the Company. The Company shall be solely responsible for payment of the benefits attributable to services performed by Directors. The Plan is unfunded for federal tax purposes. Any amounts set aside to defray the liabilities assumed by the Company will remain the general assets of the Company and shall remain subject to the claims of the Company’s creditors until such amounts are distributed to the Participants.
Article II
Definitions
2.1 Account. Account means a bookkeeping account maintained by the Committee to record the payment obligation of the Company to a Participant as determined under the terms of the Plan. The Committee may maintain an Account to record the total obligation to a Participant and component Accounts to reflect amounts payable at different times and in different forms. Subaccounts may be maintained for the purpose of tracking amount subject to different vesting schedules. Reference to an Account means any such Account established by the Committee, as the context requires.
2.2 Account Balance. Account Balance means, with respect to any Account, the total payment obligation owed to a Participant from such Account as of the most recent Valuation Date.
2.3 Beneficiary. Beneficiary means a natural person, estate, or trust designated by a Participant in accordance with Section 5.4 hereof to receive payments to which a Beneficiary is entitled in accordance with provisions of the Plan.
2.4 Board of Directors. Board of Directors means the Company’s board of directors.
2.5 Business Day. Business Day means each day on which the New York Stock Exchange is open for business.
2.6 Claimant. Claimant means a Participant or Beneficiary filing a claim under Article X of this Plan.
2.7 Code. Code means the Internal Revenue Code of 1986, as amended from time to time.
Page 1 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
2.8 Code Section 409A. Code Section 409A means section 409A of the Code, and regulations and other guidance issued by the Treasury Department and Internal Revenue Service thereunder.
2.9 Committee. Committee means the Company or a committee appointed by the Company to administer the Plan.
2.10 Company. Company means Integra LifeSciences Holdings Corporation.
2.11 Compensation. Compensation means a Participant’s director fees paid as RSUs and separate awards of RSUs. Compensation excludes any compensation that has been previously deferred under this Plan or any other arrangement subject to Code Section 409A.
2.12 Compensation Deferral Agreement. Compensation Deferral Agreement means an agreement between a Participant and the Company that specifies: (i) the amount of Compensation that the Participant has elected to defer to the Plan in accordance with the provisions of Article IV and (ii) the Payment Schedule applicable to the amounts deferred, indicated by an allocation of Deferrals to or among an established Separation Account, an In-Service Account or a Specified Year Account.
2.13 Deferral. Deferral means a credit to a Participant’s Account(s) that records that portion of the Participant’s Compensation that the Participant has elected to defer to the Plan in accordance with the provisions of Article IV. Unless the context of the Plan clearly indicates otherwise, a reference to Deferrals includes Earnings attributable to such Deferrals.
2.14 Director. Director means a non-employee member of the Board of Directors of the Company.
2.15 Earnings. Earnings means an adjustment to the dollar value of an Account based on the current market value of the Company’s common shares. Dividend equivalents paid on RSUs are not Earnings and are not deferred under this Plan.
2.16 Effective Date. Effective Date means April 1, 2026.
2.17 In-Service Account. In-Service Account means an Account established by the Committee to record the amounts payable on the earlier of a future calendar year as specified in the Participant’s Compensation Deferral Agreement and as set forth in Section 5.2 or the Participant’s Separation from Service as provided under Section 5.3.
2.18 Participant. Participant means an individual described in Article III.
2.19 Payment Schedule. Payment Schedule means the calendar year when payment of a Separation Account, In-Service Account or Specified Year Account will commence under Sections 5.2 or 5.3, as is applicable, and the form in which payment of such Account will be made, as specified in the Participant’s Compensation Deferral Agreement that establishes the Account.
2.20 Plan. Plan means “Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan” as documented herein and as may be amended from time to time
Page 2 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
hereafter. However, to the extent permitted or required under Code Section 409A, the term Plan may in the appropriate context also mean a portion of the Plan that is treated as a single plan under Treas. Reg. Section 1.409A-1(c), or the Plan or portion of the Plan and any other nonqualified deferred compensation plan or portion thereof that is treated as a single plan under such section.
2.21 Plan Year. Plan Year means the calendar year.
2.22 RSU RSU means a restricted stock unit granted to Directors under the terms of the Company’s equity compensation program(s), the settlement date of which is deferred under the terms of this Plan.
2.23 Separation Account. Separation Account means an Account established by the Committee in accordance with a Participant’s Compensation Deferral Agreement to record Deferrals allocated to such Account by the Participant and which are payable upon the Participant’s Separation from Service as set forth in Section 5.3.
2.24 Separation from Service. Separation from Service means a Director’s termination of service as a Director. Separation from Service shall be determined in accordance with Code Section 409A.
2.25 Specified Year Account. Specified Year Account means an Account established by the Committee to record the amounts payable in a future calendar year as specified in the Participant’s Compensation Deferral Agreement as set forth in Section 5.2. The Payment Schedule of a Specified Year Account is unaffected by a Participant’s earlier Separation from Service.
2.26 Valuation Date. Valuation Date means each Business Day.
Article III
Eligibility and Participation
3.1 Eligibility and Participation. All Directors may enroll in the Plan. Directors become Participants on the date on which the first Compensation Deferral Agreement becomes irrevocable under Article IV.
3.2 Duration. A Director shall remain a Participant as long as his or her Account Balance is greater than zero (0). An individual shall cease being a Participant in the Plan when his or her Account has been reduced to zero (0).
Article IV
Deferrals
Deferrals
4.1 Deferral Elections, Generally.
(a)A Director may make an initial election to defer Compensation by submitting a Compensation Deferral Agreement during the enrollment periods established by the Committee and in the manner specified by the Committee, but in any event, in accordance with Section 4.2. Unless an earlier date is specified in the Compensation Deferral Agreement, deferral elections with respect to
Page 3 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Compensation source become irrevocable on the latest date applicable to such Compensation source under Section 4.2.
(b)A Compensation Deferral Agreement that is not timely filed with respect to specified Compensation, or that is submitted by a Participant who experiences a Separation from Service prior to the latest date such agreement would become irrevocable under Section 409A, shall be deemed to have elected to make no Deferrals for the applicable Plan Year. The Committee may modify or revoke any Compensation Deferral Agreement prior to the date the election becomes irrevocable under the rules of Section 4.2.
(c)Unless otherwise specified by the Committee in the Compensation Deferral Agreement, Participants may defer a minimum of five percent (5%) and a maximum of one hundred percent (100%) of RSUs granted during the Plan Year.
Deferrals expressed as a percentage of a grant of RSUs will be rounded down to the nearest whole stock unit.
If a Director has the option to elect to receive a percentage of cash retainer as an RSU, such election need not be set forth in the Participant’s Compensation Deferral Agreement in which such RSUs are deferred but must be made in writing in the manner specified by the Committee in accordance with the election timing rules under Section 4.2 and is irrevocable at the same time as such Compensation Deferral Agreement.
(d)The Director shall specify on his or her Compensation Deferral Agreement the amount of Deferrals and whether to allocate Deferrals to his or her Separation Account, an In-Service Account or Specified Year Account. If no designation is made, Deferrals shall be allocated to the Participant’s Separation Account. In the Compensation Deferral Agreement in which the Participant establishes their first In-Service Account, he or she must also establish a Separation Account (if the Separation Account has not been established previously) and shall elect a Payment Schedule for such Separation Account, regardless of any Deferrals that may or may not be allocated to the Separation Account. If no Separation Account is established, the Participant will be deemed to have established a Separation Account that pays under Section 5.3 in a single lump sum.
4.2 Timing Requirements for Compensation Deferral Agreements.
(a)Initial Eligibility. The Committee may permit a Director to defer Compensation earned in the first year of eligibility, as determined in accordance with Treas. Reg. Section 1.409A-2(a)(7)(ii) and the “plan aggregation rules” provided in Treas. Reg. Section 1.409A-1(c)(2). The Compensation Deferral Agreement must be filed within 30 days after the Effective Date or, if later, within 30 days after being seated as a Director and becomes irrevocable not later than the 30th day.
A Compensation Deferral Agreement filed under this paragraph applies to Compensation related to service performed after the date on which the Compensation Deferral Agreement becomes irrevocable.
(b)Prior Year Election. Except as otherwise provided in this Section 4.2, the Committee may permit a Director to defer Compensation by filing a
Page 4 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Compensation Deferral Agreement no later than December 31 of the year next preceding the year in which RSU grants are made. A Compensation Deferral Agreement filed under this paragraph shall become irrevocable with respect to such Compensation not later than the December 31 next preceding the Plan Year in which such Compensation will be earned.
(c)Certain Forfeitable Rights. With respect to a legally binding right to a payment in a subsequent year that is subject to a forfeiture condition requiring the Director’s continued services for a period of at least 12 months from the date the Director obtains the legally binding right, the Committee may permit a Director to defer such Compensation by filing a Compensation Deferral Agreement in accordance with its rules and procedures, on or before the 30th day after the legally binding right to the Compensation accrues, provided that the Compensation Deferral Agreement is submitted at least 12 months in advance of the earliest date on which the forfeiture condition could lapse, as determined in accordance with Treas. Reg. Section 1.409A-2(a)(5). The Compensation Deferral Agreement described in this Section 4.2(c) becomes irrevocable not later than such 30th day after the Director obtains the legally binding right to the Compensation subject to such Compensation Deferral Agreement. If the forfeiture condition applicable to the payment lapses before the end of such 12-month period as a result of the Participant’s death or disability (as defined in Treas. Reg. Section 1.409A-3(i)(4)) or upon a change in control (as defined in Treas. Reg. Section 1.409A-3(i)(5)), the Compensation Deferral Agreement will be void unless it would be considered timely under another rule described in this Section.
(d)“Evergreen” Deferral Elections. The Committee, in its discretion, may provide that Compensation Deferral Agreements will continue in effect for subsequent years or performance periods by communicating that intention to Participants in writing prior to the date Compensation Deferral Agreements become irrevocable under this Section 4.2. An evergreen Compensation Deferral Agreement may be revoked or modified in writing prospectively by the Participant or the Committee with respect to Compensation for which such election remains revocable under this Section 4.2.
A Compensation Deferral Agreement is deemed to be revoked for subsequent years if the Participant is not a Director as of the last permissible date for making elections under this Section 4.2 or if the Compensation Deferral Agreement is cancelled in accordance with Section 4.3.
A Compensation Deferral Agreement is deemed to be revoked for subsequent years if the Participant is not a Director as of the last permissible date for making elections under this Section 4.2 or if the Compensation Deferral Agreement is cancelled in accordance with Section 4.3.
4.3 Cancellation of Deferrals. The Committee in its sole discretion, may cancel a Participant’s Deferrals: (i) for the balance of the Plan Year in which an Unforeseeable Emergency occurs, and (ii) during periods in which the Participant is unable to perform the duties of his or her position or any substantially similar position due to a mental or physical impairment that can be expected to result in death or last for a continuous period of at least six months, provided cancellation occurs by the later of the end of the taxable year of the Participant or the 15th day of the third month following the date the Participant incurs the disability (as defined in this clause (ii)).
Page 5 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Article V
Payments from Accounts
Payments from Accounts
5.1 General Rules. A Participant’s Account(s) become payable upon the first to occur of the payment events applicable to such Account under Sections 5.2 or 5.3, as elected in such Participant’s Compensation Deferral Agreement, or as set forth in Section 5.4.
References to an “election” means the Payment Schedule specified for an Account as designated in the Compensation Deferral Agreement that established such Account and which was made in accordance with Article IV or in a valid modification election applicable to such Account as described in Section 5.8.
Payment amounts are based on the vested Account Balances as of the first Valuation Date of the month in which actual payment will be made.
For purposes of Article V, Account Balance is determined as the number of stock units held in the applicable Account. Stock units will be paid in shares of stock as provided under the terms of the equity compensation program under which such deferred stock awards were made. Any fractional units will be paid in cash.
The vested portion of RSUs as of a payment event in this Article V is determined under the terms of the Company’s equity compensation program. Unvested RSUs shall be forfeited as provided in such program.
5.2 In-Service Accounts and Specified Year Accounts.
Commencement. An In-Service Account or Specified Year Account will be paid or commence payment in the third calendar year following the Plan Year in which such Specified Year Account is established, unless the Participant elected a later calendar year for such Account.
Form of Payment. Payment will be made in a lump sum, unless the Participant elected to receive an Account in a designated number of annual installments not to exceed five (5) installment payments.
Effect of Earlier Separation from Service. Notwithstanding the foregoing, a Participant’s In-Service Account(s) are payable under Section 5.3 in the event of the Participant’s Separation from Service prior to the elected payment commencement year for such Account(s).
The time and form of payment of a Specified Year Account is unaffected by the Participant’s earlier Separation from Service.
The time and form of payment of a Specified Year Account is unaffected by the Participant’s earlier Separation from Service.
5.3 Separation Account. Upon a Participant’s Separation from Service other than death, the Participant is entitled to receive the vested portion of his or her Separation Accounts and the vested portion of any In-Service Accounts having payment commencement years under Section 5.2 that occurs later than the calendar year in which the Participant’s Separation from Service occurs.
Commencement. A Participant’s Separation Account commences payment in the calendar year next following the calendar year in which Separation from Service occurs, unless the Participant elected a later calendar year for such Account.
Page 6 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Notwithstanding any other provision of this Plan, payment under this Section 5.3 to a Participant who is a “specified employee” as defined in Code Section 409A(a)(2)(B) will commence no earlier than the seventh month following the month of his or her Separation from Service.
Form of Payment. A Participant’s Separation Account will be paid in a single lump sum unless the Participant elected to receive such Account in a designated number of annual installments not to exceed five (5) installment payments.
In-Service Accounts. A Participant’s In-Service Account(s) payable under this Section 5.3 shall be paid at the same time and in the same form of payment that applies to the Participant’s Separation Account.
5.4 Death. Notwithstanding anything to the contrary in this Article V, upon the death of the Participant (regardless of whether such Participant is a Director at the time of death), all remaining vested Account Balances shall be paid to his or her Beneficiary in a single lump sum no later than December 31 of the calendar year following the year of the Participant’s death.
(a)Designation of Beneficiary in General. The Participant shall designate a Beneficiary in the manner and on such terms and conditions as the Committee may prescribe. No such designation shall become effective unless filed with the Committee during the Participant’s lifetime. Any designation shall remain in effect until a new designation is filed with the Committee; provided, however, that in the event a Participant designates his or her spouse as a Beneficiary, such designation shall be automatically revoked upon the dissolution of the marriage unless, following such dissolution, the Participant submits a new designation naming the former spouse as a Beneficiary. A Participant may from time to time change his or her designated Beneficiary without the consent of a previously-designated Beneficiary by filing a new designation with the Committee.
(b)No Beneficiary. If a designated Beneficiary does not survive the Participant, or if there is no valid Beneficiary designation, amounts payable under the Plan upon the death of the Participant shall be paid to the Participant’s spouse, or if there is no surviving spouse, then to the duly appointed and currently acting personal representative of the Participant’s estate.
5.5. Administrative Cash-Out of Small Balances. Notwithstanding anything to the contrary in this Article V, the Committee may at any time and without regard to whether a payment event has occurred, direct in writing (no later than the date of the payment) an immediate lump sum payment of the Participant’s Accounts if the balance of such Accounts, combined with any other amounts required to be treated as deferred under a single plan pursuant to Code Section 409A, does not exceed the applicable dollar amount under Code Section 402(g)(1)(B), provided any other such aggregated amounts are also distributed in a lump sum at the same time.
5.6 Acceleration of or Delay in Payments. Notwithstanding anything to the contrary in this Article V, the Committee, in its sole and absolute discretion, may elect to accelerate the time or form of payment of a vested Account, provided such acceleration is permitted under Treas. Reg. Section 1.409A-3(j)(4). The Committee may also, in its sole and
Page 7 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
absolute discretion, delay the time for payment of an Account, to the extent permitted under Treas. Reg. Section 1.409A-2(b)(7).
5.7 Rules Applicable to Installment Payments. If a Payment Schedule specifies annual installment payments, payments will be made commencing in the designated calendar year for the applicable payment under this Article V (as may be modified under Section 5.8) with subsequent installments paid in successive calendar years until the number of installment payments specified in the applicable Payment Schedule has been paid. The amount of each installment payment shall be determined by dividing (a) by (b), where (a) equals the vested Account Balance as of the first Valuation Date in the month actual payment will be made in accordance with this Article V and (b) equals the remaining number of annual installment payments. For purposes of Section 5.8, installment payments will be treated as a single payment. Accounts payable in installments will continue to be credited with Earnings in accordance with Article VI hereof until the Account is completely distributed.
5.8 Modifications to Payment Schedules. A Participant may modify the time or form of the Payment Schedule elected by him or her with respect to an Account, consistent with the permissible Payment Schedules available under the Plan for the applicable payment event, provided such modification complies with the requirements of this Section 5.8.
(a)Time of Election. The modification election must be submitted to the Committee not less than 12 months prior to the first day of the calendar year payments (or the initial payment) would have commenced under the Payment Schedule in effect prior to modification (the “Prior Election”).
(b)Date of Payment under Modified Payment Schedule. The calendar year in which payments are to commence under the modified Payment Schedule must be no earlier than the fifth calendar year after the calendar year payment would have commenced under the Prior Election. Under no circumstances may a modification election result in an acceleration of payments in violation of Code Section 409A. If the Participant modifies only the form, and not the commencement date for payment, payments shall commence in the fifth calendar year following the calendar year payment would have commenced under the Prior Election.
(c)Irrevocability; Effective Date. A modification election is irrevocable when filed and becomes effective 12 months after the filing date.
(d)Effect on Accounts. An election to modify a Payment Schedule is limited to the designated Account(s) and payment time or event to which such Payment Schedule applies and shall not be construed to affect any Payment Schedule for an alternative payment time or event applicable to such Account(s) or any Payment Schedule applicable to any other Account.
Article VI
Valuation of Account Balances
6.1 Valuation. Valuation of Accounts shall be performed under procedures approved by the Committee in its sole and absolute discretion, in accordance with this 0.
Deferrals of RSUs will be credited to a Participant’s Account in the designated number of units with each unit equal in value to one share of common stock of the Company.
Page 8 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Dividend equivalents payable with respect to deferred RSUs are not deferred under this Plan and shall be paid under the terms of the Company’s equity compensation plan.
Article VII
Administration
7.1Plan Administration. This Plan shall be administered by the Committee which shall have discretionary authority to make, amend, interpret and enforce all appropriate rules and regulations for the administration of this Plan and to utilize its discretion to decide or resolve any and all questions, including but not limited to eligibility for benefits and interpretations of this Plan and its terms, as may arise in connection with the Plan. Claims for benefits shall be filed with the Committee and resolved in accordance with the claims procedures in Article X. The Committee shall have the powers necessary to accomplish the Plans purposes, including, but not limited to, the following: (i) to select investment options available from time to time, (ii) to construe and interpret the terms and provisions of this Plan, (iii) to compute and certify to the amount and kind of benefits payable to Participants and their Beneficiaries, to determine the time and manner in which such benefits are paid, and to determine the amount of any withholding taxes to be deducted, (iv) to maintain all records that may be necessary for the administration of the Plan, (v) to provide for the disclosure of all information and the filing or provision of all reports and statements to Participants, Beneficiaries or governmental agencies as shall be required by law, (vi) to make and publish such rules and regulations of this Plan and procedures for the administration of this Plan as are not inconsistent with terms hereof, including method of bookkeeping for Accounts, (vii) to administer the claims procedure for this Plan, (viii) to approve election forms and procedures for use under this Plan, and (ix) to appoint a plan recordkeeper or any other agent, and to delete to the such powers and duties in connection with administration of this Plan as the Committee may from time to time prescribe.
7.2Administration Upon Change in Control. Upon a change in control affecting the Company, the Committee, as constituted immediately prior to such change in control, shall continue to act as the Committee. The Committee, by a vote of a majority of its members, shall have the authority (but shall not be obligated) to appoint an independent third party to act as the Committee. For purposes of this Section 7.2, a “change in control” means a change in control within the meaning of the rabbi trust agreement associated with the Plan or if no such definition is provided, the term shall have the meaning under Code Section 409A.
Upon such change in control, the Company may not remove the Committee or its members, unless a majority of Participants and Beneficiaries with Account Balances consent to the removal and replacement of the Committee. Notwithstanding the foregoing, the Committee shall not have authority to direct investment of trust assets under any rabbi trust described in Section 9.2.
The Company shall, with respect to the Committee identified under this Section: (i) pay all reasonable expenses and fees of the Committee, (ii) indemnify the Committee (including individuals serving as Committee members) against any costs, expenses and liabilities including, without limitation, attorneys’ fees and expenses arising in connection with the performance of the Committee’s duties hereunder, except with respect to matters resulting from the Committee’s gross negligence or willful misconduct, and (iii) supply
Page 9 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
full and timely information to the Committee on all matters related to the Plan, any rabbi trust, Participants, Beneficiaries and Accounts as the Committee may reasonably require.
7.3Withholding. The Company shall have the right to withhold from any payment due under the Plan (or with respect to any amounts credited to the Plan) any federal, state, and local income, employment and other taxes required by law to be withheld in respect of such payment (or credit). Withholdings with respect to amounts credited to the Plan shall be deducted from Compensation that has not been deferred to the Plan.
7.4Indemnification. The Company shall indemnify and hold harmless each employee, officer, director, agent or organization, to whom or to which are delegated duties, responsibilities, and authority under the Plan or otherwise with respect to administration of the Plan, including, without limitation, the Committee, its delegees and its agents, against all claims, losses, damages, liabilities, fines and penalties, and all expenses reasonably incurred by or imposed upon him or it (including but not limited to reasonable attorney fees) which arise as a result of or in connection with his or its actions or failure to act in connection with the operation and administration of the Plan to the extent lawfully allowable and to the extent that such claim, liability, fine, penalty, or expense is not paid for by liability insurance purchased or paid for by the Company. Notwithstanding the foregoing, the Company shall not indemnify any person or organization if his or its actions or failure to act are due to gross negligence or willful misconduct or for any such amount incurred through any settlement or compromise of any action unless the Company consents in writing to such settlement or compromise.
7.5Delegation of Authority. In the administration of this Plan, the Committee may, from time to time, employ agents and delegate to them such administrative duties as it sees fit, and may from time to time consult with legal counsel who shall be legal counsel to the Company.
7.6Binding Decisions or Actions. The decision or action of the Committee in respect of any question arising out of or in connection with the administration, interpretation and application of the Plan and the rules and regulations thereunder shall be final and conclusive and binding upon all persons having any interest in the Plan.
Article VIII
Amendment and Termination
Amendment and Termination
8.1Amendment and Termination. The Company may at any time and from time to time amend the Plan or may terminate the Plan as provided in this Article III.
8.2Amendments. The Company, by action taken by its Board of Directors, may amend or modify the Plan at any time and for any reason, in whole or in part, provided that any such amendment shall not reduce the vested Account Balances of any Participant accrued as of the date of any such amendment or restatement (as if the Participant had incurred a voluntary Separation from Service on such date). The Board of Directors of the Company may delegate to the Committee the authority to amend the Plan without the consent of the Board of Directors for the purpose of: (i) conforming the Plan to the requirements of law; (ii) facilitating the administration of the Plan; (iii) clarifying provisions based on the Committee’s interpretation of the Plan documents; and (iv) making such other amendments as the Board of Directors may authorize.
Page 10 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
8.3Termination. The Company, by action taken by its Board of Directors, may terminate the Plan and pay all Participants and Beneficiaries their Account Balances in a single lump sum at any time, to the extent and in accordance with Treas. Reg. Section 1.409A-3(j)(4)(ix).
8.4Accounts Taxable Under Code Section 409A. The Plan is intended to constitute a plan of deferred compensation that meets the requirements for deferral of income taxation under Code Section 409A. The Committee, pursuant to its authority to interpret the Plan, may sever from the Plan or any Compensation Deferral Agreement any provision or exercise of a right that otherwise would result in a violation of Code Section 409A.
Article IX
Informal Funding
Informal Funding
9.1General Assets. Obligations established under the terms of the Plan may be satisfied from the general funds of the Company or a trust described in this Article IX. No Participant, spouse or Beneficiary shall have any right, title or interest whatever in assets of the Company. Nothing contained in this Plan, and no action taken pursuant to its provisions, shall create or be construed to create a trust of any kind, or a fiduciary relationship, between the Company and any Director, spouse, or Beneficiary. To the extent that any person acquires a right to receive payments hereunder, such rights are no greater than the right of an unsecured general creditor of the Company.
9.2Rabbi Trust. The Company may, in its sole discretion, establish a grantor trust, commonly known as a rabbi trust, as a vehicle for accumulating assets to pay benefits under the Plan. Payments under the Plan may be paid from the general assets of the Company or from the assets of any such rabbi trust. Payment from any such source shall reduce the obligation owed to the Participant or Beneficiary under the Plan.
Article X
Claims
Claims
10.1Filing a Claim. Any controversy or claim arising out of or relating to the Plan shall be filed in writing with the Committee which shall make all determinations concerning such claim. Any claim filed with the Committee and any decision by the Committee denying such claim shall be in writing and shall be delivered to the Participant or Beneficiary filing the claim (the “Claimant”). Notice of a claim for payments shall be delivered to the Committee within 90 days of the latest date upon which the payment could have been timely made in accordance with the terms of the Plan and Code Section 409A, and if not paid, the Participant or Beneficiary must file a claim under this Article X not later than 180 days after such latest date. If the Participant or Beneficiary fails to file a timely claim, the Participant forfeits any amounts to which he or she may have been entitled to receive under the claim.
(a)In General. Notice of a denial of benefits will be provided within 90 days of the Committee’s receipt of the Claimant’s claim for benefits. If the Committee determines that it needs additional time to review the claim, the Committee will provide the Claimant with a notice of the extension before the end of the initial 90-day period. The extension will not be more than 90 days from the end of the initial 90-day period and the notice of extension will explain the special
Page 11 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
circumstances that require the extension and the date by which the Committee expects to make a decision.
(b)Contents of Notice. If a claim for benefits is completely or partially denied, notice of such denial shall be in writing. The notice of denial shall set forth the specific reasons for denial in plain language. The notice shall: (i) cite the pertinent provisions of the Plan document, and (ii) explain, where appropriate, how the Claimant can perfect the claim, including a description of any additional material or information necessary to complete the claim and why such material or information is necessary. The claim denial also shall include an explanation of the claims review procedures and the time limits applicable to such procedures, including the right to appeal the decision, the deadline by which such appeal must be filed and a statement of the Claimant’s right to bring a civil action and the specific date by which such a civil action must commence under Section 10.4.
10.2Appeal of Denied Claims. A Claimant whose claim has been completely or partially denied shall be entitled to appeal the claim denial by filing a written appeal with a committee designated to hear such appeals (the “Appeals Committee”). A Claimant who timely requests a review of the denied claim (or his or her authorized representative) may review, upon request and free of charge, copies of all documents, records and other information relevant to the denial and may submit written comments, documents, records and other information relating to the claim to the Appeals Committee. All written comments, documents, records, and other information shall be considered “relevant” if the information: (i) was relied upon in making a benefits determination, (ii) was submitted, considered or generated in the course of making a benefits decision regardless of whether it was relied upon to make the decision, or (iii) demonstrates compliance with administrative processes and safeguards established for making benefit decisions. The review shall consider all comments, documents, records, and other information submitted by the Claimant relating to the claim, without regard to whether such information was submitted or considered in the initial benefit determination. The Appeals Committee may, in its sole discretion and if it deems appropriate or necessary, decide to hold a hearing with respect to the claim appeal.
(a)In General. Appeal of a denied benefits claim must be filed in writing with the Appeals Committee no later than 60 days after receipt of the written notification of such claim denial. The Appeals Committee shall make its decision regarding the merits of the denied claim within 60 days following receipt of the appeal (or within 120 days after such receipt, in a case where there are special circumstances requiring extension of time for reviewing the appealed claim). If an extension of time for reviewing the appeal is required because of special circumstances, written notice of the extension shall be furnished to the Claimant prior to the commencement of the extension. The notice will indicate the special circumstances requiring the extension of time and the date by which the Appeals Committee expects to render the determination on review. The review will consider comments, documents, records and other information submitted by the Claimant relating to the claim without regard to whether such information was submitted or considered in the initial benefit determination.
(b)Contents of Notice. If a benefits claim is completely or partially denied on review, notice of such denial shall be in writing. Such notice shall set forth the reasons for denial in plain language.
Page 12 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
The decision on review shall set forth: (i) the specific reason or reasons for the denial, (ii) specific references to the pertinent Plan provisions on which the denial is based, (iii) a statement that the Claimant is entitled to receive, upon request and free of charge, reasonable access to and copies of all documents, records, or other information relevant (as defined above) to the Claimant’s claim, and (iv) a statement of the Claimant’s right to bring an action following an adverse decision on review and the specific date by which such a civil action must commence under Section 10.4.
10.3Claims Appeals Upon Change in Control. Upon a change in control, the Appeals Committee, as constituted immediately prior to such change in control, shall continue to act as the Appeals Committee. The Company may not remove any member of the Appeals Committee but may replace resigning members if 2/3rds of the members of the Board of Directors of the Company and a majority of Participants and Beneficiaries with Account Balances consent to the replacement. For purposes of this Section 10.3, a “change in control” means a change in control within the meaning of the rabbi trust agreement associated with the Plan or if no such definition is provided, the term shall have the meaning under Code Section 409A.
The Appeals Committee shall have the exclusive authority at the appeals stage to interpret the terms of the Plan and resolve appeals under the Claims Procedure.
The Company shall, with respect to the Committee identified under this Section: (i) pay all reasonable expenses and fees of the Appeals Committee, (ii) indemnify the Appeals Committee (including individual committee members) against any costs, expenses and liabilities including, without limitation, attorneys’ fees and expenses arising in connection with the performance of the Appeals Committee hereunder, except with respect to matters resulting from the Appeals Committee’s gross negligence or willful misconduct, and (iii) supply full and timely information to the Appeals Committee on all matters related to the Plan, any rabbi trust, Participants, Beneficiaries and Accounts as the Appeals Committee may reasonably require.
10.4Legal Action. A Claimant may not bring any legal action, including commencement of any arbitration, relating to a claim for benefits under the Plan unless and until the Claimant has followed the claims procedures under the Plan and exhausted his or administrative remedies under Sections 10.1 and 10.2. No such legal action may be brought more than twelve (12) months following the notice of denial of benefits under Section 10.2, or if no appeal is filed by the applicable appeals deadline, twelve (12) months following the appeals deadline.
If a Participant or Beneficiary prevails in a legal proceeding brought under the Plan to enforce the rights of such Participant or any other similarly situated Participant or Beneficiary, in whole or in part, the Company shall reimburse such Participant or Beneficiary for all legal costs, expenses, attorneys’ fees and such other liabilities incurred as a result of such proceedings. If the legal proceeding is brought in connection with a change in control as defined in Section 10.3, the Participant or Beneficiary may file a claim directly with the trustee for reimbursement of such costs, expenses and fees. For purposes of the preceding sentence, the amount of the claim shall be treated as if it were an addition to the Participant’s or Beneficiary’s Account Balance.
Page 13 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
10.5Discretion of Appeals Committee. All interpretations, determinations and decisions of the Appeals Committee with respect to any claim shall be made in its sole discretion and shall be final and conclusive.
10.6Arbitration.
(a)Prior to Change in Control. If, prior to a change in control as defined in Section 10.3, any claim or controversy between the Company and a Participant or Beneficiary is not resolved through the claims procedure set forth in Article X, such claim shall be submitted to and resolved exclusively by expedited binding arbitration by a single arbitrator. Arbitration shall be conducted in accordance with the following procedures:
The complaining party shall promptly send written notice to the other party identifying the matter in dispute and the proposed remedy. Following the giving of such notice, the parties shall meet and attempt in good faith to resolve the matter. In the event the parties are unable to resolve the matter within 21 days, the parties shall meet and attempt in good faith to select a single arbitrator acceptable to both parties. If a single arbitrator is not selected by mutual consent within ten Business Days following the giving of the written notice of dispute, an arbitrator shall be selected from a list of nine persons each of whom shall be an attorney who is either engaged in the active practice of law or recognized arbitrator and who, in either event, is experienced in serving as an arbitrator in disputes between employers and employees, which list shall be provided by the main office of either JAMS, the American Arbitration Association (“AAA”) or the Federal Mediation and Conciliation Service. If, within three Business Days of the parties’ receipt of such list, the parties are unable to agree on an arbitrator from the list, then the parties shall each strike names alternatively from the list, with the first to strike being determined by the flip of a coin. After each party has had four strikes, the remaining name on the list shall be the arbitrator. If such person is unable to serve for any reason, the parties shall repeat this process until an arbitrator is selected.
Unless the parties agree otherwise, within 60 days of the selection of the arbitrator, a hearing shall be conducted before such arbitrator at a time and a place agreed upon by the parties. In the event the parties are unable to agree upon the time or place of the arbitration, the time and place shall be designated by the arbitrator after consultation with the parties. Within 30 days of the conclusion of the arbitration hearing, the arbitrator shall issue an award, accompanied by a written decision explaining the basis for the arbitrator’s award.
In any arbitration hereunder, the Company shall pay all administrative fees of the arbitration and all fees of the arbitrator, except that the Participant or Beneficiary may, if he/she/it wishes, pay up to one-half of those amounts. Each party shall pay its own attorneys’ fees, costs, and expenses, unless the arbitrator orders otherwise. The prevailing party in such arbitration, as determined by the arbitrator, and in any enforcement or other court proceedings, shall be entitled, to the extent permitted by law, to reimbursement from the other party for all of the prevailing party’s costs (including but not limited to the arbitrator’s compensation), expenses, and attorneys’ fees. The arbitrator shall have no authority to add to or to modify this Plan, shall apply all applicable law, and shall have no lesser and no
Page 14 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
greater remedial authority than would a court of law resolving the same claim or controversy. The arbitrator shall, upon an appropriate motion, dismiss any claim without an evidentiary hearing if the party bringing the motion establishes that it would be entitled to summary judgment if the matter had been pursued in court litigation.
The parties shall be entitled to discovery as follows: Each party may take no more than three depositions. The Company may depose the Participant or Beneficiary plus two other witnesses, and the Participant or Beneficiary may depose the Company, pursuant to Rule 30(b)(6) of the Federal Rules of Civil Procedure, plus two other witnesses. Each party may make such reasonable document discovery requests as are allowed in the discretion of the arbitrator.
The decision of the arbitrator shall be final, binding, and non-appealable, and may be enforced as a final judgment in any court of competent jurisdiction.
This arbitration provision of the Plan shall extend to claims against any parent, subsidiary, or affiliate of each party, and, when acting within such capacity, any officer, director, shareholder, Participant, Beneficiary, or agent of any party, or of any of the above, and shall apply as well to claims arising out of state and federal statutes and local ordinances as well as to claims arising under the common law or under this Plan.
Notwithstanding the foregoing, and unless otherwise agreed between the parties, either party may apply to a court for provisional relief, including a temporary restraining order or preliminary injunction, on the ground that the arbitration award to which the applicant may be entitled may be rendered ineffectual without provisional relief.
Any arbitration hereunder shall be conducted in accordance with the Federal Arbitration Act: provided, however, that, in the event of any inconsistency between the rules and procedures of the Act and the terms of this Plan, the terms of this Plan shall prevail.
If any of the provisions of this Section 10.6(a) are determined to be unlawful or otherwise unenforceable, in the whole part, such determination shall not affect the validity of the remainder of this section and this section shall be reformed to the extent necessary to carry out its provisions to the greatest extent possible and to insure that the resolution of all conflicts between the parties, including those arising out of statutory claims, shall be resolved by neutral, binding arbitration. If a court should find that the provisions of this Section 10.6(a) are not absolutely binding, then the parties intend any arbitration decision and award to be fully admissible in evidence in any subsequent action, given great weight by any finder of fact and treated as determinative to the maximum extent permitted by law.
The parties do not agree to arbitrate any putative class action or any other representative action. The parties agree to arbitrate only the claims(s) of a single Participant or Beneficiary.
(b)Upon Change in Control. Upon a change in control as defined in Section 10.3, Section 10.6(a) shall not apply and any legal action initiated by a Participant or
Page 15 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
Beneficiary to enforce his or her rights under the Plan may be brought in any court of competent jurisdiction. Notwithstanding the Appeals Committee’s discretion under Sections 10.3 and 10.5, the court shall apply a de novo standard of review to any prior claims decision under Sections 10.1 through 10.3 or any other determination made by the Company, its Board of Directors, the Committee, or the Appeals Committee.
Article XI
General Provisions
General Provisions
11.1Assignment. No interest of any Participant, spouse or Beneficiary under this Plan and no benefit payable hereunder shall be assigned as security for a loan, and any such purported assignment shall be null, void and of no effect, nor shall any such interest or any such benefit be subject in any manner, either voluntarily or involuntarily, to anticipation, sale, transfer, assignment or encumbrance by or through any Participant, spouse or Beneficiary. Notwithstanding anything to the contrary herein, however, the Committee has the discretion to make payments to an alternate payee in accordance with the terms of a domestic relations order (as defined in Code Section 414(p)(1)(B)).
The Company may assign any or all of its liabilities under this Plan in connection with any restructuring, recapitalization, sale of assets or other similar transactions affecting the Company without the consent of the Participant.
11.2No Legal or Equitable Rights or Interest. No Participant or other person shall have any legal or equitable rights or interest in this Plan that are not expressly granted in this Plan. Participation in this Plan does not give any person any right to be retained in the service of the Company. The Company makes no representations or warranties as to the tax consequences to a Participant or a Participant’s Beneficiaries resulting from a deferral of income pursuant to the Plan.
11.3No Employment Contract. Nothing contained herein shall be construed to constitute a contract of employment between a Director and the Company. Nothing in this Plan shall be deemed to give a Participant the right to be retained in the service of the Company or to interfere with the right of the Company to discipline or discharge the Participant at any time.
11.4Notice. Any notice or filing required or permitted to be delivered to the Committee under this Plan shall be delivered in writing, in person, or through such electronic means as is established by the Committee. Notice shall be deemed given as of the date of delivery or, if delivery is made by mail, as of the date shown on the postmark on the receipt for registration or certification. Written transmission shall be sent by certified mail to:
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
1000 CAMPUS ROAD
PRINCETON, NJ 08540
ATTN: HUMAN RESOURCES
1000 CAMPUS ROAD
PRINCETON, NJ 08540
ATTN: HUMAN RESOURCES
Any notice or filing required or permitted to be given to a Participant under this Plan shall be sufficient if in writing or hand-delivered or sent by mail to the last known address of the Participant.
Page 16 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
11.5Headings. The headings, sections, and paragraphs are included solely for convenience of reference, and if there is any conflict between such headings and the text of this Plan, the text shall control.
11.6Invalid or Unenforceable Provisions. If any provision of this Plan shall be held invalid or unenforceable, such invalidity or unenforceability shall not affect any other provisions hereof and the Committee may elect in its sole discretion to construe such invalid or unenforceable provisions in a manner that conforms to applicable law or as if such provisions, to the extent invalid or unenforceable, had not been included.
11.7Facility of Payment to a Minor. If a distribution is to be made to a minor, or to a person who is otherwise incompetent, then the Committee may, in its discretion, make such distribution: (i) to the legal guardian, or if none, to a parent of a minor payee with whom the payee maintains his or her residence, or (ii) to the conservator or committee or, if none, to the person having custody of an incompetent payee. Any such distribution shall fully discharge the Committee, the Company, and the Plan from further liability on account thereof.
11.8Governing Law. The laws of the State of New Jersey shall govern the construction and administration of the Plan.
11.9Compliance With Code Section 409A; No Guarantee. This Plan is intended to be administered in compliance with Code Section 409A and each provision of the Plan shall be interpreted consistent with Code Section 409A. Although intended to comply with Code Section 409A, this Plan shall not constitute a guarantee to any Participant or Beneficiary that the Plan in form or in operation will result in the deferral of federal or state income tax liabilities or that the Participant or Beneficiary will not be subject to the additional taxes imposed under Section 409A. The Company has no legal obligation to a Participant with respect to taxes imposed under Code Section 409A.
11.10Successors. The provisions of this Plan shall bend and inure to the benefit of the Company and its successors and assigns and the Participant and the Participant’s Beneficiaries.
Page 17 of 20
Integra LifeSciences Holdings Corporation Directors Deferred Compensation Plan
IN WITNESS WHEREOF, the undersigned executed this Plan as of the 18th day of February, 2026, to be effective as of the Effective Date.
INTEGRA LIFESCIENCES HOLDINGS CORPORATION
By: Mojdeh Poul_______________________ (Print Name)
Its: President and Chief Executive Officer____ (Title)
/s/ Mojdeh Poul________________________ (Signature)
Page 1 of 20
IART-2025.12.31-Exhibit 21.1
Subsidiaries of Integra LifeSciences Holdings Corporation
| Name of Subsidiary | State or Country of Incorporation or Organization | |||||||
| Acclarent, Inc. | Delaware | |||||||
| ACell, Inc. | Delaware | |||||||
| Arkis Biosciences Inc. | Delaware | |||||||
| Ascension Orthopedics Limited | United Kingdom | |||||||
| BIMECO, Inc. | Florida | |||||||
| BioD, LLC | Delaware | |||||||
| BioDlogics, LLC | Delaware | |||||||
| BioRecovery, LLC | Delaware | |||||||
| CardioDyne, Inc. | Massachusetts | |||||||
| Cathtec Incorporated | Massachusetts | |||||||
| Caveangle Limited | United Kingdom | |||||||
| Confluent Surgical, Inc. | Delaware | |||||||
| Derma First Aid Products, Inc. | Pennsylvania | |||||||
| Derma Sciences Europe Limited | United Kingdom | |||||||
| Derma Sciences, Inc. | Delaware | |||||||
| EndoSolutions, Inc. | Delaware | |||||||
| Fiber Imaging Technologies, Inc. | Massachusetts | |||||||
| GMS, Gesellschaft für medizinische Sondentechnik mbH | Germany | |||||||
| ILS Financing (Ireland) Limited | Ireland | |||||||
| ILS Financing Corporation | Delaware | |||||||
| ILS Services Switzerland Ltd. | Switzerland | |||||||
| ILS Surgical Investments, LLC | Delaware | |||||||
| INS Sweden AB | Sweden | |||||||
| Integra Burlington MA, Inc. (formerly known as Integra Radionics, Inc.) | Delaware | |||||||
| Integra Canada ULC (formerly known as Canada Microsurgical ULC) | Canada | |||||||
| Integra CI, Inc. | Cayman Islands | |||||||
| Integra Euro Holdings, Inc. | Delaware | |||||||
| Integra France Holdings SAS | France | |||||||
| Integra German Holdings GmbH | Germany | |||||||
| Integra GmbH | Germany | |||||||
| Integra Japan K.K. | Japan | |||||||
| Integra LifeSciences (Canada) Holdings, Inc. | Delaware | |||||||
| Integra LifeSciences (Ireland) Limited | Ireland | |||||||
| Integra LifeSciences (Shanghai) Co., Ltd. | China | |||||||
| Integra LifeSciences (Suzhou) Co., Ltd. | China | |||||||
| Integra LifeSciences (Thailand) Limited | Thailand | |||||||
| Integra LifeSciences Austria GmbH | Austria | |||||||
| Integra LifeSciences Brazil Ltda. | Brazil | |||||||
| Integra LifeSciences Corporation | Delaware | |||||||
| Integra LifeSciences Enterprises, LLLP | Delaware | |||||||
| Integra LifeSciences Financing (Cyprus) Limited | Cyprus | |||||||
| Integra LifeSciences Israel Ltd. | Israel | |||||||
| Integra LifeSciences Italy S.r.l. | Italy | |||||||
| Integra LifeSciences Korea Ltd. | Korea | |||||||
| Integra LifeSciences Middle East FZ-LLC | Dubai | |||||||
| Integra LifeSciences Production Corporation | Delaware | |||||||
| Integra LifeSciences Sales LLC (f/k/a Integra Healthcare Products LLC) | Delaware | |||||||
| Integra LifeSciences Services (France) SAS | France | |||||||
| Integra LifeSciences Shared Services (Ireland) Limited | Ireland | |||||||
| Integra LifeSciences Singapore Pte. Ltd. | Singapore | |||||||
| Integra LifeSciences Spain, S.L. | Spain | |||||||
| Integra LifeSciences Switzerland Sárl | Switzerland | |||||||
| Integra LifeSciences Taiwan Company Limited | Taiwan | |||||||
| Integra LS (Benelux) NV | Belgium | |||||||
| Integra LS Mexico, S. DE R. L. DE C.V. | Mexico | |||||||
| Integra Luxtec, Inc. | Massachusetts | |||||||
| Integra ME GmbH | Germany | |||||||
| Integra Medical Devices India Private Limited | India | |||||||
| Integra MicroFrance SAS | France | |||||||
| Integra NeuroSciences (International), Inc. | Delaware | |||||||
| Integra NeuroSciences Holdings (UK) Limited | United Kingdom | |||||||
| Integra NeuroSciences Holdings B.V. | Netherlands | |||||||
| Integra NeuroSciences Implants (France) SAS | France | |||||||
| Integra NeuroSciences Limited | United Kingdom | |||||||
| Integra Neurosciences Pty Ltd. (AUS) | Australia | |||||||
| Integra Neurosciences Pty Ltd. (NZ) | New Zealand | |||||||
| Integra Receivables LLC | Delaware | |||||||
| Integra Sales, Inc. | Delaware | |||||||
| Integra Selector LLC | Delaware | |||||||
| Integra Switzerland Holdings Sárl | Switzerland | |||||||
| Integra York PA, Inc. (formerly known as Miltex, Inc.) | Delaware | |||||||
| IsoTis NV | Netherlands | |||||||
| IsoTis T.E. Facility B.V. | Netherlands | |||||||
| J. Jamner Surgical Instruments, Inc. | Delaware | |||||||
| Jarit GmbH | Germany | |||||||
| LXU Healthcare, Inc. - Medical Specialty Products | Delaware | |||||||
| MedEfficiency, Inc. | Delaware | |||||||
| Minnesota Scientific, Inc. | Minnesota | |||||||
| Newdeal SAS | France | |||||||
| Newdeal, Inc. | Texas | |||||||
| Precise Dental Holding Corp. | New Jersey | |||||||
| Precise Dental Internacional, S.A. de C.V. | Mexico | |||||||
| Precise Dental Products, Ltd. | California | |||||||
| Precision Dental International, Inc. | California | |||||||
| Rebound Therapeutics Corporation | Delaware | |||||||
| Spembly Cryosurgery Limited | United Kingdom | |||||||
| Spembly Medical Limited | United Kingdom | |||||||
| Surgical Innovation Associates, Inc. | Delaware | |||||||
| Tarsus Medical Inc. | Delaware | |||||||
| TEI Biosciences (UK) Limited | United Kingdom | |||||||
| TEI Biosciences Inc. | Delaware | |||||||
| TEI Medical Inc. | Delaware | |||||||
| TGX Medical Systems, LLC | Delaware | |||||||
IART-2025.12.31-EXHIBIT 23.1
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We hereby consent to the incorporation by reference in the Registration Statements on Form S-8 (Nos. 333-231709, 333-221210, 333-216212, 333-170210, 333-155263, 333-127488, 333-109042, 333-261744, 333-266353, 333-282990, and 333-289145) of Integra LifeSciences Holdings Corporation of our report dated February 26, 2026 , relating to the financial statements and the effectiveness of internal control over financial reporting, which appears in this Form 10-K.
/s/ PricewaterhouseCoopers LLP
Florham Park, New Jersey
February 26, 2026
Exhibit 31.1
Certification of Principal Executive Officer
Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Mojdeh Poul, certify that:
1.I have reviewed this annual report on Form 10-K of Integra LifeSciences Holdings Corporation;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and we have:
a.designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b.designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c.evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d.disclosed in this report any change in the registrant's internal control over financial reporting that occurred during the registrant's most recent fiscal quarter (the registrant's fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant's internal control over financial reporting; and
5. The registrant's other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a.all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b.any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
| Date: | February 26, 2026 | /s/ Mojdeh Poul | ||||||
| Mojdeh Poul | ||||||||
| President and Chief Executive Officer | ||||||||
Exhibit 31.2
Certification of Principal Financial Officer
Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Lea Knight, certify that:
1.I have reviewed this annual report on Form 10-K of Integra LifeSciences Holdings Corporation;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and we have:
a.designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b.designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c.evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d.disclosed in this report any change in the registrant's internal control over financial reporting that occurred during the registrant's most recent fiscal quarter (the registrant's fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant's internal control over financial reporting; and
5.The registrant's other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a.all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b.any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
| Date: | February 26, 2026 | /s/ Lea Knight | ||||||
| Lea Knight | ||||||||
| Executive Vice President and Chief Financial Officer | ||||||||
Exhibit 32.1
Certification of Principal Executive Officer
Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of Integra LifeSciences Holdings Corporation (the “Company”) on Form 10-K for the year ended December 31, 2025 as filed with the Securities Exchange Commission on the date hereof (the “Report”), I, Mojdeh Poul, President and Chief Executive Officer of the Company, hereby certify, pursuant to 18 U.S.C. §1350, as adopted pursuant to §906 of the Sarbanes-Oxley Act of 2002, that to the best of my knowledge:
1.The Report fully complies with the requirement of Section 13(a) or Section 15(d), as applicable, of the Securities Exchange Act of 1934, as amended; and
2.The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
| Date: | February 26, 2026 | /s/ Mojdeh Poul | ||||||
| Mojdeh Poul | ||||||||
| President and Chief Executive Officer | ||||||||
Exhibit 32.2
Certification of Principal Financial Officer
Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of Integra LifeSciences Holdings Corporation (the “Company”) on Form 10-K for the year ended December 31, 2025 as filed with the Securities Exchange Commission on the date hereof (the “Report”), I, Lea Knight, Executive Vice President and Chief Financial Officer of the Company, hereby certify, pursuant to 18 U.S.C. §1350, as adopted pursuant to §906 of the Sarbanes-Oxley Act of 2002, that to the best of my knowledge:
1.The Report fully complies with the requirement of Section 13(a) or Section 15(d), as applicable, of the Securities Exchange Act of 1934, as amended; and
2.The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
| Date: | February 26, 2026 | /s/ Lea Knight | ||||||
| Lea Knight | ||||||||
| Executive Vice President and Chief Financial Officer | ||||||||